A REVIEW ON TRANSDERMAL PATCHES AS A NOVEL DRUG DELIVERY SYSTEM
HTML Full TextREVIEW ON TRANSDERMAL PATCHES AS A NOVEL DRUG DELIVERY SYSTEM
Rohitashwa Sonkar * 1, S. K. Prajapati 1, Dilip Kumar Chanchal 2, Rohit Kumar Bijauliya 2 and Santosh Kumar 3
Department of Pharmaceutics 1, Department of Pharmacognosy 2, Institute of Pharmacy, Bundelkhand University, Jhansi - 284128, Uttar Pradesh, India.
Hygia Institute of Pharmaceutical Education and Research 3, Lucknow - 226020, Uttar Pradesh, India.
ABSTRACT: Transdermal drug delivery system was introduced to overcome the difficulties of drug delivery through the oral route. Despite their relatively higher costs, transdermal delivery systems have proved advantageous for delivery of selected drugs, such as estrogens, testosterone, clonidine, and nitro-glycerine. Transdermal delivery provides a leading edge over injectable and oral routes by increasing patient compliance and avoiding the first-pass metabolism, respectively. Topical administration of therapeutic agents offers many advantages over conventional oral and invasive methods of drug delivery. Skin is an effective medium from which absorption of the drug takes place and enters into systematic circulation over some time. The present article reviews the selection of drug and polymers suitable to be formulated as a transdermal system, advantages, disadvantages of formulation design, and the methods of preparation and evaluation of it.
Keywords: Transdermal, Delivery, Patches
INTRODUCTION: Nowadays, about 74% of drugs are taken orally and are found not to be as valuable as most wanted. To advance such characters, the transdermal drug delivery system emerged. With the creation of the current time of pharmaceutical dosage forms, transdermal drug delivery system (TDDS) recognized itself as an important part of novel drug delivery systems.
Transdermal dosage forms, still a costly alternative to conventional formulations, are becoming popular because of their exclusive advantages.Improved bioavailability, controlled absorption, extra uniform plasma levels, painless and reduced side effects easy application and flexibility of terminating drug administration by simply removing the patch to the skin are some of the potential advantages of transdermal drug delivery 1.
Oral Conventional dosage forms like tablets and capsules are the most widely used drug delivery system, but both dosage forms face the problem of gastric drug/enzyme instability first pass metabolism. Oral route has many further problems like the unpleasant taste, odor, and color.
Numerous additional problems are arising during taking pills; hence, problems are being faced during treatment. Sometimes patients become non-compliant. TDDS patches drugs are used by continuous release, so they show their effect for the exact duration and the transdermal patch is non-irritating and noninvasive technique. It is attractive alternative techniques over conservative techniques for systemic administration of drug 2.
The first transdermal drug delivery (TDD) system, Transderm-Scop developed in 1980, contained the drug Scopolamine for treatment of motion sickness. The transdermal device is a membrane-moderated system. The membrane in this system is a microporous polypropylene film. The drug reservoir is a solution of the drug in a mixture of mineral oil and polyisobutylene. This study release is maintained over three days 3.
A transdermal patch is defined as an adhesive medicated patch that is placed on to the above skin to deliver an exact dose of the drug through the skin into the bloodstream with a predetermined rate of release to reach in the body. Today the most common transdermal system present in the market mainly based on semi-permeable membranes which were called as patches. Transdermal drug delivery systems (TDDS), also known as “Transdermal patches” or “Skin patches” are dosage forms designed to deliver a therapeutically effective amount of drug across a patient‟s skin and in the bloodstream.
Transdermal drug delivery system has been in existence for a long time. Cardiovascular / anti-hypertensive diseases are the major concern of death in India. Cardiovascular diseases (CVD) are the cause of more than 30% of deaths, not only in the developed countries. The World Health Organization (WHO) estimates that low and middle-income countries are disproportionately affected. 82% of CVD deaths take place in low- and middle-income countries and occur almost equally in men and women.
To deliver therapeutic agents through the human skin for systemic effects of the cardiovascular/ antihypertensive diseases, the comprehensive morphological, biophysical, and physicochemical properties of the skin are to be considered 4.
Advantages of Transdermal Drug Delivery Systems: 5 Delivery via the transdermal route is an interesting option because the transdermal route is convenient and safe. The positive features of delivering drugs across the skin to achieve systemic effects are:
- Avoidance of first pass metabolism.
- Avoidance of gastro intestinal incompatibility.
- Predictable and extended duration of the activity.
- Minimizing undesirable side effects.
- Provides utilization of drugs with short biological half-lives, narrow therapeutic window.
- Improving physiological and pharmacological response.
- Avoiding the fluctuation in drug levels.
- Inter and intrapatient variations.
- Maintain the plasma concentration of potent drugs
- Termination of therapy is easy at any point in time.
- Greater patient compliance due to the elimination of multiple dosing profiles.
- Ability to deliver the drug more selectively to a specific site.
- Provide suitability for self-administration.
- Enhance therapeutic efficacy.
Disadvantages of TDDS: 6, 7, 8
- Some patients develop contact dermatitis at the site of application from one or more of the system components, necessitating discontinuation.
- Higher cost.
- Should not use the ionic drug.
- May cause allergic reactions.
- The molecular weight of less than 500 Da is essential.
- Sufficient aqueous and lipid solubility, a log P (octanol/ water) between 1 and 3 is required for permeate to transverse SC and underlying aqueous layers.
- Transdermal therapy is feasible for certain potent drugs only.
- Transdermal therapy is not feasible for ionic drugs.
- It cannot deliver the drug in a pulsatile fashion.
- Only relatively potent drugs are suitable candidates for transdermal delivery because of the natural limits of drug entry imposed by the skin’s impermeability.
Limitations of TDDS:
- Limited skin permeability.
- Restricted to the potent drug.
- Cannot use for a large molecule (>500 Dalton).
- Significant lag time.
- The difficulty for adhesion.
- The drug undergoes degradation in the skin.
- Variation in absorption efficiency at different sites of skin.
Conditions in which Transdermal Patches are used: 9 Transdermal patches are used when:
- When the patient has intolerable side effects (including constipation) and who is unable to take oral medication (dysphagia) and is requesting an alternative method of drug delivery.
- Where pain control might be improved by reliable administration, this might be useful in patients with cognitive impairment or those who for other reasons are not able to self-medicate with their analgesia.
- It can be used in combination with other enhancement strategies to produce synergistic effects.
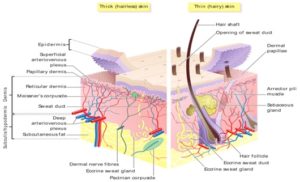
Anatomy and Physiology of Skin: 10, 11 Human skin comprises of three distinct but mutually dependent tissues:
A) The stratified, vascular, cellular epidermis,
B) The underlying dermis of connective tissues and
C) Hypodermis
FIG. 1: ANATOMICAL AND PHYSIOLOGICAL STRUCTURE OF SKIN
A) Epidermis: The multilayered epidermis varies in thickness, depending on cell size and number of cell layers of the epidermis, ranging from 0.8 mm on palms and soles down to 0.06 mm on the eyelids. It gives thickness, water permeability, and diffusivity of water through the epidermis. It consists of outer stratum corneum and viable epidermis.
1) Horney Layer (Stratum Corneum): This is the outermost layer of skin, also called a horney layer. It is approximately 10 μm thick when dry, but swells to several times this thickness when fully hydrated. It has 10 to 30 layers of dead, keratinized cells called corneocytes. There are three possible ways that drug molecules can pass through stratum corneum. The drug can be absorbed by various pathways through the skin depending on the physicochemical properties of the drug. Both lipophilic and hydrophilic drugs are absorbed from different routes like Transfollicular route, Trans-cellular route, and Intercellular route.
2) Viable Epidermis: This is situated beneath the outermost layer and varies in thickness ranging from 0.06 mm on the eyelids sole up to 0.8 mm on the palms. Going inwards, it consists of various layers as stratum granulosum, stratum lucidum, stratum spinosum and the stratum basal. In the basal layer, mitosis divisions of the cells constantly reproduce the epidermis, and this proliferation compensates the loss of dead horney cells from the skin surface.
B) Dermis: Dermis is 3 to 5 mm thick layer and is composed of a matrix of connective tissue, which contains blood vessels, lymph vessels, and nerves. The cutaneous blood supply has an essential function in the regulation of body temperature. It also provides nutrients and oxygen to the skin while removing toxins and waste products. Capillaries reach to within 0.2 mm of the skin surface and provide sink conditions for most molecules penetrating the skin barrier. The blood supply thus keeps the dermal concentration of a permeant very low, and the resulting concentration difference across the epidermis provides the essential concentration gradient for transdermal permeation.
C) Hypodermis: The hypodermis or subcutaneous fat tissue supports the dermis and epidermis. It serves as a fat storage area. This layer helps to regulate temperature, provides nutritional support and mechanical protection. It carries principal blood vessels and nerves to the skin and may contain sensory pressure organs.
Routes of Penetration: 12, 13 For a long term treatment and for a multi-dose treatment the most suitable system is transdermal drug delivery system because different patches are prepared for a long period of time in a suitable dose providing treatment. There are two diffusion pathways to penetrate a molecule in normal human skin: the appendageal and the transepidermal pathway. For the ions and large polar molecules, the appendageal route is best and for the unionized molecule which can cross the intact layer, the skin follow the transepidermal pathway. A molecule should have adequate lipophillicty and optimum molecular weight to penetrate in to the skin. Hydrophilic drugs partitioned preferentially via intercellular domains and the lipophilic permeants partitioned the subcutaneous via intercellular route. Most of the molecule traverses the stratum corneum by both routes. The transport of various drug molecules through the skin, promptly restricted by the barrier properties of the epidermis. To avoid these difficulties in permeation through SC, penetration enhancers are used.
General Features of Skin: 14
Color: Skin color represent an aggregate of the remitted and reflected light, the wavelength of which depends largely on the presence of 4 biochromes. Two biochromes are present in the epidermis:
Melanin: This is brown and absorbs UV and visible light.
Carotenoids: This is yellow.
Other two are present in the dermis.
Oxyhemoglobin: This is bright red found in arterioles and capillaries of the papillary layer.
Reduced Hemoglobin: This is bluish red and is found in the sub-papillary venous plexus.
Functions of Skin: 15, 16, 17 It is also called the integumentary system, which simply means “covering” the skin’s function go well beyond serving as a large, opaque bag for the body contents. It is essential because it keeps water and another precious molecule in the body. It also keeps water out structurally the skin is a marvel. It insulates and cushions the deeper body organs and protects the entire body from mechanical damage (bumps and cuts), chemical damage (from acid and base), UV radiation, sunlight, and bacteria.
Sensation: The skin contains abundant nerve ending and receptors that detect stimuli related to temperature, touch, pressure, and pain.
Regulation of Body Temperature: In response to high temperature or strenuous exercise, the evaporation of sweat from the skin surface helps lower elevated body temperature to normal body temperature. Changes in the flow of blood to the skin also help regulate body temperature.
Immunity: It provides immunologic information obtained during antigen processing to the appropriate effector cells in the lymphatic tissue.
Excretion: Excretion is carried out by exocrine secretions of sweat, sebaceous and apocrine gland. These excretory secretions are being utilized now to detect certain diseases of the blood.
Drug Delivery Route: Certain lipid-soluble substances and low molecular weight drugs have been successfully absorbed systemically, across the skin by transdermal patches, for example, nitroglycerin, hormones, nicotine, scopolamine, etc.
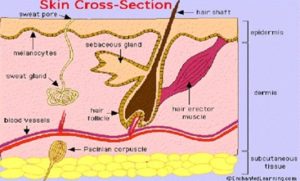
Drug Permeation Pathway: 10, 11 Percutaneous absorption involves passive diffusion of the substances through the skin.
FIG. 2: DRUG PENETRATION PATHWAYS ACROSS SKIN
A molecule may use two diffusional routes to penetrate normal intact skin, the appendageal route, and the epidermal route. There are critically three ways in which a drug molecule can cross the intact stratum corneum: via skin appendages (shunt routes); through the intercellular lipid domains; or by a transcellular route Fig. 2.
Appendageal Route: Appendageal route comprises transport via sweat glands and hair follicles with their associated sebaceous glands. These routes circumvent penetration through the stratum corneum and are therefore known as “shunt” routes. This route is considered to be of minor importance because of its relatively small area, approximately 0.1 % of the total skin area.
Epidermal Route:
1) Transcellular: Pathway means the transport of molecules across the epithelial cellular membrane. These include passive transport of small molecules, active transport of ionic and polar compounds and endocytosis and transcytosis of macromolecules.
2) Paracellular: Paracellular pathway means the transport of molecules around or between the cells. Tight junctions or similar situations exist between the cells. The principal pathway taken by a permeant is decided mainly by the partition coefficient (log k). Hydrophilic drugs partition preferentially into the intracellular domains, whereas lipophilic permeants traverse the stratum corneum via the intercellular route. Most permeants permeate the stratum corneum by both routes. However, the tortuous intercellular pathway is widely considered to provide the principal route and major barrier to the permeation of most the drugs.
Transdermal Patches: 18, 19 A transdermal patch is a medicated adhesive patch that is placed on the skin to deliver a specific dose of medication through the skin and into the bloodstream. In this system, drug therapy can be stopped instantly in a situation where drug input is no longer desirable. The system allows reducing the frequency of dosing, which is particularly favorable for the compound with short biological half-life. The transdermal drug delivery is affected by limitations as well that is due to the primary function of human skin.
Several drugs can be administered transdermally. For example, scopolamine patches to check motion sickness and fentanyl patches to treat cancer pain or chronic pain syndromes are being used currently by the transdermal route.
Types of Transdermal Patches: 20, 21, 22
Single Layer Drug in Adhesive: In this type of patches, the adhesive layer contains drugs. Here the property of the adhesive layer does not only adhere to the various layer together, although this type of layer is responsible for releasing the drug to the skin. The backing and the temporary liner is surrounded on the adhesive layer.
Multi-layer Drug in Adhesive: The multi-layer drug in the adhesive is similar to the single layer, but it contains an immediate drug release layer which is different from another layer which will be a controlled release along with the adhesive layer. For releasing the drug, the adhesive layer is responsible. This patch also has a temporary liner-layer and a permanent backing.
Vapour Patch: The adhesive layer in this patch not only serve as adhere the various layer together but also serves a market, commonly used for releasing of essential oils in decongestion. Other marketed vapor patches are used to improve the quality of sleep and reduces cigarette smoking conditions.
Reservoir System: In this system, the two layers; an impervious backing layer and a rate controlling membrane are responsible for embedded drug reservoir in between them. There is a rate controlling membrane which can be microporous or nonporous which is responsible for the drug release. The drug in the form of suspension, gel, solution, or dispersed in a solid polymer matrix. The hypoallergenic adhesive polymer can be applied as an outer surface polymeric membrane, which is compatible with the drug.
Microreservoir System: This system is the combination of the reservoir and matrix-dispersion system. By suspending the drug in an aqueous solution of a water-soluble polymer, the drug reservoir is formed and then dispersing the solution homogeneously in a lipophilic polymer to form thousand of unreachable, microscopic spheres of drug reservoirs.
By immediately crosslinking the polymer in situ b using cross-linking agent, this thermodynamically unstable dispersion is stabilized.
Matrix System:
Drug-in-adhesive System: The drug reservoir is formed by dispersing the drug in an adhesive polymer and then spreading the medicated adhesive polymer by solvent casting or melting on an impervious backing layer. For protection purpose, the unmediated adhesive polymer layers are applied on top of the reservoir.
Matrix-dispersion System: In hydrophilic or lipophilic polymer matrix, the drug is homogeneously dispersed. This drug containing polymer disk is fixed on to an occlusive base plate in a compartment fabricated from a drug impermeable backing layer. To form a strip of adhesive film, applying the adhesive on the face of the drug reservoir, it is spread along with the circumference.
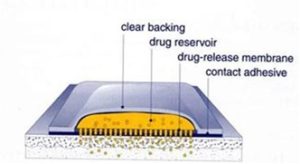
Formulation Design: 23, 24, 25, 26, 27 A transdermal therapeutic system is essentially a multi-laminate structure that is composed of the following constituents:
- Drug
- Polymer matrix
- Penetration enhancers
- Adhesives
- Backing membrane
- Release linear
FIG. 3: DIFFERENT LAYERS OF TRANSDERMAL PATCHES
Drug: Transdermal route of administration cannot be employed for all types of drugs. It depends upon optimal physicochemical properties of the drug, its biological properties. Also, consideration of the pharmacokinetic and pharmacodynamic properties of the drug is necessary. The most important requirement of the drug to be delivered transdermally is demonstrated by the need for controlled delivery, such as short half-life, an adverse effect associated with other route or a complex oral or i.v. dose regimen. The drug parameter required for ideal drug candidate for transdermal drug delivery as above:
Polymer: Advances in transdermal drug delivery technology have been rapid because of the sophistication of polymer science that now allows incorporation of polymers in the transdermal system (TDS) in adequate quantity. The release rate from TDS can be tailored by varying polymer composition. Selection of polymeric membrane is very important in designing a variety of membrane permeation controlled TDS.
The Criteria for the Polymers are:
- The polymer should be chemically non-reactive, or it should be an inert drug carrier;
- The polymer must not decompose on storage or during the life span;
- Molecular weight, physical characteristic and chemical functionality of the polymer must allow the diffusion of the drug substance at a desirable rate;
- The polymer and its decomposed product should be nontoxic. It should be biocompatible with skin;
- The polymer must be easy to manufacture and fabricate into the desired product. It should allow incorporation of large amounts of the active agent.
Penetration Enhancer: To increase the permeability of stratum corneum to attain higher therapeutic levels of the drug penetration enhancers interact with structural components of stratum corneum, i.e. proteins or lipids. The enhancement in the absorption of oil-soluble drugs is apparently due to the partial leaching of the epidermal lipids by the chemical enhancers, resulting in the improvement of the skin conditions for wetting and transepidermal and transfollicular penetration. The miscibility and solution properties of the enhancers used could be responsible for the enhanced transdermal permeation of water-soluble drugs. Pharmaceutical scientists have made great efforts in transdermal permeation studies using various enhancers for several drug moieties. Sorption promotes act by interaction with intracellular lipids leading to disruption of their organization and increasing their fluidity. Some of them also interact with the intercellular protein, keratin denaturation (azone and oleic acid) while others act by both mechanism (DMSO and propylene glycol). Another possible mechanism is by altering the skin hydration.
Ideal Penetration Enhancers Should Possess the Following Properties:
- They should be non-toxic, non-irritating, and non-allergic.
- They would ideally work rapidly; the activity and duration of effect should be both predictable and reproducible.
- They should have no pharmacological activity within the body.
- The penetration enhancers should work uni-directionally, e., they should allow therapeutic agents into the body whilst preventing the loss of endogenous materials from the body.
- When removed from the skin, barrier properties should return both rapidly and fully to normal.
- They should be cosmetically acceptable with an appropriate skin feel.
- Pharmacologically inert.
Adhesive Layer: The adhesive must possess sufficient property to firmly secure the system to the skin surface and to maintain it in position for as long as desired, even in the presence of water. After removal of the patch, any traces of adhesive left behind must be capable of being washed with water and soap. Pressure sensitive adhesives are used to achieve contact between the transdermal patch and the skin.
Adhesion is understood to be the Net Effect of Three Phenomena, namely:
Peel: The resistance against the breakage of the adhesive bond.
Track: The ability of a polymer to adhere to a substrate with little contact Pressure and
Creep: The viscous relaxation of the adhesive bond upon shear.
The Ideal Characters of Adhesive Materials are:
- High biocompatibility (low irritancy, toxicity, allergic reaction, etc.);
- Good adhesive to oily, wet, wrinkled and hairy skin;
- Good environment resistance against water and humidity;
- Easy to remove from the skin;
- The high permeability of moisture to avoid excessive occlusion and for the drug itself and;
- Non-reactive towards the drug.
There are Three Types of Adhesive Used Mainly:
- Silicone type adhesive;
- Polyisobutylene adhesive and;
- Poly-acrylate based adhesive.
Backing Layer: The backing layer must be impermeable to drug and permeation enhancers. The backing membrane serves the purpose of holding the entire system together and at the same time protects the drug reservoir from exposure to the atmosphere, which could result in the breakage or loss of the drug by volatilization.
The most commonly used backing materials are polyester, aluminized poly-ethylene terapthalate, siliconised polyethylene terapthalate, and aluminum foil of metalized polyester laminated with polyethylene.
Release Liner: The peel strip prevents the loss of the drug that has migrated into the adhesive layer during storage and protects the finished device against contamination. Polyesters foils and other metalized laminates are typical materials which are commonly used.
Preparation of Transdermal Patches: Trans-dermal drug delivery patches can be prepared by various methods:
Mercury Substrate Method: 28, 29, 30, 31, 32 In this method the required the amount of drug is dissolved in a predetermined amount of polymer solution along with plasticizer. The above solution is to be stirred for some time to produce a homogenous dispersion, and it is kept aside until air bobbles removed completely and then poured into a glass ring which is placed over the mercury surface in a glass petri dish. The rate of evaporation of the solvent is controlled by placing an inverted funnel over the petri dish. The dried films are to be stored in desiccators.
Circular Teflon Mould Method: 33, 34 Solutions containing polymers in various ratios are used in an organic solvent. The calculated amount of drug is dissolved in half the quantity of the same organic solvent. Plasticizer added into the drug-polymer solution. The total contents are to be stirred and then poured into a circular Teflon mould. And the rate of solvent vaporization controlled with placing inverted glass funnel on Teflon mould. The solvent is allowed to evaporate for 24 h. The dried films are to be stored in desiccators.
Glass Substrate Method: 35-42 The polymeric solutions are kept aside for swelling, then the required quantity of plasticizer and drug solution are added and stirred for 10 min. Further, it is set-aside for some time to exclude any entrapped air and is then poured in a clean and dry an umbra Petri plate. The rate of solvent evaporation is controlled by inverting a glass funnel over the Petri plate. After overnight, the dried films are taken out and stored in desiccators.
Using IPM Membranes Method: 40, 41 In this method, the drug is dispersed in a mixture of water and propylene glycol containing carbomer 940 polymers and stirred for 12 h in magnetic stirrer. The dispersion is to be neutralized and made viscous by the addition of triethanolamine. Buffer pH 7.4 can be used to obtain solution gel if the drug solubility in aqueous solution is very poor. The formed gel will be incorporated in the IPM membrane.
By Using EVAC Membranes Method: 40, 41 To prepare the target transdermal therapeutic system, 1% carbopol reservoir gel, polyethylene (PE), ethylene vinyl acetate copolymer (EVAC) membranes can be used as rate control membranes. If the drug is not soluble in water, propylene glycol is used for the preparation of gel. The drug is dissolved in propylene glycol; carbopol resin will be added to the above solution and neutralized by using 5% w/w sodium hydroxide solution. The drug (in gel form) is placed on a sheet of a backing layer covering the specified area. A rate-controlling membrane will be placed over the gel, and the edges will be sealed by heat to obtain a leak-proof device.
Aluminium Backed Adhesive Film Method: 42, 43 Transdermal drug delivery system may produce unstable matrices if the loading dose is greater than 10 mg. Aluminum backed adhesive film method is a suitable one.
For the preparation of same, chloroform is the choice of solvent, because most of the drugs, as well as adhesive, are soluble in chloroform. The drug is dissolved in chloroform, and adhesive material will be added to the drug solution and dissolved. A custom made aluminum former is lined with aluminum foil and the ends blanked off with tightly fitting cork blocks.
Asymmetric TPX Membrane Method: 40 A prototype patches can be fabricated by a heat sealable polyester film (type 1009, 3m) with a concave of 1 cm diameter used as the backing membrane. Drug sample is dispensed into the concave membrane, covered by a TPX {poly (4-methyl-1-pentene)} asymmetric membrane, and sealed by an adhesive.
Evaluation Test of Transdermal Patch:
Mass Uniformity and Thickness: 44, 45 The film of patches was dried at 60 °C for 4 h. Then the patches were weighed directly on the digital balance for the determination of mass and thickness of each patch at five different locations were done by using screw gauge.
Folding Endurance: 46, 47 Folding endurance was determined by repeatedly folding the patch at the same place up to 200 times without breaking or until it breaks. The number of times the film could be folded at the same place without breaking was the folding endurance.
Drug Content Determination: 48, 49 For a drug content uniformity, the patch was dissolved in a 100 ml of isotonic phosphate buffer saline pH 7.4 for 12 hours under occasional stirring. The contents were filtered by Whatman filter paper, and then the filtrate was analyzed by U.V spectrophotometer at a specific λ max.
Flexibility or Tensile Strength: 50 The pulley system is used to determine the flexibility and tensile strength. With the help of two small catchers, the patch was pulled in the opposite direction by gradually increasing the force until the patch was broken. The tensile strength was noted from the scale of the pulley in kg/cm2.
Elongation Break: 51 For elongation break study the longitudinal strips were cut from the transdermal patch. By using vernier calliper the flatness was determined at various points. By noting the length just before the breakpoint the percentage elongation was determined and substituted in the equation.
Elongation (%) = L1 - L2 / L2 × 100
Where, L1 = final length of each strip, L2 = initial length of each strip
Percentage of Moisture Uptake: 52, 53 To check the physical stability of film in high humidity condition the film was placed in a desiccator containing a solution of aluminum chloride at room temperature for 24 h and exposed to 85% relative humidity until a constant weight of film was obtained. The film was reweighed, and the percentage of moisture uptake was determined by the following formula.
Percentage moisture uptake = Final weight – Initial weight / Initial weight × 100
Percentage Moisture Loss: 54, 55 To check the extent of moisture loss, the films were weighed accurately and kept in the desiccators containing anhydrous calcium chloride. After 3 days the film was carried out and reweighed.
Percentage of moisture loss = Initial weight – Final weight / Initial weight × 100
Water Vapour Transmission Rate (WVT) Studies 56 It is defined as the quantity of moisture transmitted through a unit area of film in unit time. Glass vials of equal diameter were used as transmission cells. The film was placed on the vials, containing 3 g of fused calcium chloride as a desiccant, with an adhesive tape. The cell was accurately weighed and kept in a desiccator containing a saturated solution of potassium chloride to maintain RH of 84%. The vials were taken out and weighed at every 24 h interval for a period of 72 h.
WVT = Final weight – Initial weight / Area
The rate of water vapor transmission was calculated from the plot of the amount of water vapor transmitted vs. time.
Differential Scanning Calorimetry: 58 The DSC analysis of drug, cholesterol, span 60, HPMC K4M, liposomal patch, niosomal patch, and control patch were carried out. The analysis was conducted in the heating range of 54 - 260 °C in a nitrogen atmosphere at the rate of 150 ml/min using DSC.
Surface Morphology Study: 59, 60 This study of formulated transdermal patches (both stable and unstable) was carried out by scanning electron microscope (SEM) at 15 kV under different magnification. SEM was confirmed by direct deposition of a film on double-sided carbon tape and coated with gold. The sample was visualized using an SEM operated with a secondary detector at different acceleration voltage and a different magnification.
Flatness: 61 The strips were cut out longitudinally from each film, i.e. one from the center and two from either side. The length of each strip was measured and the variation in the length because of non-uniformity in flatness was measured by determining percent constriction, considering 0% constriction is equivalent to 100% flatness:
% Constriction = L1 - L2 / L2 × 100
Where, L1= initial length of each strip, L2= final length of each strip
Stability Study: 62 For stability study, the prepared patch were sealed in polyethylene coated aluminum foils and kept at 25 °C for a few months. The samples were withdrawn at 0, 30, 60, and 90 days, and the drug content was analyzed by a UV spectrophotometer method.
Physical Appearance: 63 The patches were visually inspected for color, flexibility, smoothness, and homogeneity.
Skin Irritation Study: 64 Skin irritation and sensitization testing can be performed on healthy rabbits (average weight of 1.2 to 1.5 kg). The dorsal surface (50 cm2) of the rabbit is to be cleaned and remove the hair from the clean dorsal surface by shaving and clean the surface by using rectified spirit and the representative formulations can be applied over the skin.
The patch is to be removed after 24 h, and the skin is to be observed and classified into 5 grades based on the severity of skin injury.
CONCLUSION: Transdermal drug delivery systems represent a beneficial innovation for drug delivery, particularly in patients who cannot swallow or remember to take their medications. Topical administration of therapeutic agents offers various advantages over conventional oral and invasive methods of drug delivery. Various important advantages of transdermal drug delivery are a limitation of hepatic first-pass metabolism, enhancement of therapeutic efficiency, and maintenance of steady plasma level of the drug. This article provides important information about the formulation and evaluation with respect to transdermal drug delivery systems. TDDS is a practical application as the next generation of drug delivery system for human.
ACKNOWLEDGEMENT: The authors thankful to Professor Dr. S. K. Prajapati and Dr. Shashi Alok for his valuable guidance.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Ahmed A, Karki N, Charde R, Charde M and Ganghare B: Transdermal drug delivery system an overview. Int J Biomed Adv Res 2011; 2: 38-56.
- Patel A, Visht S and Sharma PK: Transdermal drug delivery system: Next generation patches. J Drug Discov Dev 1: 43-65.
- Chowdary KPR and Naidu RAS: Transdermal drug delivery, a review of current status. Indian Drugs 1995, 32(9): 414-422.
- Vyas SP and Khar RK: Controlled drug delivery: Concepts and advances, Vallabh Prakashan, Edition 1st, 2002: 411-445.
- Hadgraft J and Guy R: In; Transdermal drug delivery, Marcel Dekker, Inc., New York and Basel, Vol. 35, 296.
- Barry BW: Dermatological Formulations: Percutaneous absorption. Drugs and Pharmaceutical Sciences. Marcel Dekker, Inc. Vol. 18, 1983: 1-39.
- Aulton ME: Pharmaceutics: The science of dosage form design. Dosage form design and manufacture Transdermal Drug Delivery, Edition 2nd, Part 4th, 2007: 499 -533.
- Merkle HP: Transdermal delivery system: Methods find. Exp Clin Pharmacol 1989; 11: 135-35.
- Mahato RA: Pharmaceutical dosage forms and drug delivery, published by CRS Press, Boca Raton, 2002: 196-197.
- Finnin BC and Morgan TM: Transdermal penetration. J Pharm Sci 1999; 88(10): 955-958.
- Allen LV, Popovich NG and Ansel HC: Ansel‟s pharma-ceutical dosage forms and drug delivery systems, Lippincott Williams and Wilkins, Edition 8th, 2005: 298-315.
- Rani S, Saroha K, Syan N and Mathur P: Der Pharmacia Sinica 2011; 2(5): 17-29.
- Nandy BC, Chourasia SK, Roy S, Mazumdar B, Meena KC, Aujha D, Makhija M and Pathak K: Der Pharmacia Sinica 2011; 2(4): 203-217.
- Freedberg IM, Eisen AZ, Wolf K, Austen KF, Goldsmith LA and Katz SI: Fitzpatrick’s dermatology in general medicine, Mc Graw-Hill Medical publishing division, Edition 6th, Vol. 1, 2003: 58-85.
- Mzrieb EN: Human anatomy and physiology. Dorling Kindersley Pvt. Ltd., India, Edition 6th, 2006: 152-157.
- Gaikwad AK: Transdermal drug delivery system: Formulation aspects and evaluation. Compr J Pharm Sci 2013; 1(1): 1-10.
- Marieb EN: Essential of human anatomy and physiology, Published by Dorling Kindersley Pvt. Ltd., India, Edition 8th, 2006: 110-116.
- Dhiman S, Thakur GS and Rehni AK: Transdermal patches: a recent approach to new drug delivery system. Int J Pharm Pharm Sci 2011; 3(5): 26-34.
- Vizseralek G, Berko S, Toth G, Balogh R, Szucs MB, Csanyi B, Sinko B and Novak KT: Eur J Pharm Sci 2015; 1-8.
- Sharma A, Saini S and Rana AC: Transdermal drug delivery system: A review. Int J Res Pharm Biomed Sci 2013; 4(1): 286-292.
- Patel MP and Gupta MM: Pharm Innov J 2013; 2(3): 149-165.
- Kumar JA, Pullakandam N, Prabu SL and Gopal V: Int J Pharm Sci Rev Res 2010; 3(2): 49-54.
- Barry BW: Mode of action of penetration enhancers on the kinetics of percutaneous absorption. J Control Release 1987; 6: 43-51.
- Qvist MH, Hoeck U, Kreilgaard B, Madson F and Frokjaer S: Release of chemical penetration enhancers from drug-in-adhesive transdermal patches. Int J Pharm 2002: 231: 253-263.
- Govil SK, Radnic EM, Sterner DG: US Patent 1993; 5: 262, 165.
- Govil SK: In Drug delivery devices, Marcel Dekker, New York 1988: 388
- Guy, Richard H, Hadgraft and Jonathan; Transdermal drug delivery Published by Informa Health Care Edition 2nd, 2002: 322.
- Aqil M, Sultana Y and Ali A: Matrix-type transdermal drug delivery systems of metoprolol tartrate, in-vitro Acta Pharm 2003; 53: 119-125.
- Basubramanian V, Iyer, Ravindra C and Vasavada: Evaluation of lanolin alcohol films and kinetics of triamcinolone acetonide release. Journal of Pharmaceutical Sciences 1979; 68(6): 119-125.
- Chowdary KPR and Naidu RAS: Preparation and evaluation of cellulose acetate films as rate controlling membranes for transdermal use. Indian Drugs 1991; 29 (7): 312-315.
- Mamatha T, Venkateswara Rao J and Mukkanti K: Development of matrix type transdermal patches of lercanidipine hydrochloride, physicochemical and in-vitro DARU 2010; 18(1): 9-16.
- Sridevi S, Chary MG, Krishna DR, Prakash V and Diwan: Pharmacodynamic evaluation of transdermal drug delivery system of glibenclamide in rats. Indian Journal of Pharmacology 2000; 32: 309-312.
- Sharma T and Rawal G: transdermal therapeutic systems- An overview. International Journal of Pharmaceutical and Biological Archives 2011; 2(6): 1581-1587.
- Wiechers J: Use of chemical penetration enhancers in transdermal drug delivery-possibilities and difficulties. Acta Pharm 1992; 4: 123.
- Manvi FV, Dandagi PM, Gadad AP, Mastiholimat VS and Jagdeesh T: Formulation of transdermal drug delivery system of ketotifen fumarate. IJPS 2003; 65(3): 239-243.
- Kanikkannan N, Jayaswal SB and Singh J: Transdermal delivery of indomethacin: Release profile of drug from polymeric patches. Indian Drugs 30(9): 441-445.
- Sankar V, Velrajan G, Palaniappan R and Rajasekar S: Design and evaluation of nifedipine transdermal patches. IJPS 2003; 65(5): 510-515.
- Ryan F, Donnelly, Paul A and McCarron: Design and physicochemical characterization of a bioadhesive patch for dose-controlled topical delivery of imiquimod. International Jour of Pharmaceutics 2006; 307: 318-325.
- Patel HJ, Patel JS and Patel KD: Transdermal patch for ketotifen fumarate as an asthmatic drug. IJPR 2009; l(1): 1297-1304.
- Rani S, Saroha K and Syan N: Transdermal patches a successful tool in transdermal drug delivery system: An overview. Der Pharmacia Sinica 2011; 2(5): 17-29.
- Ahmed A, Karki N, Charde R, Charde M and Gandhare B: Transdermal drug delivery systems- an overview. International Journal of Biomedical and Advance Research 2011; 02(01): 38-56.
- Parthasarathy G, Bhaskar reddy K and Prasanth VV: Formulation and characterization of transdermal patches of naproxen with various polymers. International Journal of Comprehensive Pharmacy 2011; 6 (07): 1-3.
- Kavitha K and Rajendra MM: Design and evaluation of transdermal films of lornoxicam. IJPBS 2011; 2(2): 54-62.
- Nazarkar S, Kondawar M, Prasad V, Khedkar S and Dayama D: Int J Curr Pharm Res 2014; 6(4): 32-36.
- Mutalik S and Udupa N: J Pharm Sci 2004; 93: 1577-1594.
- Kumar SR, Jain A and Satish N: Der Pharm L 2012; 4(1): 330-343.
- Vaja D, Seth AK, Sailor GU, Patel J, Patel J, Pandya K, Patel M, Ghelani TK and Joshi U: Int J Pharm Sci 2011; 2(4): 89-103.
- Kumar JRK, Murlidharan S, Dhanaraj SA: J Pharm Sci Res 2012; 4(6): 1840-1843.
- Nawaz A, Khan GM, Shah SU, Shah SU, Rehman A, Shah KU and Hussain A: Pak Acad Sci 2011; 48(2): 95-100.
- Sarkar G, Saha NR, Roy I, Bhattacharyya A, Bose M, Mishra R, Rana D, Bhattacharjee A and Chattopadhyay D: Int J Biol Macromol 2014; 66: 158-165.
- Mohd A, Mohd E, Chand S, Hanifa, Sabreesh M, Asia R and Kumar GS: Adv Res Pharm Biol 2011; 1(2): 109-119.
- Yadav SK, Laxmi MV and Krishna JV: J Global Trends Pharm Sci 2013; 4(1): 999-1006.
- Koteswararao P, Duraivel S Kumar KPS and Bhowmik D: Indian J Res Pharm Biotechnol 2013; 1(5): 629-634.
- Ganju G, Ganju K and Pathak AK: Int J Res Pharm Chem 2011; 1(4): 1115-1118.
- Oza NA, Patadiya DD, Patel PU and Patel DM: Int J Res Pharm Chem 2013; 2(1): 151-162.
- Reddy MV, Reddy VJ, Ramesh Y and Venkateswarlu I: Int J Inst Pharm Life Sci 2011; 1(1): 18-29.
- Akhtar N, Arkvanshi S, Bhattacharya SS, Verma A and Pathak K: J Liposome Res 2014; 1-11.
- Ali MM and Rajab NA: World J Pharm Res 2014; 3(7): 50-70.
- Panchaxari DM, Pampana S, Pal T, Devabhaktuni B and Aravapalli AK: DARU J Pharm Sci 2013; 21(6): 1-14.
- Arora P and Mukherjee B: J Pharm Sci 2002; 91: 2076-2089.
- Jamakandi VG, Mulla JS, Vinay BL and Shivakumar HN: Asian J Pharm 2009; 59-64.
- Patel KN, Patel HK and Patel VA: Int J Pharm Pharm Sci 2012; 4(1): 296-299.
- Nair RS, Ling TN and Shukkoor MSA: J Pharm Res 2013; 6: 774-779.
- Gavali P, Gaikwad A, Radhika PR and Sivakumar T: Design and development of hydroxypropyl methyl cellulose based polymeric film of enalapril maleate. International Journal of Pharmtech Research 2010; 2(1): 274-282.
How to cite this article:
Sonkar R, Prajapati SK, Chanchal DK, Bijauliya RK and Kumar S: A review on transdermal patches as a novel drug delivery system. Int J Life Sci & Rev 2018; 4(4): 52-62. doi: 10.13040/IJPSR.0975-8232.IJLSR.4(4).52-62.
Article Information
1
52-62
728
1282
English
IJLSR
R. Sonkar *, S. K. Prajapati, D. K. Chanchal, R. K. Bijauliya and S. Kumar
Department of Pharmaceutics, Institute of Pharmacy, Bundelkhand University, Jhansi, Uttar Pradesh, India.
rohitashwasonkar06@gmail.com
11 January 2018
28 March 2018
23 April 2018
10.13040/IJPSR.0975-8232.IJLSR.4(4).52-62
30 April 2018