A REVIEW ON MICROSPHERES A NOVEL DRUG DELIVERY SYSTEM FOR MULTI-PARTICULATE DRUG RELEASE
HTML Full TextA REVIEW ON MICROSPHERES A NOVEL DRUG DELIVERY SYSTEM FOR MULTI-PARTICULATE DRUG RELEASE
Neha Agnihotri *, G. C. Soni, Dilip Kumar Chanchal, Afrin Khan and Sakshi Tiwari
Institute of Pharmacy, Bundelkhand University Jhansi - 284128, Uttar Pradesh, India.
ABSTRACT: A new era of science and technology has evolved in pharmaceutical research focused on the development of different novel drug delivery systems. The evolution of an existing drug from its traditional form to a novel delivery system may considerably improve its performance in aspects of efficacy, safety, and patient compliance. The goal of any drug delivery system is to provide a therapeutic amount of drug to the proper site in the body and then maintain the desired drug concentration. The microsphere is typically injected, but oral dosage forms are being developed to improve the treatment of gastrointestinal conditions. In future microspheres will find the central place in novel drug delivery, particularly in diseased cell sorting, diagnostics, genetic materials, targeted and effective drug delivery. Microspheres are multiparticulate drug delivery systems which are prepared to obtain prolonged or controlled drug delivery to improve bioavailability, stability and to target the drug to a specific site at a predetermined rate. These delivery systems offer numerous advantages compared to conventional dosage forms, which include improved efficacy, reduced toxicity, improved patient compliance, and convenience. Such systems often use macromolecules as carriers for the drugs. The current aim of this review is to study various aspects of the microparticulate drug delivery system including a method of formulation, evaluation & characterization. The present review highlights various types of microspheres, different methods of preparation, its applications and also various parameters to evaluate their efficiency. Microspheres are various types like bioadhesive microspheres, magnetic microspheres, floating microspheres, radioactive microspheres, polymeric microspheres, biodegradable polymeric microspheres, synthetic polymeric microspheres and are prepared by methods like spray drying, solvent evaporation, single emulsion technique, double emulsion technique, emulsion cross-linking method, multiple emulsion method, phase separation coacervation technique, spray drying and solvent extraction. A most important application is that it is used for targeting tumors using anticancer drugs. Being more stable, it has an advantage over other delivery systems like liposomes. It is an important carrier for safe and effective in-vivo drug delivery.
Keywords: Microsphere, Polymers, Release of drug and delivery of drug
INTRODUCTION: The concept of packing microscopic quantities of material within microspheres dates to 1930’s the work of Bungenberg de Jong and co-workers on the entrapment of substance with coacervates 1.
To obtain maximum therapeutic efficacy, it becomes necessary to deliver the agent to the target tissue in the optimal amount in the right period thereby causing little toxicity and minimal side effects 2.
Oral route drug administration is by far the preferable route for taking medications. However, their short circulating half-life and restricted absorption via a defined segment of intestine limit the therapeutic potential of many drugs. Such a pharmacokinetic limitation leads in many cases to frequent dosing of medication to achieve the therapeutic effect. A rational approach to enhance bioavailability and improve pharmacokinetic and pharmacodynamic profile is to release the drug in a controlled manner and site-specific manner 3. There are various approaches in delivering a therapeutic substance to the target site in a sustained, controlled release fashion. One such approach is using microspheres as carriers for drugs. The development of new delivery systems for the controlled release of drugs is one of the most interesting fields of research in pharmaceutical sciences. A well designed controlled drug delivery system can overcome some of the problems of conventional therapy and enhance the therapeutic efficacy of a given drug. To obtain maximum therapeutic efficacy, it becomes necessary to deliver the agent to the target tissue in the optimal amount in the right period thereby causing little toxicity and minimal side effects. There are various approaches in delivering a therapeutic substance to the target site in a sustained controlled release fashion 4.
Microspheres are defined as “Monolithic sphere or therapeutic agent distributed throughout the matrix either as a molecular dispersion of particles” (or) can be defined as a structure made up of the continuous phase of one or more miscible polymers in which drug particles are dispersed at the molecular or macroscopic level. Microspheres are small spherical particles, with diameters in the micrometer range (typically 1 μm to 1000 μm). Microspheres are sometimes referred to as microparticle
Advantages:
- They facilitate accurate delivery of small quantities of potent drug and reduced concentration of drug at a site other than the target organ or tissue.
- They protect the unstable drug before and after administration, before their availability at the site of action.
- They provide the ability to manipulate the in-vivo action of the drug, pharmacokinetic profile, tissue distribution and cellular interaction of the drug. They enable the controlled release of the drug - examples: narcotic, antagonist, steroid hormones 5.
- Particle size reduction for enhancing the solubility of the poorly soluble drug.
- Provide constant and prolonged therapeutic effect.
- Provide constant drug concentration in the blood thereby increasing patent compliance.
- Decrease dose and toxicity.
- Protect the drug from enzymatic and photolytic cleavage hence found to be best for drug delivery.
- Reduce the dosing frequency and thereby improve patient
- Better drug utilization will improve the bioavailability and reduce the incidence or intensity of adverse effects.
- Microsphere morphology allows a controllable variability in degradation and drug release.
- Convert liquid to solid form & to mask the bitter taste.
- Protects the GIT from irritant effects of the drug.
- Biodegradable microspheres have the advantage over large polymer implants in that they do not require surgical procedures for implantation and removal.
- Controlled release delivery biodegradable microspheres are used to control drug release rates thereby decreasing toxic side effects, and eliminating the inconvenience of repeated injection 6.
Polymers Used in Preparation of Microsphere Development: Microspheres used usually are polymers. They are classified into two types: Synthetic Polymers and Natural Polymers.
1. Synthetic Polymers:
Non-biodegradable Polymers: Polymethyl methacrylate (PMMA), acrolein, glycidyl methacrylate, epoxy polymers.
Biodegradable Polymers: Lactides, their glycolides, and their copolymers, polyalkyl cyanoacrylate, polyanhydrides.
2. Natural Polymers: These are obtained from different sources like proteins, carbohydrates, and chemically modified carbohydrates.
Proteins: Albumin, gelatin, and collagen, carbohydrates: agarose, carrageenan, chitosan, starch, chemically modified carbohydrates: poly (acryl) dextran, poly (acryl) starch 7, 8.
Types of Microspheres: 12, 13, 14
1. Bioadhesive Microspheres: These kinds of microspheres exhibit a prolonged residence time at the site of application and causes intimate contact with the absorption site and produces better therapeutic action 9, 10, 11. Adhesion can be defined as sticking of the drug to the membrane by using the sticking property of the water-soluble polymers. Adhesion of drug delivery device to the mucosal membrane such as buccal, ocular, rectal, nasal, etc. can be termed as bio-adhesion. These kinds of microspheres called bioadhesive microsphere.
2. Magnetic Microspheres: This kind of delivery system is very much important which localizes the drug to the disease site. In this larger amount of freely circulating drug can be replaced by a smaller amount of magnetically targeted drug. Magnetic carriers receive magnetic responses to a magnetic field from incorporated materials that are used for magnetic microspheres are chitosan, dextran, etc. The different types are therapeutic magnetic microspheres and diagnostic microspheres 18. Magnetic microspheres are supramolecular particles that are small enough to circulate through capillaries without producing embolic occlusion (<4μm) but are sufficiently susceptible (ferromagnetic) to be captured in micro vessels and dragged into the adjacent tissues by the magnetic field of 0.5-0.8 tesla 9, 19.
Therapeutic Magnetic Microspheres: It is used to deliver a chemotherapeutic agent to the liver tumor. Drugs like proteins and peptides can also be targeted through this system.
Diagnostic Microspheres: It can be used for imaging liver metastases and also can be used to distinguish bowel loops from other abdominal structures by forming nano-size particles supramagnetic iron oxides 9, 19.
3. Floating microspheres: In floating types, the bulk density is less than the gastric fluid and so remains buoyant in the stomach without affecting the gastric emptying rate. The drug is released slowly at the desired rate if the system is floating on gastric content and increases gastric residence and increases fluctuation in plasma concentration. Moreover, it also reduces the chances of striking and dose dumping. One another way it produces a prolonged therapeutic effect and therefore reduces dosing frequencies 20, 21. Gastroretentive floating microspheres are low-density systems that have sufficient buoyancy to float over gastric contents and remain in the stomach for a prolonged period without affecting gastric emptying rate 9, 22, 23.
4. Polymeric Microspheres: The different types of polymeric microspheres can be classified as follows and they are biodegradable polymeric microspheres & synthetic polymeric microspheres.
Biodegradable Polymeric Microspheres: Natural polymers such as starch are used with the concept that they are biodegradable, biocompatible, and also bioadhesive in nature. Biodegradable polymers prolong the residence time when contact with mucous membrane due to its high degree of swelling property with an aqueous medium, results in gel formation. The rate and extent of drug release are controlled by the concentration of polymer and the release pattern in a sustained manner. The main drawback is in clinical use drug loading efficiency of biodegradable microspheres is complex and is difficult to control the drug release.
Synthetic Polymeric Microspheres: The interest of synthetic polymeric microspheres are widely used in the clinical application. Moreover, that also used as a bulking agent, fillers, embolic particles drug delivery vehicles, etc. and proved to be safe and biocompatible. But the main disadvantage of these kinds of microspheres tends to migrate away from the injection site and lead to potential risk, embolism and further organ damage 24.
5. Radioactive Microspheres: Radioactive microspheres deliver a high radiation dose to the targeted areas without damaging the normal surrounding tissues. They are injected to the arteries that lead to a tumor of interest. The different kinds of radioactive microspheres are α emitters, β emitters and γ emitters 9, 25.
Limitation: Some of the disadvantages were found to be as follows:
- The modified release from the formulations.
- The release rate of the controlled release dosage form may vary from a variety of factors like food and the rate of transit through the gut.
- Differences in the release rate from one dose to another.
- Controlled release formulations generally contain a higher drug load, and thus any loss of integrity of the release characteristics of the dosage form may lead to potential toxicity.
- Dosage forms of this kind should not be crushed or chewed 26.
Criteria for Microsphere Preparation: Incorporation of solid, liquid or gases into one or more polymeric coatings can be done by microencapsulation technique 27. The different methods used for various microspheres preparation depends on particle size, route of administration, duration of drug release and these above characters related to rpm, a method of cross-linking, the drug of cross-linking, evaporation time, co-precipitation, etc. 28 Preparation of microspheres should satisfy certain criteria 29:
- The ability to incorporate reasonably high concentrations of the drug.
- Stability of the preparation after synthesis with a clinically acceptable shelf life.
- Controlled particle size and dispersibility in aqueous vehicles for injection.
- Release of the active reagent with good control over a wide time scale.
- Biocompatibility with a controllable bio-degradability and
- Susceptibility to chemical modification.
Method of Preparation: The various methods of preparations are:
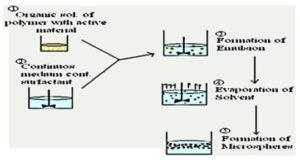
1. Emulsion Solvent Evaporation Technique: In this technique, the drug is dissolved in the polymer which was previously dissolved in chloroform, and the resulting solution is added to an aqueous phase containing 0.2% sodium of PVP as an emulsifying agent. The above mixture was agitated at 500 rpm then the drug and polymer (eudragit) were transformed into a fine droplet which solidified into rigid microspheres by solvent evaporation and then collected by filtration and washed with demineralized water and desiccated at room temperature for 24 h 30.
2. Emulsion Solvent Diffusion Technique: The colon floating microspheres were prepared using the emulsion solvent diffusion technique to improve the residence time. The drug-polymer mixture was dissolved in a mixture of ethanol and dichloromethane (1:1) and then the mixture was added dropwise to sodium lauryl sulphate (SLS) solution. The solution was stirred with propeller type agitator at room temperature at 150 rpm for 1 hr. Thus the formed floating microspheres were washed and dried in a desiccator at room temperature 7, 8, 30.
FIG. 1: EMULSION SOLVENT EVAPORATION TECHNIQUE 34
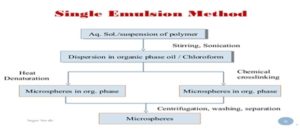
3. Single Emulsion Technique: The micro-particulate carriers of natural polymers, i.e. those of proteins and carbohydrates are prepared by single emulsion technique. The natural polymers are dissolved/dispersed in an aqueous medium followed by dispersion in the non-aqueous medium, e.g. oil. In the second step of preparation, cross-linking of dispersed globule is carried out. The cross-linking is achieved by two methods, i.e. either by heat or using chemical cross linking agents including glutaraldehyde, formaldehyde, di-acid chloride, etc. 7, 8, 9
FIG. 2: MICROSPHERES BY SINGLE EMULSION TECHNIQUE
4. Double Emulsion Technique: This method involves the formation of the multiple emulsion or double emulsion of type w/o/w. It is best suited to water-soluble drugs, peptides, proteins, and vaccines. This method can be used with both the natural as well as the synthetic polymers.
The aqueous protein solution is dispersed in a lipophilic organic continuous phase.This protein solution may contain active constituents. The continuous phase generally consists of the polymer solution that eventually encapsulates of the protein homogenization or the sonication before addition to the aqueous solution of the polyvinyl alcohol (PVA). This results in the formation of a double emulsion. The emulsion is then subjected to solvent removal either by solvent evaporation or by the solvent extraction process. The solvent evaporation is carried out by maintaining emulsion at reduced pressure or by stirring the emulsion so that the organic phase evaporates out. The emulsion is then added to a large quantity of water into which organic phase diffuses out. The solid microspheres are subsequently obtained by filtration and washing with n-hexane, acetone or an organic solvent to remove traces of oil from the surface.
FIG. 3: MICROSPHERES BY DOUBLE EMULSION TECHNIQUE
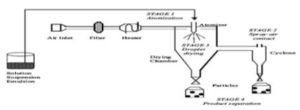
5. Spray Drying Method: The polymer is first dissolved in a suitable volatile organic solvent such as dichloromethane, acetone, etc.
FIG. 4: MICROSPHERES BY SPRAY DRYING TECHNIQUE
The drug in the solid form is then dispersed in the polymer solution under high-speed homogenization.
This dispersion is then atomized in a stream of hot air. The atomization leads to the formation of small droplets or the fine mist from which the solvent evaporates instantaneously leading the formation of microspheres 7, 8, 33.
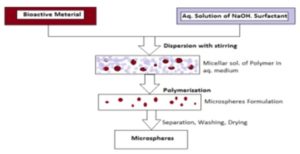
6. Polymerization Method: The polymerization techniques conventionally used for the preparation of the microspheres, are mainly classified as Normal Polymerization and Interfacial Polymerization 7, 8, 9, 37.
Normal Polymerization: Normal polymerization proceeds and is carried out using different techniques as bulk, suspension precipitation, emulsion, and micellar polymerization processes. In bulk polymerization, a monomer or a mixture of monomers along with the initiator or catalyst is usually heated to initiate polymerization. The polymer so obtained may be molded as microspheres. Drug loading may be done during the process of polymerization. Suspension polymerization also referred to as a bead or pearl polymerization. Here, it is carried out by heating the monomer or mixture of monomers as droplets dispersion in a continuous aqueous phase. The droplets may also contain an initiator and other additives. Emulsion polymerization differs from suspension polymerization as due to the presence of an initiator in the aqueous phase, which later on diffuses to the surface of micelles. Bulk polymerization has an advantage of formation of pure polymers 38.
FIG. 5: POLYMERIZATION METHOD
Interfacial Polymerization: It involves the reaction of various monomers at the interface between the two immiscible liquid phases to form a film of polymer that essentially envelops the dispersed phase. In this technique, two reacting monomers are employed, one of which is dissolved in the continuous phase while the other being dispersed in the continuous phase 8.
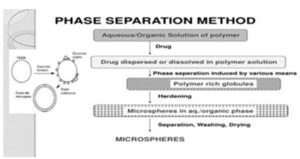
7. Phase Separation Method: Phase separation method is mainly designed for preparing the reservoir type of the system. This method is used to encapsulate water soluble drugs, e.g. peptides, proteins and some of the preparations having matrix type particular when the drug is hydrophobic, e.g. steroids. In this technique, the polymer is first dissolved in a suitable solvent, and then the drug is dispersed by making it's aqueous 7, 8, 39.
FIG. 6: PHASE SEPARATION METHOD
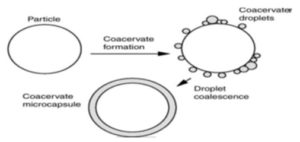
8. Coacervation Method:
Coacervation Thermal Change: Performed by the weighed amount of ethyl cellulose was dissolved in cyclohexane with vigorous stirring at 80 ºC by heating. Then the drug was finely pulverized and added with vigorous stirring on the above solution, and phase separation was done by reducing temperature and using an ice bath. Then above product was washed twice with cyclohexane and air dried then passed through a sieve (sieve no. 40) to obtain individual micro-capsule 25.
FIG. 7: COACERVATION METHOD
Coacervation Non-Solvent Addition: Developed by weighed amount of ethyl cellulose was dissolved in toluene containing polyisobutylene in the closed beaker with magnetic stirring for 6 h at 500 rpm, and the drug is dispersed in it and stirring is continued for 15 min. Then phase separation is done by petroleum benzoin 5 times with continuous stirring 1. After that, the microcapsules were washed with n-hexane and air dried for 2 h and then in an oven at 50 ºC for 4h 40.
9. Solvent Extraction: Solvent extraction method is used for the preparation of the microparticles, involves removal of the organic phase by extraction of the organic solvent. The method involves water-miscible organic solvents such as isopropanol. The organic phase is removed by extraction with water. This process decreases the hardening time for the microspheres. The process involves the direct addition of the drug or protein to polymer organic solution. The rate of solvent removal by extraction method depends on the temperature of water, the ratio of emulsion volume to the water and the solubility profile of the polymer 8.
FIG. 8: SOLVENT EXTRACTION
10. Emulsion Cross-Linking Method: In this method, the drug was dissolved in an aqueous gelatine solution which was previously heated for 1 h at 40 ºC. The solution was added dropwise to liquid paraffin while stirring the mixture at 1500 rpm for 10 min at 35 ºC, results in w/o emulsion then further stirring is done for 10 min at 15 ºC. Thus the produced microspheres/were washed respectively three times with acetone and isopropyl alcohol which then air dried and dispersed in 5 ml of aqueous glutaraldehyde saturated toluene solution at room temperature for 3 h for cross-linking and then was treated with 100 ml of 10 mm glycine solution containing 0.1% w/v of tween 80 at 37 ºC for 10 min to block unreacted glutaraldehyde 18. Examples for this technique is gelatin A microspheres.
11. Multiple Emulsion Method: Oral controlled release drug delivery of various drugs was prepared by this technique. In the beginning, powder drug was dispersed in solution (methylcellulose) followed by emulsification in ethyl cellulose solution in ethyl acetate. The primary emulsion was then re-emulsified in an aqueous medium. Under optimized condition, discrete microspheres were formed during this phase 41.
Physicochemical Evaluation:
Characterization: The characterization of the microparticulate carrier is an important phenomenon, which helps to design a suitable carrier for the proteins, drug or antigen delivery. These microspheres have different microstructures. These microstructures determine the release and the stability of the carrier 48.
Particle Size and Shape: The most widely used procedures to visualize microparticles are conventional light microscopy (LM) and scanning electron microscopy (SEM). Both can be used to determine the shape and outer structure of microparticles. LM provides control over coating parameters in case of double-walled microspheres. The microspheres structures can be visualized before and after coating, and the change can be measured microscopically. SEM provides higher resolution in contrast to the LM. SEM allows investigations of the microspheres surfaces and after particles are cross-sectioned 52.
Electron Spectroscopy for Chemical Analysis: The surface chemistry, atomic composition of surface and surface degradation of biodegradable microspheres can be determined using the electron spectroscopy for chemical analysis (ESCA).
Attenuated Total Reflectance Fourier Transform Infrared Spectroscopy: FTIR is used to determine the degradation of the polymeric matrix of the carrier system. The surface of the microspheres is investigated measuring alternated total reflectance (ATR). The ATR-FTIR provides information about the surface composition of the microspheres depending upon manufacturing procedures and conditions.
Density Determination: The density of the microspheres can be measured by using a multi-volume pycnometer. The accurately weighed sample in a cup is placed into the multi-volume pycnometer. Helium is introduced at a constant pressure in the chamber and allowed to expand. This expansion results in a decrease in pressure within the chamber. Two consecutive readings of reduction in pressure at different initial pressure are noted. From two pressure readings, the volume and hence the density of the microsphere carrier is determined.
Isoelectric Point: The microelectrophoresis is an apparatus used to measure the electrophoretic mobility of microspheres from which the isoelectric point can be determined. The electrophoretic mobility can be related to surface contained charge, ionizable behavior or ion absorption nature of the microspheres.
Surface Carboxylic Acid Residue: The surface carboxylic acid residue is measured by using radioactive glycine. The radioactive glycine conjugates are prepared by the reaction of C14-glycine ethyl ester hydrochloride with the microspheres. The radioactivity of the conjugate is then measured using a liquid scintillation counter. Thus, the carboxylic acid residue can be compared and correlated.
Surface Amino Acid Residue: Surface-associated amino acid residue is determined by the radioactive C14-acetic acid conjugate. The carboxylic acid residue is measured through the liquid scintillation counter, and hence the amino acid residue can be determined indirectly.
Capture Efficiency: The capture efficiency of the microspheres or the percent entrapment can be determined by allowing washed microspheres to lose. The lysate is then subjected to the determination of active constituents as per monograph requirement. The percent encapsulation efficiency is calculated using the following equation:
% Entrapment = Actual content / Theoretical content × 100
Angle of Contact: The angle of contact is measured to determine the wetting property of a microparticulate carrier. It determines the nature of microspheres in terms of hydrophilicity or hydrophobicity. The angle of contact is measured at the solid/air/water interface 53.
Applications:
Microspheres in Vaccine Delivery: An ideal vaccine must fulfill the requirement of efficacy, safety, convenience in application and cost.
Biodegradable delivery systems for vaccines that are given by parenteral route may overcome the shortcoming of the conventional vaccines. The interest in parenteral (subcutaneous, intramuscular, intradermal) carrier lies since they offer specific advantages including:
- Improved antigenicity by adjuvant action
- Modulation of antigen release
- Stabilization of antigen.
Targeting using Microparticulate Carriers: The concept of targeting, i.e., site-specific drug delivery is a well-established dogma, which is gaining full attention. The therapeutic efficacy of the drug relies on its access and specific interaction with its candidate receptors. The ability to leave the pool in a reproducible, efficient and specific manner is center to drug action mediated by the use of a carrier system.
Monoclonal Antibodies Mediated Microspheres Targeting: Monoclonal antibodies targeting microspheres are immune microspheres. This targeting is a method used to achieve selective targeting to specific sites. Monoclonal antibodies are extremely specific molecules. Mabs can be directly attached to the microspheres using a covalent coupling. The Mabs can be attached to microspheres by any of the following methods:
- Nonspecific adsorption and specific adsorption.
- Direct coupling.
- Coupling via
Chemoembolization: Chemoembolization is an endovascular therapy, which involves the selective arterial embolization of a tumor together with simultaneous or subsequent local delivery the chemotherapeutic agent.
Imaging: The particle size range of microspheres is an important factor in determining the imaging of particular sites using radiolabelled microspheres. The particles injected intravenously apart from the portal vein will become entrapped in the capillary bed of the lungs. This phenomenon is exploited for the scintigraphic imaging of the tumor masses in lungs using labeled human serum albumin microspheres.
Topical Porous Microspheres: Microsponges are porous microspheres having a myriad of inter-connected voids of particle size range 5-300 μm. These microsponges have the capacity to entrap a wide range of active ingredients such as emollients, fragrances, essential oils, etc., are used as the topical carrier system 54.
Medical Application:
- The release of proteins, hormones, and peptides over an extended period.
- Gene therapy with DNA plasmids and also the delivery of insulin.
- Vaccine delivery for treatment of diseases like hepatitis, influenza, pertussis, ricin toxoid, diphtheria, birth control.
- Passive targeting of leaky tumor vessels, active targeting of tumor cells, antigens, by the intraarterial/ intravenous application.
- Tumour targeting with doxorubicin and also
- Treatments of leishmaniasis.
- Magnetic microspheres can be used for stem cell extraction and bone marrow purging.
- It is used in isolation of antibodies, cell separation & toxin extraction by affinity-chromatography.
- It is used for various diagnostic tests for infectious diseases like bacterial, viral, and fungal 43.
Radioactive Microsphere’s Application: 4
- Can be used for radio embolization of liver and spleen tumors.
- It is used for radio synovectomy of arthritis joint, local radiotherapy, interactivity treatment.
- Imaging of liver, spleen, bone marrow, lung and even imaging of thrombus in deep vein thrombosis can be done.
Future Challenges: Future challenges of microspheres look bright particularly in the area of medicinal field because of its wide spectrum of application in molecular biology, e.g., microsphere-based genotyping platform is used to detect six single nucleotide polymorphism, yttrium-90 microspheres are used to prevent tumor after liver transplantation, and it’s advanced way in delivery of vaccines and proteins.
CONCLUSION: The present review article shows that microspheres are a better choice of drug delivery system than many other types of drug delivery system. In future by combining various other strategies, microspheres will find the central and significant place in novel drug delivery, particularly in diseased cell sorting, diagnostics, gene & genetic materials, safe, targeted, specific and effective in-vivo delivery and supplements as miniature versions of diseased organ and tissues in the body. Drug absorption in the gastrointestinal tract is a highly variable procedure and prolonging gastric retention of the dosage form extends the time for drug absorption.
Microspheres by ionotropic gelation technique promise to be a potential approach for gastric retention. Although, there are a number of difficulties to be worked out to achieve prolonged gastric retention, a large number of companies are focusing on commercializing this technique. In future by combining various other strategies, microspheres will find the central place in novel drug delivery, particularly in diseased cell sorting, diagnostics, gene & genetic materials, safe, targeted and effective in-vivo delivery and supplements as miniature versions of diseased organ and tissues in the body.
ACKNOWLEDGEMENT: The authors thankful to Mr. G. C. Soni for his valuable guidance during the work.
CONFLICT OF INTEREST: Nil
REFERENCES:
- https://www.slideshare.net/DivyaThakur17/microspheres-67311278
- Jain NK: Controlled and Novel drug delivery, CBS Publishers New Delhi, India Edition 4th, 21: 236-237.
- Jamini M and Rawat S: A review on the microsphere. Res J Pharm Boil Chem Sci 2013; 4(1): 1223-33.
- Prasad BSG, Gupta VRM, Devanna N, and Jayasurya K: Microspheres as drug delivery system- A review. Journal of Global Trends in Pharmaceutical Science 2014; 5(3): 1961-1972.
- Omkar T, Alagusundaram M, Madhu SC, Umashankari K, Attuluri VB, Lavanya C and Ramkanth B: Microspheres as a novel drug delivery system. Int J of Chem-Tech and Res 2009; 3(1): 526-534.
- Kadam NR and Suvarna V: Microspheres: A brief review. Asian Journal of Biomedical and Pharmaceutical Sciences 2015; 5(47): 13-19.
- Patel NR, Patel DA, Bharadia PD, Pandya V and Modi V: Microsphere as a novel drug delivers. Int J of Pharm & Life Sci 2011; 2(8): 992-997.
- Vyas and Khar: Targeted and Controlled drug delivery. CBS Publishers and distributors 2001.
- Prasanth VV, Moy AC, Mathew ST and Mathapan R: Microspheres: an overview. Int J of Pharm & Biomedical Sci 2011; 2(2): 332-338.
- Vasir JK and Tambekar K: Bioadhesive microspheres as a controlled drug delivery system. Int Jou Pharm 2003; 255: 13-32.
- Senthil A, Narayanswamy VB, Galge DS, Bhosale RS, Tadwee IAK and Shahi S: Mucoadhesive microspheres. IJRAP 2011; 1(2): 55-59.
- Thube M and Ankit S: Review on microspheres. International Journal of Pharmaceutical Research Allied Sciences 2012; 1(1): 24-33.
- Kumar SK, Reddy JP and Sekhar CKB: A review on microsphere for novel drug delivery system. Journal of Pharmacy Research 2012; 5(1): 420-424.
- Kataria S, Middha A, Sandhu P, Bilandi A and Kapoor B: Microsphere: A review. International Journal of Research In Pharmacy and Chemistry 2011; 1(4): 1184-1198.
- Sipai AB, Yadav MV, Mamatha Y and Prasanth VV: Mucoadhesive microsphere an overview. American Journal of Pharmtech Research 2012; 2(1): 237-258.
- Hardenia SS, Jian A, Patel JR and Kaushal A: Formulation and evaluation of mucoadhesive microsphere of ciprofloxacin. Journal of Advanced Pharmacy Education and Research 2011; 1(4): 214-224.
- Nalini AM, Jain ASK and Jain NK: Con-A conjugated mucoadhesive microspheres for the colonic delivery of diloxanide furoate. International Journal of Pharmaceutics 2008; 3(59): 182-189.
- Liu G, Yang H and Zhou J: Preparation of magnetic microsphere from water-in-oil emulsion stabilized by block copolymer dispersant. Biomacromolecules 2005; 6: 1280-1288.
- Chandrawanshi P and Patidar H: Magnetic microspheres: as targeted drug delivery. Journal of Pharm Res 2009; 2(5): 964-966.
- Dutta P, Struti J, Niranajan PC and Bhaojirao ME: Floating Microsphere: Recent trends in the development of gastro-retentive floating drug delivery system. International Journal of Pharmaceutical Science and Nanotechnology 2011; 4(1): 1293-1306.
- Kawashima Y, Niwa T, Takeuchi H, Hino T and Ito Y: Preparation of multiple unit hollow microspheres (microbial loons) with an acrylic resin containing tranilast and their drug release characteristics (in vitro) and floating behavior (in-vivo), J Control. Release 1991; 16: 279-290.
- Chawla C, Gupta P, Karadia V and Bansal AK: Gastro-retention - A means to address regional variability in intestinal drug absorption. Phar Tech 2003; 7(2): 2, 50-68.
- Gholap SP, Banrjee SK, Gaikwad DD, Jadhav SL and Thorat RM: Hollow microspheres: A review. Int J of Pharm Sci Review and Res 2010; 1(1): 74-79.
- Andrianov AK and Payne LG: Polymeric carriers for oral uptake of microparticulates. Advanced Drug Delivery Reviews 1998; 34: 155-170.
- Hafeli U, Atcher RW, Morris CE, Beresford B, Humm JL and Macklis RM: Polymeric radiopharmaceutical delivery systems. Radioactivity & Radiochemistry 1992; 3: 11-14.
- Vyas SK and Khar RK: Targeted and Controlled drug delivery, Vallabh Prakashan, New Delhi India, Edition 7th, 420-445.
- Ghulam M, Mahmood A, Naveed A and Fatima RA: Comparative study of various microencapsulation techniques Effect of polymer viscosity on microcapsule characteristics. Pak J Sci 2009; 22(3): 291-300.
- Li, SP, Kowalski CR, Feld KM and Grim WM: Recent Advances in microencapsulation technology & equipment. Drug Dev Ind Pharm 1988; 14: 353-376.
- Alagusundaram M, Chetty MSC, Umashankari K, Badarinath AV, Lavanya C and Ramkanth S: Micro-spheres as a novel drug delivery system - a review. International Journal of Chem Tech Research 2009; 1(3): 526-534.
- Trivedi P, Verma AML and Garud N: Preparation and Characterization of aceclofenac microspheres. Asian Journal of Pharmaceutics 2008; 2(2): 110-115.
- Das MK and Rao KR: Evaluation of zidovudine encapsulated ethyl cellulose microspheres prepared by water-oil-water double emulsion solvent diffusion technique. Acta Poloniae Pharmaceutica & Drug Research 2006; 63(2): 141-148.
- Jain D, Panda AK and Majumdar DK: Eudragit S100 entrapped insulin microspheres for oral delivery. AAPS Pharm Sci Tech 2005; 6(1): 101-107.
- Mahajan H, Gattani S and Surana S: Spray dried mucoadhesive microspheres of ondansetron for nasal administration. Int J of Pharma Sci and Nanotech 2008; 1(3): 267-273.
- https://www.google.co.in/url?sa=i&source=images&cd.
- http://www.authorstream.com/Presentation/v.ramani007-1706786-microsphere-vdr/
- Singh P: Indian Journal of Novel Drug Delivery 2011; 3(2): 70-82.
- Chinna GB, Shyam SR, Vimal KM, Sleeva RM and Sai KM: Formulation and evaluation of indomethacin microspheres using natural and synthetic polymers as controlled release dosage forms. Int J of Drug Discovery 2010; 2(1): 8-16.
- Zhou WQ, Gu TY, Su ZG and Ma GH: Synthesis of macroporous poly (styrene-divinyl benzene) microspheres by surfactant reverse micelles swelling method. Science Direct Polymer 2007; 48: 1981-1988
- Sunitha S, Amareshwar P, Santhosh KM and Chakravarti P: Preparation and evaluation of tramadol hydrochloride microspheres by phase separation coacervation technique using various solvents and nonsolvents. Journal of Global Pharm Tech 2011; 3(4):33-41.
- Ghulam M, Mahmood A, Naveed A and Fatima RA: Comparative study of various microencapsulation techniques. Effect of polymer viscosity on microcapsule characteristics. Pak J Sci 2009; 22(3): 291-300.
- Sam MT, Devi Gayathri S, Prasanth VV and Vinod B: NSAIDs as microspheres. The Internet Journal of Pharmacology 2008; 6(1): 67-73.
- Rashi S, Singh D, Chanchal DK, Singh M, Anand S and Alok S: Cubosomes: a bicontinuous cubic crystalline phase. International Journal of Life Sciences and Review 2015; 1(10): 291-301.
- Blanco-Prieto MJ, Besseghir K, Orsolini P and Heimgartner F: Importance of the test medium for the release kinetics of a somatostatin analogue from poly (D, L-lactide-co-glycolide) microspheres. International Journal of Pharmaceutics 1999; 184: 243-250.
- Venkatesh H and Badri Narayan N: A buccal delivery system of salbutamol sulphate. Utilization and Evaluation of Plant Products in Pharmaceutical Formulations 1991.
- Save T and Venkatachalam P: Bioadhesive tablets of Nifedipine: Standardization of a novel buccoadhesive erodible carrier. Drug Delivery Ind Pharm 1994; 20(19): 3005-14.
- Lopez CR, Portero A, Vila-Jato JC and Alonso MJ: Design and Evaluation of chitosan/ ethylcellulose mucoadhesive bilayered devices for buccal drug delivery. J Control Rel 1998; 55: 143-52.
- Alagusundaram M, Chetty MSC, Umashankari K, Badarinath AV, Lavanya C and Ramkanth S: Microspheres as a novel drug delivery system- A review. Int J of Chem-Tech Res 2009; 1: 526-34.
- Rathone MJ: Human buccal absorption. A method for Estimating the transfer kinetics of drugs across the human buccal membrane. Int J Pharm 1991; 69: 103.
- Rathone MJ: Human buccal absorption I.A method for Estimating the transfer kinetics of drugs across the human buccal membrane. Int J Pharm 1991; 69: 103.
- Widder KJ, Sanyci AE, Ovadia H and Paterson PQ: Clin Immuno Immunopathol 1979; 14: 395.
- Mathews BR and Nixon JR: J Pharm Pharmacol 1974; 26: 283.
- https://www.researchgate.net/publication/221900556 Biodegradable Polymeric
- Prasad SG, Gupta VRM, Devanna N and Jayasurya K: Microspheres as drug delivery system – A review. Journal of Global Trends in Pharma Sci 2014; 5(3): 1961-1972.
How to cite this article:
Agnihotri N, Soni GC, Chanchal DK, Khan A and Tiwari S: A review on microsphereas a novel drug delivery system for multi-particulate drug release. Int J Life Sci & Rev 2019; 5(1): 6-15. doi: 10.13040/IJPSR.0975-8232.IJLSR.5(1).6-15.
All © 2015 are reserved by International Journal of Life Sciences and Review. This Journal licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.
Article Information
2
6-15
790
1536
English
IJLSR
N. Agnihotri *, G. C. Soni, D. K. Chanchal, A. Khan and S. Tiwari
Institute of Pharmacy, Bundelkhand University Jhansi, Uttar Pradesh, India.
nehaagnihotri1235@gmail.com
13 December 2018
20 January 2019
23 January 2019
10.13040/IJPSR.0975-8232.IJLSR.5(1).6-15
31 January 2019