A REVIEW ON FAST DISSOLVING FILMS FOR BUCCAL DELIVERY OF LOW DOSE DRUGS
HTML Full TextA REVIEW ON FAST DISSOLVING FILMS FOR BUCCAL DELIVERY OF LOW DOSE DRUGS
Tuhina Banerjee *, Vaseem A. Ansari, Satyaprakash Singh, Tarique Mahmood and Juber Akhtar
Faculty of Pharmacy, Integral University, Kursi Road, Lucknow - 226026, Uttar Pradesh, India.
ABSTRACT: Fast-dissolving drug-delivery systems were first developed in the late 1970s as an alternative to tablets, capsules, and syrups for pediatric and geriatric patients who experience difficulties swallowing traditional oral solid- dosage forms. Fast dissolving films have become a novel approach to oral drug delivery system as it provides convenience and ease of use over other dosage forms such as orally disintegrating tablets, buccal tablets, and sublingual tablets, so mouth dissolving films are gaining the interest of a large number of pharmaceutical industries. Mouth dissolving films are thin solid dosage forms which, when placed in the oral cavity, dissolve within a few seconds without chewing and intake of water. The oral, buccal mucosa being highly vascularized, drugs can absorb directly and can enter the systemic circulation without undergoing first-pass hepatic metabolism. This advantage can be exploited in preparing products with improved oral bioavailability of molecules that undergo first pass effect. These films offer a convenient way of dosing medication pediatric, geriatric, and bedridden patients. The sublingual and buccal delivery of a drug via thin film has the potential to improve the onset of action, lower the dosing, and enhance the efficacy and safety profile of medicament. An ideal film should have properties like pleasant taste, high stability, and ease of handling. The present review provides an account of various formulation methods and their evaluation used in film formulations and applications of mouth dissolving films.
Keywords: Fast disintegrating, Site-specific, Polymer, Plasticizer, Solvent casting, Solid dispersion
INTRODUCTION: Among the various routes, oral administration is the most preferred route. Most of the drugs are taken orally in the form of tablets, capsules, etc. by all patients, including adult, pediatric, and geriatric patients.
But these dosage forms have to face many problems such as-
- Need for water for their disintegration.
- Choking problems
- Poor patient compliance
- Unpleasant taste and odor
- Difficult to administer in children, aged people, mental patients, in unconscious states, 1
These problems are overcome by the development of other dosage forms such as fast dissolving films which are devoid of such problems. Fast dissolving film is a thin film with an area of 5-20 sq.cm containing an active pharmaceutical ingredient 2. Fast dissolving oral films (FDOFs) are the most advanced form of solid oral dosage form due to their flexibility and comfort. It improves the efficacy of APIs by getting dissolved within a few minutes in the oral cavity after coming in contact with saliva, without chewing and no need for water for administration.
It gives quick onset of action, long onset time, and instant bioavailability of drugs due to high blood flow and permeability of oral mucosa. FDOFs are useful in patients such as pediatrics, geriatrics, bedridden, emetic patients, diarrhea, sudden episode of allergic attacks, or coughing for those who have an active life style. It is also useful where local action is desired, such as local anesthetic for toothaches, oral ulcers, cold sores, or teething. FDFs also have an established shelf-life of 2-3 years, depending on the API but are extremely sensitive to environmental moisture. The special features of fast dissolving films are- Thin elegant, various sizes and shapes, un-obstructive, mucoadhesion, fast disintegration, quick dissolving, rapid release, etc. 3
Advantages: 4 The advantages of fast dissolving films are-
- Administered without water, anywhere, any time.
- Due to the larger surface area, provides rapid disintegration and dissolution in the oral cavity.
- Dose accuracy.
- The acidic environment of the stomach can be avoided.
- Site-specific action and local action.
- Flexible and portable so provides ease in transportation during consumer handling and storage.
- Suitable for geriatric and pediatric patients, who experience difficulties in swallowing, mentally ill, the developmentally disable and the patients who are uncooperative, or are on the reduced liquid intake or are nauseated.
- Beneficial in motion sickness, acute pain, allergic attack, or coughing, where the rapid onset of action is required.
- Stable for a longer duration of time, since the drug remains in solid dosage form until it is consumed.
- The oral or buccal mucosa is highly vascularized, drugs get absorbed directly and enter the systemic circulation without undergoing first‐pass hepatic metabolism.
- The sublingual and buccal delivery of a drug via film has the potential to improve the onset of action, lower the dosing, and enhance the efficacy and safety profile of the medicament.
- Provide new business opportunities like product differentiation, product promotion and patent extension 5.
Disadvantages: High dose cannot be incorporated, expensive packaging is required, dose uniformity is a technical challenge 6.
Common Ingredients:
Active Pharmaceutical Ingredient (1-25%): Variety of APIs can be delivered through fast dissolving films. Potent drugs are the best candidates for incorporation in fast dissolving film. It is useful to use micronized API as it improves the texture of the film and also results in better dissolution and uniformity in the fast dissolving film. Many APIs, which are potential candidates for fast dissolving film technology, has a bitter taste which makes the formulation unpalatable, especially for pediatric preparations.
Thus before incorporating such APIs in fast dissolving films, the taste masking is an essential step. Various methods can be used to improve the palatability of the formulation, but the simplest method is mixing and co-processing of bitter tasting API with flavoring or sweetening agent 7. Some of the examples of suitable API that can be incorporated in the FDFs are given in the Table 1.
Film Forming Polymer (40-50%): To prepare a film formulation that is water-soluble, excipients or polymer must be water soluble with low molecular weight and excellent film-forming capacity. The polymer that is to be used should be non-toxic, non-irritant, and devoid of leachable impurities. It should have good wetting and spreadability property. It should exhibit sufficient peel, shear, and tensile strengths. It should be readily available and should not be very expensive. Some of the examples of suitable polymers that can be incorporated in the FDFs are HPMC, CMC, gelatin, pullulan, etc. 9
TABLE 1: SOME SUITABLE DRUG CANDIDATES 8
| Category of drugs | Examples |
| Selective serotonin reuptake inhibitors | Fluoxetine, Sertraline, Paroxetine, Fluvoxamine, Citalopram, and
Alaproclate |
| Anti-emetics | Ondansetron, Granisetron, Palonosetron, Dronabinol, Aprepitant, Ramosetron, Metopimazine, Nabilone, Tropisetron, Metoclopramide, Prochlorperazine, Trimethobenzamide, Dimenhydrinate, Prochlorperazine and Dolasetron |
| 5HT3 antagonists | Alosetron, Ondansetron, Granisetron, Palonosetron, Ramosetron and Tropisetron |
| Anti-epileptics | Carbamazepine, Clonazepam, Diazepam, Divalproex Sodium, Fosphenyloin, Gabapentin, Lamotrigine, Levetiracetam, Oxcarbazepine, Phenyloin, Pregabalin, Primidone, Tiagabine, Topiramate, Valproate sodium , Vigabatrin and Zonisamide |
| Anti-migrains | Almotriptan, Dihydroergotamine Mesylate, Eletriptan, Frovatriptan, Naratriptan, Rizatriptan, Sumatriptan and Zolmitriptan |
| Dopamine D1 and D2 antagonists | Amisulpride, Bromperidol, Cabergoline, Domperidone, Fenoldopam, Haloperidol, Metoclopramide, Metopimazine, Pergolide Mesylate, Prochlorperazine, Quetiapine |
Plasticizer (0-20%): Plasticizer helps to improve the flexibility of films and reduces the brittleness of the films. However, inappropriate use of plasticizer may lead to film cracking, splitting, and peeling. Common plasticizers that can be used are glycerol, propylene glycol, etc. 10
Sweetening Agents (3-6%): Sweeteners have become an important part of the formulation that disintegrates or dissolves in the oral cavity. Both natural, as well as artificial sweeteners, can be used in the formulation of these fast dissolving films. Polyhydric alcohols such as sorbitol, mannitol, and isomalt can be used in combination as they provide good mouth feel and cooling sensation. However, the use of natural sugars should be restricted in people who are on a diet or in diabetic patients. Due to this reason, artificial sweeteners have gained more popularity in food and pharmaceutical preparations. Saccharin, cyclamate, and aspartame are artificial sweeteners.
Saliva Stimulating Agent (2-6%): The purpose of using saliva stimulating agents is to increase the rate of production of saliva that would aid in the faster disintegration of the rapid dissolving films. Generally, acids which are used in the preparation of food can be used as salivary stimulants such as citric acid, malic acid, lactic acid, ascorbic acid, and tartaric acid 11.
Flavouring Agents (0-10%): The acceptance of an orally disintegrating or dissolving formulation by an individual depends on the flavor quality which is observed in first few seconds after the product has been consumed and the after taste of the formulation which lasts for at least about 10min. The selection of flavor depends on the type of drug to be incorporated in the formulation. It was observed that age plays a significant role in the taste fondness. The geriatric population like mint or orange flavors, while the younger generation like flavors like fruit punch, raspberry etc. Flavoring agents can be selected from synthetic flavor oils, oleo resins, extract derived from various parts of the plants like leaves, fruits and flowers. Flavors can be used alone or in combination. Peppermint oil, cinnamon oil, spearmint oil, oil of nutmeg are examples of flavor oils, while vanilla, cocoa, coffee, chocolate, and citrus are fruity flavors. Apple, raspberry, cherry, pineapple are a few examples of fruit essence type 12.
Colouring Agents (1%): FD & C approved coloring agents are used for fast dissolving films like titanium dioxide.
Surfactants: Surfactants act as solubilizing or wetting or dispersing agent in the formulation so that the film gets dissolved within seconds and releases active agent quickly. Some of the commonly used surfactants are sodium lauryl sulfate, benzalkonium chloride, tweens, etc. One of the most important surfactants is Polaxamer 407 that is used as solubilizing, wetting, and dispersing agent 13.
Methods of Preparation: One or more combinations of the following process can be used to manufacture the fast dissolving films-
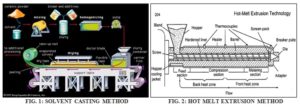
Solvent Casting Method: In solvent casting method water-soluble polymers are dissolved in water, and the drug along with other excipients is dissolved in suitable solvent then both the solutions are mixed and stirred and finally cast into the Petri plates, dried and cut into uniform dimensions 14.
Semisolid Casting Method: In the semisolid casting method firstly a solution of water-soluble film-forming the polymer is prepared. The resulting solution is added to a solution of acid insoluble polymer (e.g., cellulose acetate phthalate, cellulose acetate butyrate), which was prepared in ammonium or sodium hydroxide. Then an appropriate amount of plasticizer is added so that a gel mass is obtained. Finally, the gel mass is cast into the films or ribbons using heat controlled drums. The thickness of the film is about 0.015-0.05 inches. The ratio of the acid insoluble polymer to film-forming polymer should be 1:4 15.
Hot Melt Extrusion Method: In hot melt extrusion method firstly the drug is mixed with carriers in solid form. Then the extruder having heaters melts the mixture. Finally, the melt is shaped into films by the dies 16.
Solid Dispersion Extrusion Method: In this method, immiscible components are extruded with the drug, and then solid dispersions are prepared. Finally, the solid dispersions are shaped into films using dies.
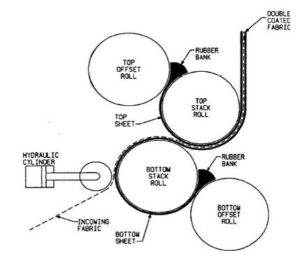
Rolling method: In the rolling method a solution or suspension containing drug is rolled on a carrier. The solvent is mainly water and a mixture of water and alcohol. The film is dried on the rollers and cut into desired shapes and sizes 17.
FIG. 3: ROLLING METHOD
Evaluation Parameters:
Organoleptic Evaluation: Colour is a vital means of identification for many pharmaceutical products and is also important for consumer acceptance. The color of the product must be uniform within a dosage form. Odour is also important for consumer acceptance of oral dosage forms and can provide an indication of the quality of oral films as the presence of an odor in a batch could indicate a stability problem. Taste is also an essential factor for consumer acceptance. Taste preference is subjective, and the control of taste in the production of oral soluble films is based on the presence or absence of a specified taste 18.
Drug Content Uniformity Test: The test for the content uniformity is carried out taking a sample film of size 2×2 sq cm, which is placed in a beaker containing 10 ml of a suitable medium. The contents are stirred in a cyclo-mixer to dissolve the film, which is then transferred to a volumetric flask (10ml). The absorbance of the solution is measured against the corresponding blank solution at a particular wavelength using a standard assay method described for the particular API mentioned in any of the standard pharmacopoeias. Content uniformity is determined by estimating the API content in the individual film. Limit of content uniformity is 85-115% 19.
Folding Endurance: Folding endurance is determined by repeated folding of the strip at the same place till the strip breaks. The number of times the film is folded without breaking is the folding endurance value. This indicates the brittleness of the film.
Surface pH: The film is placed on a Petri dish and moistened with 0.5 ml of distilled water. Kept for 30 sec. The pH is noted after bringing the electrode of the pH meter in contact with the surface of the film. The final result is determined by taking the mean of 3 readings.
Thickness Test: The thickness of the film is measured using a Micrometer Screw Gauge at different locations. The final result is determined by taking the mean of 6 readings. The thickness of the film ranges from 1-10 mm 20.
Disintegration Test: The disintegration of orally fast dissolving films requires USP disintegration apparatus. The disintegration time limit of 30 seconds or less for orally disintegrating tablets described in CDER guidance can be applied to fast dissolving oral strips. Disintegration time vary depending on the formulation but typically the disintegration range from 5 to 30 sec. Although, no official guidance is available for oral fast disintegrating films strips.
Dissolution Test: Dissolution studies of films are performed by using USP type II apparatus in 6.8 phosphate buffer (900 ml) and 0.1N HCl (900 ml). The temperature (37±0.5°C) and the rotation speed is 50 rpm. 5 ml samples are withdrawn at various time intervals and analyzed spectrophotometrically 21.
Elongation Test: Kinston Universal Testing Instrument is used. Films are pulled by 2 clamps at a rate of 100 mm/min. Force and elongation is measured when the film breaks.
% Elongation = Increase in length / Original length × 100
Tensile Strength Test: It is the maximum stress applied to a point at which the strip specimen breaks. It is performed to check the strength and elasticity of the film by using the tensile tester. It is calculated by the applied load at rupture divided by the cross‐sectional area of the strip as given in the equation below: 22
Tensile strength = (Load at Failure / Strip thickness × Strip width) × 100
Applications: Oral mucosal delivery via buccal, sublingual, and mucosal route by use of FDFs could become a preferential delivery method for therapies in which rapid absorption is desired, including those used to manage pain, allergies, sleep difficulties, and central nervous system disorders. Dissolvable FDFs evolved over the past few years from the confection and oral care markets in the form of breath strips and became a novel and widely accepted form by consumers for delivering vitamins and personal care products.
Topical Applications: The use of dissolvable films may be feasible in the delivery of active agents such as analgesics or antimicrobial ingredients for wound care and other applications.
Gastro-retentive Dosage System: Dissolvable films are being considered in dosage forms for which water-soluble, and poorly soluble molecules of various molecular weights are contained in a film format. Dissolution of the films could be triggered by the pH or enzyme secretions of the gastrointestinal tract, and could potentially be used to treat gastrointestinal disorders.
Diagnostic Devices: Dissolvable films may be loaded with sensitive reagents to allow controlled release when exposed to a biological fluid or to create isolation barriers for separating multiple reagents to enable a timed reaction within a diagnostic device 23.
TABLE 2: MARKETED FORMULATIONS OF FAST DISSOLVING FILMS 24, 25
| Brand name | API | Manufacturer |
| Orazel | Menthol/ Pectin | Del |
| Gas-X | Simethicone | Novartis |
| Benadryl | Diphenylhydramine HCl | Pfizer |
| Chloraseptic | Benzocaine | Prestige |
| Ondansterone | Ondansetrone | Labtec |
| Maxalt MLT | Rizatriptan | Merck |
| Zoming-ZMT | Zolmitriptan | AstraZeneca |
| Febrectol | Paracetamol | Prographarm |
| Nimulid | Nimesulide | Panacea Biotech |
| Torrox MT | Rofecoxib | Torrent Pharmaceuticals |
| Romilast | Montelukast | Ranbaxy |
| NuLev | Hyoscyamine sulfate | Cima Labs |
| FazaClo | Clozapine | AzurPharma |
| Mirtazapine | Mirtazapine | Teva Pharmaceuticals |
| Parcopa | Carbidopa/Levodopa | Schwarz Pharma |
| Theraflu | Dextromethorphan HBr | Novartis |
| Triaminic | Phenylephrine HCl | Novartis |
| Donepezil | Donepezil HCl | Labtec |
| Sudafed | Phenylephrine HCl | Pfizer |
| Rapid film | Ondansetron | GmbH |
CONCLUSION: Fast dissolving oral films have several advantages over conventional dosage forms. So, they are of great importance during emergency cases such as allergic reactions and asthmatic attacks whenever immediate onset of action is desired.
ACKNOWLEDGEMENT: I would like to thank organizers of the national conference entitled ‘Novel Tools and Treatment Approaches in Health Care System’ for selecting my paper for poster presentation, organized at Faculty of Pharmacy, Integral University, Lucknow on 3rd March 2015.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Corniello C: Quick dissolving strips: from concept to commercialization. Drug Delivery Technology 2006; 6: 68-71.
- Vishwakarma DK, Tripathi AK, Yogesh P and Maddheshiya B: Review article on mouth dissolving film. Journal of Global Pharma Technology 2011; 3(1): 1-8.
- Bhyan B, Jangra S, Kaur M and Singh H: Orally fast dissolving films: innovations in formulation and technology. International Journal of Pharmaceutical Sciences Review and Research 2011; 50-57.
- Vollmer U and Galfetti P: Rapid film: Oral thin films as an innovative drug delivery System and dosage form. Drug Dev Report 2006; 64-67.
- Arya A, Chandra A, Sharma V and Pathak K: Fast dissolving oral films: An innovative drug delivery system and dosage form. International Journal of Chem Tech Research 2010; 2(1): 576-583.
- Gavaskar B, Vijayakumar S, Sharma G and Rao YM: Overview of fast dissolving films. International Journal of Pharmacy and Pharmaceutical Science 2010; 2(3): 29-33.
- Bhura N and Sanghvi: A review on fast dissolving film. International Journal of Pharmaceutical Research and Bio-Science 2012; 1(3): 66-89.
- Bhowmik D, Chiranjib B, Krishnakanth and Chandira RM: Fast dissolving tablet: an overview. Journal of Chemical and Pharmaceutical Research 2009; 1(1): 163-177.
- Bala R, Pawar P, Khanna S and Arora S: Orally dissolving strips: A new approach to oral drug delivery system. Int J of Pharmaceutical Investigations 2013; 3: 67-76.
- Mashru RC, Sutariya BC and Parikh PP: Development and evaluation of fast dissolving films of salbutamol sulfate. Drug Dev Ind Pharm 2005; 31: 25-34.
- Desai P and Basu B: Design and evaluation of fast dissolving films of Domperidone. International Research Journal of Pharmacy 2012; 3(9): 134-142.
- Puthil SP and Dixit RP: Oral strip technology: overview and future potential. Journal of Controlled Release 2009; 139: 94-107.
- Patel AR, Prajapati DS and Raval JA: Fast dissolving films (FDFs) as a newer venture in fast dissolving dosage forms. International Journal of Drug Development and Research 2010; 2(2): 232-246.
- Ghodake P and Karande M: Mouth dissolving films: innovative vehicle for oral delivery. International Journal of Pharma Research and Review 2013; 2(10): 41-47.
- Kaur M, Rana AC and Seth N: Fast dissolving films: An innovative drug delivery system. Int J of Pharmaceutical Research & Allied Sciences 2013; 2(1): 14-24.
- Goel H, Rai P, Rana V and Tiwari KA: Orally disintegration systems: innovations in formulation and technology. Recent Patents on Drug Delivery and Formulation 2008; 2(3): 258-274.
- Mahtre S and Khanekar P: Rapid dissolving oral dosage form. J of Global Pharma Technology 2012; 10(4); 1-14.
- Malke M, Shidhaye S and Kadam VJ: Formulation and evaluation of oxcarbazepine fast dissolve tablets. Indian J Pharmaceutical Sci 2007; 69(2): 211-214.
- Gohel MC, Sharma R, Soniwala MM: Development of taste masked film of Valdecoxib for oral use. Ind J Pharm Sci 2007; 69: 318-320.
- Singh S, Gangwar S, Garg G, Garg V and Sharma PK: Formulation and evaluation of rapidly disintegrating film of levocetirizine hydrochloride. Scholars Research Library Der Pharmacia Lettre 2010; 2(2): 434-439.
- Nagar P, Chauhan I and Yasir M: Insights into Polymers: film formers in mouth dissolving films. Drug Invention Today 2011; 3(12): 280-289.
- Suresh B, Halloran D and James L: Quick dissolving films: A novel approach to drug delivery. Drug Dev Tech 2006; 1-7.
- Rathi V, Senthil V, Kammili L and Hans R: A brief review on oral film technology. International Journal of Research in Ayurveda and Pharmacy 2010; 2(4): 1138-1147.
- Saini S, Nanda A and Hooda M: Fast dissolving films (FDF). Innovative Drug Delivery System 2011; 919-928.
- Sau-hung S, Robert S and Lori D: Fast dissolving orally consumable films. U.S. Patent, 2003; 6: 596, 298.
How to cite this article:
Banarjee T, Ansari VA, Singh S, Mahmood T and Akhtar J: A review on fast dissolving films for buccal delivery of low dose drugs. Int J Life Sci & Rev 2015; 1(4): 117-23. doi: 10.13040/ IJPSR.0975-8232.IJLSR.1(4).117-23.
All © 2015 are reserved by International Journal of Life Sciences and Review. This Journal licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.
Article Information
3
117-123
704
2559
English
IJLSR
T. Banarjee, V. A. Ansari, S. Singh, T. Mahmood and J.Akhtar
Faculty of Pharmacy, Integral University, Kursi Road, Lucknow, Uttar Pradesh, India.
er.pandeykullu@gmail.com
19 February 2015
15 March 2015
27 March 2015
10.13040/IJPSR.0975-8232.IJLSR.1(4).117-23
01 April 2015