FAST DISSOLVING ORAL FILM: OVERVIEW
HTML Full TextFAST DISSOLVING ORAL FILM: OVERVIEW
Dalia Abdelaty Mostafa
October University for Modern Science and Art, Cairo, Egypt.
ABSTRACT: The purpose of the current review is to enlighten the present and the future perspective on oral fast dissolving films (OFDFs) as a drug delivery system as they are gaining interest as a substitute of fast dissolving tablets. Tablets/capsules to modified release tablets/capsules to oral disintegrating tablet to wafer to the recent development of fast dissolving oral films. Fast dissolving drug delivery systems were first invented in the late 1970s to overcome swallowing difficulties associated with tablets and capsules for pediatric and geriatric patients. Also, solid oral delivery systems do not require sterile conditions and are therefore less expensive to manufacture, but oral drug delivery systems still need some advancements to be made because of their drawbacks related to particular class of patients which includes geriatric, pediatric and dysphagia patients associated with many medical conditions as they have difficulty in swallowing or chewing solid dosage forms. Many pediatric and geriatric patients are unwilling to take solid preparations due to fear of choking because of tablet appearance, and patients experienced difficulty in swallowing tablets. Even fast dissolving tablets, there is a fear of choking due to its tablet type appearance. For the last two decades, there had been an enhanced demand for patient-compliant dosage forms. Research and development in the oral drug delivery segment had led to the transition of dosage form from simple conventional tablets/capsules to modified release tablets/capsules too. The oral disintegrating tablet (ODT) to wafer to the recent development of oral dissolving film, "a thin film that is prepared using hydrophilic polymers that rapidly dissolves on the tongue or buccal cavity." Fast dissolving oral films (FDOFs) are the most advanced form of oral solid dosage form due to more flexibility and comfort. It improves the efficacy of APIs by dissolving within a minute in the oral cavity after the contact with saliva without chewing and no need of water for administration. It gives quick absorption, and instant bioavailability of drugs due to high blood flow, and permeability of oral mucosa is 4-1000 times greater than that of skin. Fast dissolving oral films are fast disintegrating thin films having an area ranging from 5 to 20 cm2 in which drug is incorporated in the form of a matrix using hydrophilic polymer. The active pharmaceutical ingredient can be incorporated up to 15 mg along with other excipients, i.e., plasticizers, colorants, sweeteners, taste masking agents, etc. Plasticizer increases workability, spreadability, and flexibility of films, thereby reducing the glass transition temperature of polymers. Fast dissolving films are very similar to an ultra-thin strip of a postage stamp in their shape, size and thickness. They quickly disintegrate and dissolve, and there is no need for water for their administration, making them suitable for pediatric and geriatric patients. The solvent casting method is commonly used for OFDFs preparation. Films prepared should be evaluated for organoleptic properties, thickness, tackiness, tensile strength, folding endurance, disintegration, and in-vitro drug release.
Keywords: Fast dissolving film, Folding endurance, Disintegration test time, Dissolution
INTRODUCTION: The oral route is the most popular route for the administration of therapeutic agents.
The epithelial lining of the oral cavity differs in type (keratinized and non-keratinized), and thickness in different areas, these differences give rise to regional variation in permeability of drugs 1.
The buccal mucosa is a promising delivery route for drug administration. It offers several advantages as the mucosa is well supplied with both vascular and lymphatic drainage. First pass metabolism in liver and pre-systemic elimination in the gastrointestinal tract (GIT) is avoided.
With the right dosage form design and formulation, the permeability and the local environment of the mucosa can be controlled and manipulated to accommodate drug permeation. Oral fast dissolving films (OFDFs) will provide an increase in the bioavailability, low cost of therapy, and ease of administration, which will lead to patient compliance 2, 3. OFDFs administered through buccal route to increase patient compliance, achieve minimum dissolution time in oral, buccal mucosa to reach systemic circulation with the fastest onset of action, solve a specific defect of other dosage form and formulating a film dosage form alternative to capsules and tablets 4. Among the different routes of drug delivery, the oral route is nominated to be the most favored and highly accepted to patients and physicians 5. The buccal region is one of the most convenient and easily approachable routes of administration for the delivery of the therapeutic agents to be used locally and systemically. The mucosa is considered one of the potential sites of drug administration. Trans-mucosal routes of drug delivery offer important advantages over oral administration 6.
The buccal drug delivery system prolongs the residence time of dosage form at the site of action, improves the therapeutic performance of the drug, and provides a better enzymatic flora for drug absorption. It provides direct entry of the drug into the systemic circulation, therefore, avoid all the drawbacks of the per-oral administration of drugs as hepatic first-pass metabolism, pre-systemic elimination of GIT by enzymatic degradation, as shown in Table 1, that prohibit oral administration of certain types of drugs especially proteins and peptides. The mucosa is relatively permeable and rich with blood supply, it is firm, strong, and it shows short recovery times after stress or damage. The important point is that the lack of Langerhans cells makes the buccal mucosa tolerant to potential allergies 7.
TABLE 1: SHOWING ENZYMATIC DEGRADATION IN GIT 7
| Major digestive enzymes | |||
| Enzyme | Produced in | Site of release | pH level |
| Carbohydrate digestion | |||
| Salivary amylase | Salivary gland | Mouth | Neutral |
| Pancreatic amylase | Pancreas | Small intestine | Basic |
| Maltase | Small intestine | Small intestine | Basic |
| Protein digestion | |||
| Pepsin | Gastric Glands | Stomach | Acidic |
| Trypsin | Pancreas | Small intestine | Basic |
| Peptidases | Small intestine | Small intestine | Basic |
| Nucleic acid digestion | |||
| Nuclease | Pancreas | Small intestine | Basic |
| Nucleosidases | Pancreas | Small intestine | Basic |
| Fat digestion | |||
| Lipase | Pancreas | Small intestine | Basic |
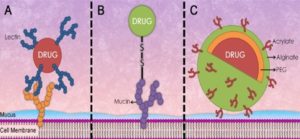
FIG. 1: INTERFACIAL ATTRACTION FORCE BY MEMBRANE COATING GRANULES (MCG) 9
The buccal cavity is an attractive and achievable site for systemic drug delivery as it increases the bioavailability. Bio-adhesion can be described as a phenomenon of interfacial attraction forces, in which two materials are held together, it occurs between the surfaces of biological, subtract of the natural or synthetic polymers, which allows the polymer to adhere to the biological surface for an extended period as shown in Fig. 1. Generally, bio-adhesion is a term that indicates adhesive interactions with biological or non-biological derived substances 8, 9.
Benefits of the Buccal Route: Buccal drug delivery has high patient acceptance compared to other routes of drug administration. It has a rapid onset of action unlike the oral route, also it helps the avoidance of the pain associated with injections, and it is more accessible for administration and the removal of the dosage form. Buccal drug delivery provides more quick and effective absorption 6, 2. Side effects of the oral route will be avoided as nausea and vomiting. The vital point is that drug absorption can be achieved in cases of the unconscious, less co-operative patients, and emergency cases 5, 4. Drugs that show poor bioavailability via oral route, can be administered conveniently, as drugs that are unstable in acidic environment of the stomach or destroyed by the enzymes or the alkaline environment of the intestine can be administered through the buccal cavity as it will also show obvious reduction of side effects that are related to the dose 6, 2. Buccal drug delivery allows the drug to directly enter the systemic circulation; it also provides a passive system that doesn’t require activation. Buccal delivery drugs must have the ability to withstand extremes of environment like changes in temperature and pH. It can deliver peptide molecules that are unstable to be administered by the oral route. Buccal drug delivery system can be used as sustained drug delivery 7, 3.
Limitations on the Drugs to be Administered by the Buccal Route: Some drugs, if taken by the buccal route may cause irritation of the buccal mucosa, allergic reactions, discoloration of the teeth, some of them have unpleasant or bitter taste, awful odor and unstable in the buccal pH; these drugs cannot be administered by this route. Also swallowing of saliva can lead to loss of suspended or dissolved drug particles. Once the dosage form is placed on the absorption site, it should not be disturbed neither by eating nor drinking; drawbacks can happen as the patient might swallow the formulation. Buccal drug delivery is mainly used only with drugs absorbed by passive diffusion. If the formulation of the drug contains the antimicrobial agent, it will affect the natural microbes present in the buccal cavity. Also, low permeability of the buccal membrane compared to the sublingual membrane and continuous secretion of saliva may lead to dilution of the drug 6, 2.
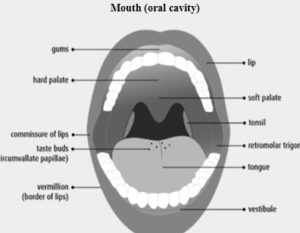
Anatomy of the Buccal Mucosa: The buccal region is a part of the mouth, bounded anteriorly and laterally by lips and cheeks, posteriorly and medially by the teeth and gums and above and below by the reflection of mucosa from the lips and cheeks to gum as shown in Fig. 2 8.
FIG. 2: ANATOMY OF THE MOUTH 8
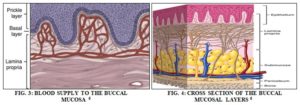
Maxillary artery supplies blood to the buccal mucosa, and blood flow is faster and richer (2.4 ml/min/ cm2) than that in sublingual and gingival regions, therefore it facilitates the passive diffusion of drug molecules across the mucosa as shown in Fig. 3 6.
The buccal mucosa is composed of several layers with different cells, it is composed of an outermost layer of stratified squamous epithelium, below it lies a basement membrane, and then an intermediate layer called lamina propria followed by the innermost layer called submucosa as shown in Fig. 4 6.
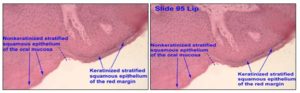
Epithelium: It is a protective layer for the tissues beneath it, it is divided into two parts; keratinized and non-keratinized surface. The non-keratinized epithelium is found in the soft palate, sublingual region, and the buccal region, which is our main concern. It contains small amounts of ceramide with noacyl-ceramides. It also contains small amounts of neutral but polar lipids, mainly cholesterol sulfate and glucosylceramides. These epithelia are found to be considerably more permeable to water than keratinized epithelia. Keratinized epithelium is found in the hard palate and non-flexible regions of the oral cavity that is impermeable to water as it is associated with barrier. The epithelium of the buccal mucosa is about 40 to 50 cells layer thick, epithelial cells increase in size and become flattered as they travel from the basal layers to the superficial layer. The epithelia are rough in texture with surface area 52.2 cm2. The turnover time of the buccal epithelium is estimated at 5-6 days. The thickness of mucosa in humans, dogs, and rabbits has been determined approximately 500-800 µm, as shown in Fig. 5 6, 2.
FIG. 5: KERATINIZED AND NON-KERATINIZED EPITHELIA 6
Basement Membrane: It provides adherence between the underlining connective tissues, and the epithelium acts as mechanical support for the epithelium.
Lamina Propria: It consists of collagen fibers, a supporting layer of connective tissues, smooth muscles, blood vessels, and capillaries that open to the internal jugular vein. The rich arterial blood supply to the mucosa membrane is derived from the external carotid artery 7, 3.
Submucosa: A gel-like secretion known as mucus covers the entire oral cavity that acts as a protective layer to the cells below and it consists of insoluble glycoproteins, water, small quantities of proteins, enzymes, electrolytes and nucleic acid 4.
The environment of the Buccal Cavity: The cells of the oral epithelia are surrounded by mucus; an intercellular ground substance, which is the principle component of which complexes are made of proteins and carbohydrates. This matrix plays a role in cell-cell adhesion as well as a lubricant; the mucus also plays a role in bio-adhesion of mucoadhesive drug delivery system. At physiological pH, the mucus network carries a negative charge so, at this pH, mucus can form a strong, cohesive gel structure that will bind to the epithelial cell surface as gelatinous layer 8.On the other hand, saliva; is an aqueous fluid with 1% organic and inorganic materials, which protects soft tissues from abrasion by rough materials and chemicals. The salivary pH ranges from 5.5 to 7. Depending on the flow rate, the daily salivary volume is between 0.5 to 2 liters, and it’s the volume of fluid that is available to hydrate oral mucosal dosage forms. The selection of hydrophilic polymeric devices as a vehicle for buccal drug delivery is due to this water-rich environment of the oral cavity 8.
Permeability of the Buccal Mucosa: Permeability of the oral mucosa is relatively low, but it differs among various parts of the oral region, taking into consideration that the buccal membrane is more permeable than other parts in the oral region. The buccal mucosa forms a barrier to drug permeation, the effects of this barrier and buccal absorption are the factors affecting the drug administration. It is estimated that the buccal mucosa permeability is 4-4000 times greater than the skin. The order of permeability of the oral cavity is decreasing in order of sublingual then the buccal, and then the palate. This order depends on the relative thickness and the degree of keratinization 6. The permeability coefficient of a drug is used to measure the ease of which the drug can permeate the membrane. The permeability coefficient is the function of the degree of keratinization of these tissues, physiochemical properties of the drug, and the membrane thickness. It is believed that the permeability barrier in the oral mucosa is a result of intercellular material derived from the so-called membrane coating granules (MCG) which are either keratinized and non-keratinized 8, 4.
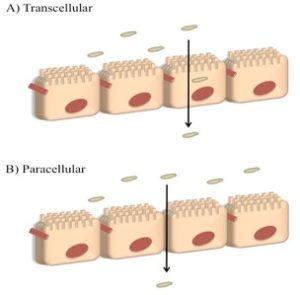
Buccal Absorption Pathway: There are two permeation pathways for passive drug transport across the oral mucosa: para-cellular and trans-cellular routes. Permeants can use these two routes simultaneously, but one route is usually preferred over the other depending on the physiochemical properties of the diffusing drug. Since; the intercellular spaces and cytoplasm are hydrophilic so that lipophilic compounds would have low solubility in this environment. The cell membrane, however, is lipophilic, so hydrophilic solutes will have difficulty permeating through the cell membrane due to the partition coefficient.
FIG. 6: PASSIVE DIFFUSION PATHWAY 4
Therefore, the intercellular space pose as the major barrier to permeation of lipophilic compound, and the cell membrane acts as the major transport barrier for hydrophilic compounds. In the oral epithelium, the permeation of the solute may involve a combination of these routes 6. There are different modes of permeation of the buccal cavity, which are passive diffusion and endocytosis.
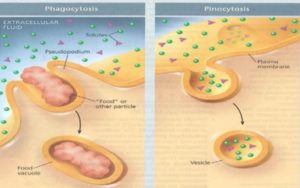
Firstly, passive diffusion involves carriers, channels, or direct diffusion through a membrane. This type of transport always operates from regions of greater concentration to regions of lesser concentration. No external source of energy is required; passive diffusion is either trans-cellular or intracellular, which is crossing the cell membrane and entering the cell, so lipophilic molecules can permeate or paracellular or intercellular, which is bypassing between the cells, so hydrophilic molecules can permeate; this is shown in Fig. 6. Secondly, for the endocytosis, which is a process where the drug molecules are engulfed by the cells. It is of two types; phagocytosis, which indicates the engulfment of solid drug molecules and pinocytosis that indicates the engulfment of liquid drug molecules, as shown in Fig. 7 10.
FIG. 7: ENDOCYTOSIS DIFFUSION 10
Barriers to Penetrate across the Buccal Mucosa: Barriers such as membrane coating granules, mucus, saliva, and basement membrane affect the rate and extent of the drug absorption through the buccal mucosa.
Membrane Coating Granules (Cores Granules): The main permeability barrier property of buccal mucosa is due to intercellular materials derived from the so-called membrane coating granules (MCG). They are spherical granules that are 100-300 nm in diameter that is found in both non-keratinized and keratinized epithelia. The main function of MCG is cell adhesion, membrane thickening effect, cell surface coat production, and permeability barrier 11.
Mucus: Mucus is composed of mucins and inorganic salts suspended in water. It acts as a lubricant which allows cells to move relative to one another. Mucus plays an important role in the adhesion of bio-adhesive drug delivery system 7.
Saliva: It initiates the digestion process, moistens the mouth, and controls the bacterial flora of the oral cavity. Constant flowing down of saliva in the oral cavity makes it very difficult for drugs to be administered for a significant amount of time to facilitate absorption in this site 11.
Basement Membrane: The basement membrane plays an important role in limiting the passage of materials across the junction between epithelium and connective tissues. The charge on the components of the basal lamina will limit the rate of penetration of the lipophilic compounds that can pass the superficial epithelial barrier relatively easy 7.
Physiochemical Properties of the Drug Affect Absorption of Drug: There are some variables that can affect drug absorption, including physico-chemical properties of the drug as 2, 3:
Molecular Size: For the hydrophilic substance, the rate of absorption is dependent upon the molecular size. Small molecules (<75-100 Daltons (Da)) cross the mucosa rapidly. However, this permeability decreases as the molecular size increases.
Lipid Solubility: The permeability is dependent upon the oil-water partition coefficient, so more lipid compounds have higher permeability.
Ionization: The degree of ionization depends on the function of both pH and pKa at the mucosal surface. For weak acids and bases, only the unionized forms are lipid soluble. As ionization increase, maximum absorption of many compounds is achieved.
Physiological Factors Affecting the Buccal Bioavailability: Other variables that can affect drug absorption are the physiological factors of the buccal cavity as:
Inherent Permeability of the Epithelium: The oral mucosal epithelium permeability is intermediate between that of the skin, which is specialized with its barrier function, and the gut epithelium, which is specialized with high absorption function.
The thickness of Epithelium: It differs among the sites in the oral cavity. The buccal mucosa is approximately 500-800 µm thick.
Blood Supply: Rich blood supply and lymphatic drainage are present in the lamina propria, the blood flow in the buccal mucosa is 2.4 ml min-1 cm, which serve the oral cavity, so the drug moieties that passes the epithelium will be absorbed into the systemic circulation.
Mucosa and Saliva: The oral mucosal surfaces are constantly washed by saliva stream with an approximation of 0.5-2L/day. Since the buccal area is exposed to a lot of salivae, this will enhance the drug dissolution increasing its bioavailability.
Metabolic Activity: Drug moieties absorbed through the oral epithelium are directly delivered to the blood circulation; therefore, avoiding the first pass effect caused by the liver and gut walls. So, oral mucosal drug delivery is an attractive way of delivering liable enzymatic drugs as therapeutic proteins and peptides.
Ability to Retain Delivery System: The buccal mucosa provides a smooth and immobile surface that is ideally suited to the use of the retentive delivery system.
Species Differences: Rodents’ buccal mucosa contains a highly keratinized epithelium, so they are not very suitable as animal models when studying the buccal cavity.
The Oral Fast Dissolving Film: Orally Fast Dissolving Film, as shown in Fig. 8 is a new drug delivery system for the oral delivery of the drugs. It was developed based on technology of the transdermal patch. The delivery system consists of a very thin oral strip, which is simply placed on the patient’s tongue or any oral mucosal tissue, instantly wet by saliva, where the film rapidly hydrates and adheres onto the site of application. It then rapidly disintegrates and dissolves to release the medication 11. Pharmaceutical industry technology is growing year by year. Researchers and scientists led to that growth in two different ways, discovering new molecules to treat a specific disease or enhancing the administration of well-known molecules by enhancing their dosage forms and route of administration. Both approaches got the same aim, which is achieving the goal of treatment.
FIG. 8: OFDFs 12
The technology of manufacturing of the exciting molecule is an advanced way to improve and discard its possible drawbacks which either due to the drug harmful side effects on its site of action or the response of the body different systems and organs on the drug, affecting its bioavailability and metabolism. Another factor that must be considered is the patient himself; they also may have a direct effect on the treatment therapy that could happen in many ways like; not following the right procedure in taking certain drugs or ignoring the required dose and concentration.
Also, patients may have a direct effect results from their state of health like consciousness, some disease affecting the drug absorption, and if the patient is susceptible to injection or not. Many more reasons could also result in not achieving the goal of treatment; those reasons led to the research centers and the pharmaceutical industries to pay more attention and focus on the developing of the dosage forms and even discovering new routes of administration. OFDFs are one of the dosage forms that are gaining a lot of interest in the pharmaceutical industry 12.
Benefits of Oral Fast Dissolving Films: Due to the presence of larger surface area, films provide rapid disintegration and dissolution in the oral cavity. OFDFs are flexible and portable, so they provide ease in transportation, during consumer handling and storage. It is suitable for dysphagia patients, patients who are mentally ill, patients who are un-cooperative or are on reduced liquid intake plans or nauseated. The film is beneficial in emergency cases such as motion sickness, acute pain, and suede episodes of allergic attack or coughing, where an ultra-rapid onset of action is required. Stable for a long duration, since the drug remains in solid dosage form until it is consumed 14. So, it combines the advantage of the solid dosage form in terms of stability and liquid dosage form in terms of bioavailability 12. The sublingual and buccal delivery of a drug via thin film has the potential to improve the onset of action, lowering the dosing, and enhance the efficacy and safety profile of the medicament, provide a new business opportunity like product differentiation, product promotion 12.
Limitations of Oral Fast Dissolving Films: The OFDFs have some drawbacks concerning the dose uniformity for each strip to another strip as it is a challenge to keep the same dose for all strips. Another drawback is the drug loading capacity, as not all drugs can be loaded on the OFDFs because there are some requirements for the drug to be loaded on the strip; one of them is the drug concentration/dose 14.
Since the maximum dose to be loaded is 75 mg; therefore, not all drugs can be loaded on the film. Moreover, there are some drawbacks concerning the packaging of the strips which needs some special requirements as it is hard to be packed and at the same time the pack must not interact with the film, also make sure that the film is stable inside it. Finally, the last drawback for the OFDFs is that there is several technical problems with the use of the film strips as an example; its thickness while casting the film 14.
Film Forming Matrix: The matrix of OFDFs has special considerations to make a suitable thin film to disintegrate within the buccal cavity. They must be safe, non-toxic, and non-irritant to be used orally. Moreover, the film components must serve in achieving the important characters of the film, like water solubility and stability. There are several ingredients used to compose a film. The most important components are polymers and plasticizers. The other formula components are used according to the desired function or type of the film example saliva stimulating agent, sweetening agent, surfactants, coloring agents, flavoring agents. All the ingredients used in the film are added with certain concentration, as shown in Table 2 14.
TABLE 1: GENERAL COMPOSITION OF OFDFs 14
| Ingredients | Concentration (%) |
| Active pharmaceutical ingredient | 1-25 |
| Polymer | 40-50 |
| Plasticizer | 0-20 |
| Colors, flavors, fillers | 0-40 |
Film Formulating Polymers: The type of polymers is chosen according to the function required in the dosage form. Concerning all those critical parameters, the polymers can change in the film stability. A variety of polymers are available for the preparation of OFDFs. The polymers can be used alone or in combination to improve hydrophilicity, flexibility, and mouthfeel and solubility characteristics of OFDFs. The stiffness of the film depends on the type of polymer and the amount of polymer in the formulation. The polymer employed should be non-toxic, non-irritant with good wetting and spreadability property.
The polymer should not be very expensive and should be readily available. Water-soluble polymer that may be used include natural gums such as xanthan, guar, acacia, tragacanth gums; other available polymers include cellulose or cellulose derivatives, hydroxyl propyl methyl cellulose (HPMC) with different grades like HPMC E15, HPMC E5, HPMC K4M, HPMC K100, hydroxyethylcellulose, hydroxypropyl cellulose, carboxymethyl cellulose, polyvinylpyrrolidone, polyvinyl alcohol, pullulan, gelatin. Modified starches are also used for preparation. There are many types of polymers with different physical properties shown in Table 3 6.
TABLE 3: POLYMERS LIST 6
| Polymer | Special characteristics |
| Pullulan | It’s a natural glucan. It contains a unique linkage alpha 1-4 and 1-6, which is important for the bio adhesive properties. When dissolved in water; 10-15% gives a tasteless and odorless non-hygroscopic solution. It decomposes at 25 ºC-28 ºC. It is highly soluble in water, dilute alkali & insoluble in alcohol, organic solvents except in formamide and dimethylsulphoxide. It is a water solubility enhancer |
| Gelatin | The higher M.W, the better quality of the film and it is an excellent carrier for flavors, smooth mouthfeel |
| Sodium alginate | It has a unique colloidal properties, and it is appropriated for loading additives and antimicrobials |
| Rosin | Hydrophobic biomaterial, glassy solid so brittle soln. add dibutyl sebacate (DBS) plasticizer à decreased tensile strength, lowered Tg, increased elongation, and flexibility of the film. It's melting point ranges from 100 ºC to 120 ºC. It is soluble in alcohol, ether, benzene, and chloroform |
| Starch | Starch films are transparent, flavorless, tasteless, and colorless. It has limited applications due to poor mechanical strength and its efficient barrier against low polarity compounds. Cova starch had good flexibility and low water permeability. By aging, starch get loose its flexibility property |
| Maltodextrose | Hygroscopic, High DE (Dextrose Equivalent) has shorter glucose chains, higher sweetness, higher the solubility and lower heat resistance |
| Chitosan | Natural, non-toxic polymer. It has poor solubility in neutral solutions. It is generally cohesive |
| Gum carrageenan | Water soluble polymer, it is less opaque than starch films |
| Cellulose derivatives | It is flexible, transparent, odorless, flavorless, tasteless, water soluble |
| Hydroxypropyl methyl cellulose HPMC(sorbitol, PEG 200, glycerol) | It is flexible, transparent, odorless, flavorless, tasteless, water soluble. Is has a high glass transition temperature, good moisture, and oxygen polymer. But it has poor water soluble, film adhesion, mechanical strength |
| Hydroxy propyl cellulose HPC (sorbitol, PEG 200, glycerol) | It is non-ionic water-soluble thermoplastic polymer
It forms highly flexible films It has a very high glass transition temperature and good carrying capacity |
| Kollicoat | It has good lubricant properties, binding, water retention, thickening and film formation |
| Poly vinyl alcohol
(PVA) |
It is odorless, non-toxic that has melting Point 230 °C and 180-190 °C for fully hydrolyzed and partially hydrolyzed respectively. Has high tensile strength and good flexibility |
| Poly vinyl alcohol (PVP) | Soluble in water and other polar solvents, excellent wetting properties and hygroscopic |
Plasticizer: The second major component after polymers to formulate the film is the plasticizer. Plasticizers control the film mechanical characteristics. The chemical structure and concentration of plasticizers play an important role in alleviating the glass transition temperature of the polymers 13. The selection of plasticizer will depend upon its compatibility with the polymer and also the type of solvent employed in the casting of the film.
The flow of polymer will get better with the use of plasticizer and enhances the strength of the polymer. Glycerol, propylene glycol, low molecular weight polyethylene glycols, phthalate derivatives like dimethyl, diethyl, and dibutyl phthalate, citrate derivatives such as tributyl, triethyl, acetyl citrate, triacetin and castor oil are some of the commonly used plasticizer excipients. However, inappropriate use of plasticizer may lead to film cracking, splitting, and peeling of the strip. It is also reported that the use of certain plasticizers may also affect the absorption rate of the drug 6.
Sweetening Agent: The sweetening agent is a vital ingredient to achieve patient convenience and to mask any bad taste from API 15. The sweetening agents are classified into natural and artificial sweeteners 13. The natural sweeteners such as glucose, sucrose, liquid glucose, isomaltose, and fructose.
Fructose sweetener perceives rapidly in the mouth; it can be combined with sorbitol or mannitol for better mouthfeel and cooling sensation 13. Artificial sweeteners have two generations; the first one includes aspartame and climate. The second generation includes acesulfame-k, sucralose neotame, and ultimate. The sweetening agent must be considerably chosen to achieve the sweetness required 14 within the range of concentration, which is between 3-6% w/w of the whole amount of ingredients added in the film 15.
Saliva Stimulating Agent: The purpose of using saliva stimulating agents is to increase the rate of production of saliva that would aid in the faster disintegration of the rapid dissolving strip formulations. Generally, acids which are used in the preparation of food can be utilized as salivary stimulants, like citric acid, malic acid, lactic acid, ascorbic acid, and tartaric acid. These agents are used alone or in combination between 2 to 6% w/w of the weight of the strip 12.
Surfactants: Surfactants are used as a solubilizing or wetting or dispersing agent so that the film is getting dissolved within seconds and release active agent immediately. Some of the commonly used are sodium lauryl sulfate, benzalkonium chloride, benzethonium chloride, tweens etc.
Flavoring Agents: Preferably up to 10% w/w flavors are added in the OFDF formulations The acceptance of the oral disintegrating or dissolving formulation by an individual is largely depends on the initial flavour quality which is observed in first few seconds after the product has been consumed and the after taste of the formulation which lasts for at least about 10 min. The selection of flavor is dependent on the type of drug to be incorporated in the formulation. It was observed that age plays a significant role in the taste fondness.
The geriatric population like mint or orange flavors while the younger generation like flavors like fruit punch, raspberry, etc. Flavoring agents can be selected from synthetic flavor oils, oleoresins, extract derived from various parts of the plants like leaves, fruits, and flowers. Flavors can be used alone or in the combination. Peppermint oil, cinnamon oil, spearmint oil, oil of nutmeg are examples of flavor oils while vanilla, cocoa, coffee, chocolate and citrus, apple, raspberry, cherry, pineapple are few examples of fruity flavors 13.
Methods of Preparation of OFDFs: The manufacture of OFDF is done by various methods such as solvent casting, hot-melt extrusion, semisolid casting, solid-dispersion extrusion, and rolling. Here we discuss these methods and the various parameters in which dissolving films are evaluated.
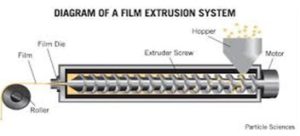
Hot Melt Extrusion: In hot melt extrusion method, as shown in Fig. 9, first we must have initial mass dried and obtained with carriers as the drug is mixed with carriers and obtained as solid mass, then the mass is introduced into an extruder which is divided into four zones having different degrees in temperature, zone 1 at 800 °C, zone 2 at 150 °C, zone 3 at 1000 °C, zone 4 at 650 °C. The speed of extruder speed should be set at 15 rpm to process the granules inside the barrel of an extruder for 3-4 min 16, 13. The film is obtained after being pressed into a cylindrical calendar. Hot melt extrusion provides a lot of advantages 16, 13 like limited operation unite. It minimizes the amount of waste product, doesn’t need the use of solvent or water (anhydrous) and produces uniform content because of intense mixing and agitation.
FIG. 9: HOT MELT EXTRUSION EQUIPMENT 16
Semi-Solid Casting Methods: In solvent casting method excipients are dissolved in water, then water-soluble polymers and the drug is added and stirred to form a homogeneous solution. Finally, the solution is cast into the Petri plate and dried in the semi-solid casting method the ratio used is between the acid, insoluble and film-forming polymers should be from 1:4.
The Steps of the Method are: 14
Solid Dispersion Extraction: The aim here is to disperse the drug into a melted polymer solution to facilitate its loading. The number of active ingredients used can be one or more that are dissolved in a suitable liquid solvent which acts as an inert carrier. This happens in the presence of amorphous hydrophilic polymer under 70 °C temperature without removing the liquid solvent to obtain the solid dispersion needed. Finally, this obtained solid dispersions are shaped into films using dyes. Knowing that the selected solvent or the dissolved drug may be immiscible with the melt of the polymeric form 16, 14.
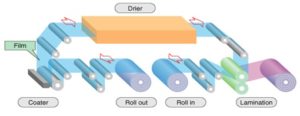
Rolling Method: In the rolling method, as shown in Fig. 10, the film is prepared first by premix preparation, then it is mixed with the drug solution and added to the roller as shown in Fig. 16. Premix consist of film-forming polymers, polar solvents, other excipients, and additives but no drug. The whole premix is added to the master batch feed tank, then feed it by a 1st metering pump and control value to one or both of the 1st and 2nd mixers 17. The required amount of drug is then added to the desired mix, blended with the master batch premix to give a uniform matrix. The specific amount of uniform matrix is then fed to the pan by second metering pumps, and the film is finally formed and carried away by support roller and the wet film is then dried using controlled bottom drying 14, 15, 16.
FIG. 10: ROLLING METHOD EQUIPMENT15
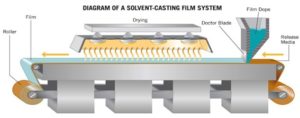
Solvent Casting Method: Solvent casting method, as shown in Fig. 11, is preferred in fast dissolving films. Two separate solvents are done with their soluble ingredients; then they are mixed and stirred to form a homogenous solution.
FIG. 11: SOLVENT CASTING EQUIPMENT 13
At first, water-soluble polymers are dissolved in water and form a clear, viscous solution. Secondly, the drug and its other excipients are dissolved in a suitable solvent 13. Then both solutions are mixed and stirred at 1000 rpm. Finally, the homogenous solution obtained undergoes vacuum removing of entrapped air, cast and dried into the Petri plate and then cut into pieces of desired size 12, 14, 16.
Evaluation and Quality Control Tests of OFDFs: Oral Fast Dissolving Films can be evaluated through many tests. The quality control of these types of dosage form requires specific tests which determine the quality and stability of the oral fast dissolving films as it includes the shape, thickness, endurance, hygroscope characteristics of the film, strength, percent elongation, weight variation, drug content, disintegration time, in-vitro dissolution test, dryness test, tears resistance, stability studies and the organoleptic evaluation of the film, surface pH of the film. Moreover, there are some evaluation tests, but it is not obligatory to be done which are the young modulus, contact angle, transparency, swelling property of the film, morphology studies, film softening, permeation studies, percent moisture loss, storage and packing of the OFDF films 12.
Thickness Test: In the previous studies of the evaluation of OFDFs films, the thickness test was done with simple techniques and methods. Later on, the development of the thickness has been increased. The film thickness is measured by two materials or equipment either by digital Vernier calipers, as shown in Fig. 12, or the screw gauge micrometer, as shown in Fig. 13. The film thickness should be tested and evaluated from five different locations, especially the four corners and at the center of the film. The thickness range of the OFDFs should be between the ranges of 5-200 micrometer. This test aims to ensure the uniformity of the drug content and accuracy of the dose 16.
Dryness Test: Dryness test or the tackiness test. The aim of this test it to evaluate the tenacity of the film being able to grip the solvent and also to see the adherence of the film as it is done with eight stages known for film drying and these are set to touch, free from dust, surface dry, dry to react, dry handling, dry to touch, dry hard and free from fingerprints or dry print free 16. Tack is defined as the persistence in which the strip stick to a piece of paper which is pressed into contact with the strip.
Tensile Strength: Tensile strength; this test aims to evaluate and to measure the maximum force applied to a point at which the film breaks. This test can be calculated by the applied load at the cut-off or the fracture divided by the cross-sectional strip. The equation below shows the calculation of how to calculate the tensile strength 12.
Tensile strength = Load at breakage/strip thickness × strip width
Folding Endurance: Folding endurance in which the film is rolled or folded for many times until it breaks; this will determine the endurance of the film. As the times of folding increase, the endurance of film increases. It is a directly proportional relation 12.
Tears Resistance: Tear resistance in which the film is presented to high shear force to evaluate the tear resistance of the film 14. Usually, the applied load is low or minimal rate 51mm/min. It is defined as the newton pound-force. It is generally found near the start of tearing which can tear the film 16. It is done by elementary tear thwing -albret tearing test as shown in Fig. 14.
FIG. 14: ELMENDORF TEAR THWINGALBRET 16
Organoleptic Evaluation: Organoleptic evaluation studies. In this test, the evaluation is carried out to see some properties of the film. Special apparatus is used to evaluate the taste of the strip by special controlled human taste panels 14. The organoleptic properties needed for the OFDFs includes the color, flavor, and taste. This formulation will dissolve and dissolute in the buccal cavity, so it is very important to take in considerations for these organoleptic properties to increase patient compliance. As for depressed people and children, the colored film will be a good choice for administration. The second organoleptic property is the odor of the film. The flavor which is loaded on the film should provide an acceptable odor to the formulation. An ideal flavoring agent should mask the odor of the excipients 16.
Surface pH: The principle of this test is to determine the pH of the film. Since the OFDF films are administered through the buccal cavity, which may cause side effects; therefore pH must be adjusted. The pH of the OFDFs should be close to neutral pH. The film is combined with a pH electrode; the pH is measured by bringing the electrode on the surface of the film, as shown in Fig. 15.
FIG. 15: THE SURFACE pH METER 16
The minimum requirements for this test are at least six oral films strips, and the mean, standard deviation is calculated 16.
Drug Content Determination: This test aims to measure the drug content inside the film or to evaluate the drug uniformity inside the film . This is measured by any standard assay method described for specific API in any standard pharmacopeia . The content uniformity is measured by estimating the API content or value. The limit of content uniformity is 85-115 percent 16, 14.
In-vitro Dissolution: Dissolution is a term which defines the amount of the drug that goes into solution per unit time under special or certain conditions of solid liquid interface, temperature and the solvent concentration 16. The apparatus used in this test is the simple paddle with sinker dissolution apparatus or the standard basket or paddle apparatus, which is described in any pharmacopeia, as shown in Fig. 16. As for the OFDFs film, it is better for us the paddle with sinker apparatus because the film might float on the surface of the aqueous solution. The dissolution medium will be selected as per the sink conditions and the highest dose of the active pharmaceutical ingredient 12.
FIG. 16: SIMPLE PADDLE WITH SINKER DISSOLUTION APPARATUS 12
Young Modulus: It is known as the elastic modulus. It is the measure of the stiffness of the film strip it is represented as the ratio of the stress applied overstrain in the area of elastic deformation. It is calculated by the following equation:
Young modulus = Force of corresponding strain/ cross-sectional area 15
Contact Angle: This test aims to evaluate the wetting properties, dissolution, and disintegration time of the OFDFs 16, 13. The contact angle is measured by goniometer (AB Lorentz and wetter Germany) apparatus at room temperature, as shown in Fig. 17. The procedure of this test requires a dry film to see the wetting properties of the film. Take the dry film and put a drop of distilled water on its surface. The images of the droplets will be recorded in 10 sec of deposition by a digital camera. Then the contact angle of the film is measured in both sides, and the average angle is calculated 15.
FIG. 17: GONIOMETER (AB LORENTZ AND WETTER GERMANY) APPARATUS 15
Swelling Property: This test aims to see the swelling amount of the film. This is done by using a simulated solution of saliva, which is used to conduct the swelling property study. The samples are weighed and placed on a stainless steel dish 15 ml were added by using a saliva solution. The film is placed inside the saliva solution, and the increase in weight of the film was observed until a constant weight is observed 14.
Transparency Test: The test is done by placing the film specimen inside the internal side of the ultraviolet spectrophotometer cell. The transparency is measured by the equation below.
Transparency = (LogT600)/b= - ɛC
In which T600 is the transmittance at 600 nm, and b is the thickness of the film in millimeters and C is the concentration 14, as shown in Fig. 18.
Percent Elongation: The percent elongation test in which the stress is applied to a film (2 × 2 cm2). This is known as strain. The strain is defined as the deformation of the film strip before it gets broken because of the stress applied. This is measured by a machine called Hounsfield universal testing machine 12. As the plasticizer content in the film increases the percent elongation. It is calculated as in the equation below.
Percent elongation = Increase in length of strip x 100 / Initial length of the strip
In-vitro Disintegration Test: This type of test requires the USP disintegration apparatus. The same test that is done to the ODTs is done to the OFDFs films. Although there are no official guidelines to the OFDFs, the disintegration will differ depending on the formulation as it will range from 5-30 sec to disintegrate 12, as shown in Fig. 19.
Characteristics of the Drug to be loaded on the OFDFs Strip: There are some requirements concerning the drug to be loaded on the film. Since not all drugs are ready to be loaded.The drug must have Pleasant taste and odor, low dose up to 75 mg, smaller or moderate molecular weight, good stability, solubility in the saliva inside the buccal cavity, it should be partially unionized at the pH of the oral cavity, and it should have the ability to permeate oral mucosal tissue. The OFDFs consists of a very thin oral strip which is simply placed on the patient tongue or any mucosal tissue, to be wetted by saliva, gets hydrated and adheres onto the site of the application than directly into the systemic circulation 15.
Drugs Approved by FDA: Over the past few years, much research and development have been directed to formulating ODF products with prescription drugs. As a result, the year 2010 was quite significant for the ODF sector of the drug delivery industry; it is the year that the first prescription ODF was approved in the European Union to the United States.
However, in the past two years, only two new prescription ODFs have made it to market. The following Table 4 provides an overview of approved prescription ODF products available in Japan, the European Union to the United States.
TABLE 4: DRUGS APPROVED BY FDA15
| OTF
developer |
Distributor/
Marketer |
Product
name |
Drug | API
content |
Country / Region | Year |
| Kyu Kyu | Mochida | Voglibose OD film 0.2 | Voglibose | 0.2 mg | Japan | 2006 launch |
| Kyu Kyu | Mochida | Voglibose OD film 0.3 | Voglibose | 0.3 mg | Japan | 2006 launch |
| Kyu Kyu | Teva-Kowa Pharma | Amlodipine OD film 2.5 | Amlodipine | 2.5 mg | Japan | January 2010 launch |
| Kyu Kyu | Teva-Kowa Pharma | Amlodipine OD film 5 | Amlodipine | 5 mg | Japan | January 2010 launch |
| Kyu Kyu | Elmed Eisai | Donepezil HCl OD film 3 | Donepezil | 3 mg | Japan | November 2011 launch |
| Kyu Kyu | Elmed Eisai | Donepezil HCl OD film 5 | Donepezil | 5 mg | Japan | November 2011 launch |
| Kyu Kyu | Mochida | Loratadine OD film 10 | Loratadine | 10 mg | Japan | November 2011 launch |
| APR/Labtec/
MonoSol Rx |
Bio Alliance
Pharma SA |
Setofilm
|
Ondansetron | 0.4 mg,
0.8 mg |
EU | March 2012
Approval |
| APR/Labtec/MonoSol Rx | Still being
sought |
Zolmitriptan
rapid film |
Zolmitriptan | 2.5 mg,
5 mg |
EU | March 2012
Approval |
OFDFs in Chemotherapeutic Induced Nausea and Vomiting: One major advantage for OFDFs in chemotherapeutic-induced nausea and vomiting (CINV) this is the most terrified-related side effect for cancer patients receiving chemotherapy who is presented to nausea and vomiting while taking this regimen treatment especially for children who cannot tolerate the pain of the intravenous anti-emetic agent, they are also presented to vomiting while taking the medication of anti-emetic Ondansetron in IV form due to irritation and GIT upset resulting from chemotherapy side effects. The optimum choice for administering the drug, in this case, is the OFDFs loaded with the drug Ondansetron as it is the drug of choice for CINV patients.
The OFDFs films overcome the intravenous dosage form drawbacks in all terms except bioavailability as its bioavailability is near to unity. The film strip is pain-free, taken without water in these cases, no irritation, and it is more convenient for the patient to administer. Moreover, the OFDFs has a convenient dosing, taste masking, site-specific, local action, and large surface area that enhance the disintegration and the dissolution time in the oral cavity. Also, the buccal delivery of a drug via thin film improves the onset of action, lower dosing, enhance the efficacy of the drug and high safety profile 15.
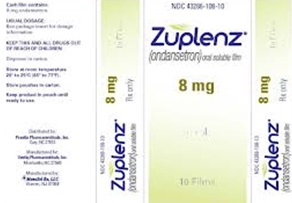
Zuplenz:
Generic name: Ondansetron
Indication: Nausea and vomiting
Desired use: Chemotherapeutic induced nausea and vomiting.
Strength: 8 mg.
FIG. 20: ZUPLENZ
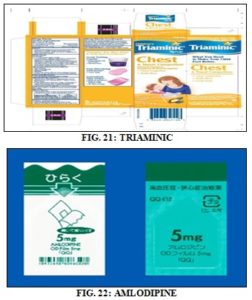
Triaminic:
Generic name: Dextromethorphan + phenylephrine.
Indication: Cough suppressant and nasal decongestant.
Strength: 5mg + 2.5mg
Amlodipine:
Indication: Anti-hypertensive drug
Class: calcium channel blocker
Strength: 5 mg
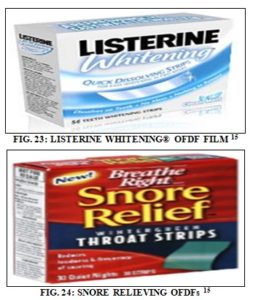
Applications of OFDFs: The OFDF doesn’t serve the pharmaceutical field only; it has other applications in terms of cosmetics, sweeteners, nicotine replacement therapy, and energy replenishment. Non-pharmaceutical oral thin films besides breath freshening applications, one logical expansion of the edible thin film industry were to develop oral care products.
As a result, several Consumer Healthcare companies have now commercialized ODF products for varying uses that include teeth whitening and the prevention of snoring 15. The first fully dissolvable whitening strips, as shown in Fig. 23, were commercialized in 2007 by McNeil (Johnson and Johnson) as Listerine Whitening® Quick Dissolve Strips. Another oral care application offshoot for ODF technology has been developed to reduce snoring during sleep, as shown in Fig. 24.
BioFilm created Snoreeze oral strips that were commercialized in 2006 via the company Passion for Life Healthcare. Each strip contains guar gum and micro-encapsulated peppermint oil to reduce snoring. The ODF is placed on the roof of the mouth and allowed to dissolve for 20-30 sec. This allows the ingredients to coat the back of the throat, and snoring is theoretically reduced.
Food industry and sensation enhancement applications one of the earlier uses for edible film technology was to develop products for the food industry. This use of edible films is becoming a popular trend. The films can be used to add flavors, maintain freshness, improve appearance, and even help prevent fungal and bacterial growth. Ascona Ingredients Ltd. is a Canadian corporation that develops and manufactures edible films for confectionary, nutraceutical, and pharmaceutical applications. An edible film used to cover meat products and made from water-soluble polysaccharides.
FIG. 25: FLAVORED ODF 15
In June 2010, American Greetings Corporation, working with First Flavor Inc., introduced a new greeting card under the name Tasties™. Each card in the new line features a flavored dissolvable ODF strip, individually pouched and sealed inside, as shown in Fig. 25. This allows the recipient to physically taste relevant flavors, which adds an extra dimension to the greeting card ritual 15. Tsukioka Film Pharma Co. is a relative newcomer to the thin film industry. The company started in hot stamping, then progressed to edible gold leaf, edible films for food and cosmetics, and most recently into pharmaceutical films. Tsukioka offers fast dissolve strips with a variety of nutraceutical ingredients as well as fresh breath strips in various flavors such as mint, orange, and lemon, as shown in Fig. 26. The company is working with Gifu International Institute of Biology to create a sustained release pharmaceutical film and has invested in a state-of-the-art plant dedicated to pharmaceutical film manufacturing in Techno Plaza in Kakamigahara-City, Japan. Tsukioka plans to obtain FDA and KFDA c-GMP certificates and expand their OEM manufacturing of pharmaceutical films in anticipation of strong business with local and global pharmaceutical companies 15.
FIG. 26: TUSKIOKA FLAVORED ORAL FAST DISSOLVING FILM 15
Permeation Studies: Permeation studies are carried using the modified Franz diffusion cell by using porcine buccal mucosa. The mucosa is mounted between the donor and receptor compartment of Franz diffusion cell. The receptor compartment is filled with buffer and maintained at 37 °C ± 0.2 °C, and the hydrodynamics were maintained by stirring with a magnetic bead at 50 rpm. One previously weighed film is placed in intimate contact with the mucosal surface of the membrane that should be previously moistened with a few drops of simulated saliva. The donor compartment is filled with 1 ml of simulated saliva of pH 6.8. Samples are withdrawn at a suitable interval, replacing the same amount with the fresh medium. The percentage of drug permeated is determined by measuring the absorbance by the selected analytical method.
Stability Study: Stability study of fast dissolving films is carried out for all the batches according to ICH guidelines. After predetermined time intervals, the films are evaluated for the drug content, disintegration time, and physical appearance 19.
Palatability Test: Palatability study is conducted based on taste, after bitterness and physical appearance. All the batches are rated A, B, and C grades as per the criteria. When the formulation scores at least one A grade, the formulation is considered as average. When the formulation scores two A grade, then it would be considered as good and the one with all three A grade it would be the very good formulation.
Grades: A= very good, B= good, C=poor.
Packaging of Fast Dissolving Film: In the pharmaceutical industry, it is vital that the package selected adequately preserve the integrity of the product. Expensive packaging, specific processing, and special care are required during manufacturing and storage to protect the dosage of other fast dissolving dosage forms. A variety of packaging options are available for fast dissolving films. Single packaging is mandatory for films, which are pharmaceutical products; an aluminum pouch is the most commonly used packaging format. APR- Labtec has developed the Rapid card, a proprietary and patented packaging system, which is specially designed for rapid films. The rapid card has the same size as a credit card and holds three raid films on each side. Every dose can be taken out individually.
The material selected must have the following characteristics:
- They must protect the preparation from environmental conditions.
- They must be FDA approved.
- They must meet the applicable tamper-resistant requirement.
- They must be non-toxic.
- They must not be reactive with the product.
- They must not impart to the product tastes or odors.
CONCLUSION: Among the different routes of drug delivery, the oral route is nominated to be the most favored and highly acceptable to patients and physicians. The buccal region is one of the most convenient and easily approachable routes of administration for the delivery of the therapeutic agents to be used locally and systemically. The mucosa is considered one of the potential sites of drug administration. There are some variables that can affect drug absorption, including physicochemical properties of the drug as molecular size; for the hydrophilic substance, the rate of absorption is dependent upon the molecular size. Small molecules (< 75-100 Da) cross the mucosa rapidly.
However, this permeability decreases as the molecular size increases. Also, lipid solubility; where the permeability is dependent upon the oil-water partition coefficient, so more lipid compounds have higher permeability. Ionization can affect the absorption of the drug as the degree of ionization depends on the function of both pH and pKa at the mucosal surface. For weak acids and bases, only the unionized forms are lipid soluble, and as ionization increase, maximum absorption of many compounds is achieved orally fast dissolving film is a new drug delivery system for the oral delivery of the drugs. It was developed based on technology of the transdermal patch.
The delivery system consists of a very thin oral strip, which is simply placed on the patient’s tongue or any oral mucosal tissue, instantly wet by saliva, where the film rapidly hydrates and adheres onto the site of application. It then rapidly disintegrates and dissolves to release the medication.
ACKNOWLEDGEMENT: Author is very much thankful to my dear student Ahmed Mohamed Shetwey, who helped me in this work.
CONFLICT OF INTEREST: The author declares that there is no conflict of interests regarding the publication of this article.
REFERENCES:
- Santosh Kumar R: An update on fast dissolving films. Wor J of Pharm and Pharma Sci 2016; 5(10): 466-486
- Venkateswarlu K, Preethi JK and Chandrasekhar KB: Formulation and in-vitro evaluation of loperamide immediate release tablets by liquisolid technique. Adv Pharm Bull 2016; 6: 385-390.
- Thakur N, Bansal M and Sharma N: Overview “A novel approach of fast dissolving films and their patients. Advances in Biological Research 2013; 7(2): 50-58.
- Nagendrakumar D: Formulation and evaluation of fast dissolving oral films of metoprolol succinate. International J of Engineering and Applied Sciences 2015; 6(4): 28-38.
- Utkarsha, CT: An overview of: orally fast dissolving sub-lingual film. Ind J of Pharm Sci and Res 2017; 7(2): 43-49.
- Nagendrakumar D, Keshavshetti GG, Pratibha M, Swati S and Harshanand S: Formulation and evaluation of fast dissolving oral films of metoprolol succinate. Int J Eng Appl Sci 2015; 6: 28-38.
- Bobade NN, Atram SC, Wankhade VP, Pande SD, Tapar KK: A review on buccal drug delivery system. Inter J of Pharm and Pharma Sci Res 2013; 3(1): 35-40.
- Vijayabhaskar K, Venkateswarlu K, Thirumalesh Naik SB, Kiran Jyothi R and Vani NG: Preparation and in-vitro evaluation of ranitidine mucoadhesive microspheres for prolonged gastric retention. Br J Phar Res 2016; 10: 1-12.
- Nibha KP: An overview of: Sublingual route for systemic drug delivery. International Journal of Research in Pharmaceutical and Biomed Sci 2012; 3(2): 913-923.
- Patil SL, Mahaparale PR, Shivnikar MA, Tiwari SS and Pawar KV: Fast dissolving oral films: An innovative drug delivery system. International Journal of Research and Reviews in Pharmacy and Applied Science 2(3): 604-610.
- Kalyan S and Bansal M: Recent trends in the development of oral dissolving film 2012; 4(2): 725-733.
- Garsuch V and Breitkreutz J: Comparative investigations on different polymers for the preparation of fast-dissolving oral films. The Journal of Pharmacy and Pharmacology 2010; 62(4): 539-545.
- Bobade NN, Atram SC, Wankhade VP, Pande SD and Tapar KK: A review on buccal drug delivery system. International Journal of Pharmacy and Pharmaceutical Science Research 2013; 3(1): 35-40.
- Ngank M and Nazish M: Fast dissolving sublingual film. A-review. Ind J of Ovel Drug Delivery 2016; 8(2): s4.61.
- Ghodake PP, Karande KM, Osmani RA, Bhosale RR, Harkare BR and Kale BB: Mouth Dissolving Films: Innovative Vehicle for Oral Drug Delivery. International Journal of Pharma Research and Review. Grant BF, Dawson DA and Stinson FS: Laboratory Animal Science 2013; 2:41-47. 1994; 44: 338–343. [PubMed]
- Thakur S: Mouth dissolving films: A review. Int J Pharm Bio Sci 2013; 4(1): (P) 899-908.
- Nagar P, Chauhan I and Yasir M: Insights into Polymers: film formers in mouth dissolving films. Drug Invention Today 2011; 3(12): 280-289.
- Arya A, Chandra A, Sharma V and Pathak K: Fast dissolving oral films: An innovative drug. Delivery System and Dosage Form. Int J of Chem Tech Research 2010; 2(1): 576-583.
- Patel AR, Prajapati DS and Raval JA: Fast dissolving films (FDFS) as a newer venture in fast dissolving dosage forms. Int J of Drug Develop and Res 2010; 2(2): 232246.
How to cite this article:
Mostafa DA: Fast dissolving oral film: Overview. Int J Life Sci & Rev 2018; 4(5): 71-87. doi: 10.13040/IJPSR.0975-8232.IJLSR.4(5).71-87.
All © 2015 are reserved by International Journal of Life Sciences and Review. This Journal licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.