FORMULATION AND EVALUATION OF FAST DISSOLVING TABLET OF AMLODIPINE BESYLATE USING DIFFERENT SUPERDISINTEGRANTS CONTAINING SOLID DISPERSION BY DIRECT COMPRESSION METHOD
HTML Full TextFORMULATION AND EVALUATION OF FAST DISSOLVING TABLET OF AMLODIPINE BESYLATE USING DIFFERENT SUPERDISINTEGRANTS CONTAINING SOLID DISPERSION BY DIRECT COMPRESSION METHOD
Jyoti Verma * 1, S. K. Prajapati 1, Dilip Kumar Chanchal 2, Rohit Kumar Bijauliya 2 and Neha Agnihotri 1
Department of Pharmaceutics 1, Department of Pharmacognosy 2, Institute of Pharmacy, Bundelkhand University, Jhansi - 284128, Uttar Pradesh, India.
ABSTRACT: Amlodipine besylate is a major calcium channel blocker for the treatment of hypertension. Solid dispersion Amlodipine besylate (SD-AB) was prepared by using EC, HPMC, and croscarmellose in different ratios. The development of rapidly disintegrating oral tablets of Amlodipine besylate by direct compression method. The tablets were prepared by using direct compression method and evaluated for weight variations, hardness, friability, wetting time, disintegration time, and dissolution study. Although oral disintegrating tablets are much useful in the cases of the geriatric, pediatric, and traveling patient because of no water required for the administration, therefore, the solid dispersions prepared by a solvent evaporation method using EC, HPMC and croscarmellose carrier can be successfully used for the improvement of dissolution of SD-AB and resulted in a faster onset of action as indicated by in-vivo studies.
Keywords: Amlodipine besylate, Superdisintegrants, Dissolution profile, Solid dispersion, Direct compression
INTRODUCTION: Cardiovascular diseases are increasing rapidly in the developing world. Hypertension is one of the most important modifiable risk factors for cardiovascular disease; it affects approximately one billion people worldwide. Treatment of high blood pressure can reduce cardiovascular mortality and morbidity. Orodispersible tablets are those when put on tongue disintegrates instantaneously, releasing the drug, which dissolves or disperses in the saliva 1.
The faster the drug release into solution, the quicker the absorption and onset of clinical effect. The advantages of mouth dissolving dosage forms are increasingly being recognized in both industry and academics. Their growing importance was underlined recently when European Pharmacopoeia adopted the term “orodispersible tablet” as a tablet to be placed in the mouth where it disperses rapidly before swallowing.
The increasing popularity of orally disintegrating dosage forms is in part owing to various factors such as fast disintegration, good mouthfeel, easy to handle, easy to swallow, and effective taste. Amlodipine is slowly and almost completely absorbed from the gastrointestinal tract. Peak plasma concentrations are reached at 6-9 h of post dose 2, 3.
Amlodipine is extensively metabolized in the liver, but there is no significant pre-systemic or first-pass metabolism and is slowly cleared with a terminal elimination half-life of 40-60 h. The volume of distribution is large (21 l/kg), and there is a high degree of protein binding (98%) 4, 5, 6. Because of less extensive and less variable first pass metabolism, its oral bioavailability is higher (60% - 80%), and more consistent. Thus, an oral dosage form is preferable. Amlodipine besylate can be administered once daily as 10 mg orally in the treatment of hypertension. Geriatric patients may have difficulty in swallowing and chewing the tablets resulting in patient noncompliance and ineffective therapy. To overcome these problems, oral dissolving tablets are a good option. Since they disintegrate and dissolve rapidly in saliva without need for drinking water. The development of a fast dissolving tablet also provides an opportunity for a line extension in the market place7.
MATERIALS AND METHODS:
Materials: Amlodipine besylate was a gift sample from Cadila Pharm. Sodium starch glycolate (SSG), aspartame; crospovidone (CP) and ethyl cellulose (EC) were obtained from Yarrow Chem. products. Croscarmellose (CCS), talc, lactose, microcrystalline cellulose, HPMC, magnesium stearate, D-mannitol was obtained from Central Drug House, India. All other reagents and solvents used were of analytical grade.
Methods:
Preparation of Solid Dispersions by the Solvent Evaporation Method: Solid dispersions were prepared using the solvent evaporation method, and the formula was mentioned in the below Table 1.
TABLE 1: SOLID DISPERSIONS FORMULATION TABLE
| Ingredients | SD1 (mg) | SD2 (mg) | SD3 (mg) |
| Amlodipine besylate | 10 | 10 | 10 |
| Ethyl cellulose | - | 10 | 10 |
| HPMC | 10 | 10 | - |
| Croscarmellose | 10 | - | 10 |
One gram of the drug was dissolved in a mixture of methanol and dichloromethane (1:1). Shaking very well was to ensure complete dissolving of the drug in the solvent mixture. A mixture of EC, HPMC, and croscarmellose equivalent to expected ratio poured in the solvent containing the drug. Stirring very well using mechanical stirrer and magnetic stirrer was till complete evaporation of the solvent. Sieving till give homogenous powder bypassed through a 200 μm sieve and retained on a 100 μm sieve. Subsequently, the sieved ground powders were stored at 25 °C in a desiccator in a screw-capped glass vial until use 8.
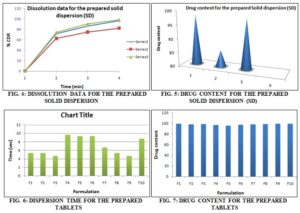
Dissolution Data for the Prepared Solid Dispersions: Dissolution study was performed for all the prepared solid dispersion by using USP-II paddle apparatus. In this 300 ml of saliva phosphate buffer pH, 6.8 was used and maintained temp 37 ± 0.5 ºC with 50 rpm. Then 2 ml of sample was withdrawn at every 5 min up to 15 min. and then it was analyzed by using UV-Visible spectrophotometer at 243 nm, and the values were tabled in below Table 2.
TABLE 2: DISSOLUTION DATA FOR THE PREPARED SOLID DISPERSION
| Time
(min) |
% CDR from the prepared solid dispersions | ||
| SD1 | SD2 | SD3 | |
| 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 |
| 5 | 71.9 ± 0.9 | 62.5 ± 0.4359 | 74.57 ± 0.3152 |
| 10 | 86.5 ± 0.6557 | 74.57 ± 1.250 | 90.73 ± 1.250 |
| 15 | 96.47 ± 0.6429 | 82.07 ± 1.901 | 98.7 ± 0.3606 |
Drug Content: The content of Amlodipine besylate in different solid dispersion was estimated using UV-Visible spectrophotometer. An accurately weighed quantity of solid dispersion (equivalent to 10 mg of Amlodipine besylate) was taken and dissolved in 10ml of methanol. From this solution, 1 ml of solution was diluted to 10 ml and assayed for drug content at 243 nm, and the values were tabled below Table 3.
TABLE 3: DRUG CONTENT FOR THE PREPARED SOLID DISPERSIONS
| Character | SD1 | SD2 | SD3 |
| Drug content | 98 ± 0.2 | 86.02 ± 1.709 | 97.97 ± 0.4509 |
Preparation of Tablets: Formulation chart for the prepared solid dispersions along with excipients were mentioned in Table 4. All the materials were passed through # 100 sieve before mixing. The solid dispersion was properly mixed with disintegrants, and then with the diluent D-mannitol, MCC. The mixture was mixed with aspartame, lactose, talc, and magnesium stearate. Finally, the blend was compressed to formulate tablet using a tablet compression machine with an average weight of 150 mg using a 6 mm flat punch in a rotary tablet press 9, 10.
TABLE 3: COMPOSITION OF TABLET FORMULATIONS (mg)
| Ingredients
(mg) |
Formulations | |||||||||
| F1 | F2 | F3 | F4 | F5 | F6 | F7 | F8 | F9 | F10 | |
| Drug solid dispersion | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 |
| Croscarmellose (Ac-Di-Sol) | 4 | 6 | 8 | - | - | - | - | - | - | - |
| Crospovidone (Polylplasdone) | - | - | - | 4 | 6 | 8 | ||||
| Sodium Starch glycol ate (Primogel) | - | - | - | - | - | - | 4 | 6 | 8 | - |
| Mannitol | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| Magnesium Stearate | 30 | 28 | 26 | 30 | 28 | 26 | 30 | 28 | 26 | 30 |
| Microcrystalline Cellulose | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 2 |
| Talc | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Lactose | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 2 |
| EC | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| HPMC | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 2 |
| Aspartame | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 2 |
Evaluation of Pre-compression Parameters (Preformulation Studies): 11, 12, 13
Bulk Density: Bulk density was determined by pouring the blend into a graduated cylimder. The bulk volume (Vb) and the weight of the blend (M) were determined. The bulk density is expressed in gm/ml and is given by:
Bulk density = M / Vb
Where, M = mass of powder taken, Vb = Bulk volume of the powder.
True Density: It tested measuring cylinder was tapped about several times, and later its true volume was measured, and true density was measured by using the following formula,
True density = {Weight of the powder (mg) / True volume of the powder (without void spaces) (ml)}.
Carr’s Index (Compressibility Index): It was determined by using the above-determined bulk density and true densities using the following formula,
Carr’s index (%) = {(Tapped density-bulk density) / Tapped density} × 100.
Hausner’s Ratio: It was also determined by using the above tapped and bulk density values using the following formula,
Hausner’s ratio = {Tapped density / Bulk density}
The Angle of Repose: Angle of repose (θ) was determined using the funnel method. The blend test sample powder was passed through a funnel that can be elevated perpendicularly until a maximum pinecone height (h) was obtained. The radius of the pile (r) was measured, and the angle of repose was calculated.
(θ) = tan-1(h/r)
Evaluation of Post-compression Parameters:14, 15
Thickness: It was measured by using Digital Vernier Calipers. Ten tablets from prepared formulation randomly taken and thickness was measured.
Weight Variation: By following USP monographs, it was calculated. In these 20 tablets from each formulation randomly taken and their total average weight was calculated, and then the individual tablet weight was calculated by comparing with the average weight.
Hardness: Hardness was calculated by using Pfizer hardness tester.
Friability Test: By following USP monograph, this test was calculated. It was measured by using Roche Friability. In this 20 tablets from each formulation was randomly taken and weighed and then placed all those 20 tablets in the plastic chamber of the Roche Friability and then it was attached to a motor for rotating with a speed of 25 rpm for 4 min, and then after 4 min all the 20 tablets weight was determined and friability was calculated.
Disintegration Time / Dispersion Time: This was determined by using glass beaker containing 6 ml of saliva buffer pH 6.8. Firstly 6 tablets from each formulation were randomly taken and dispersion time was calculated.
Drug Content: This was tested by taking 10 mg equivalent of Amlodipine besylate was taken from all formulation, and then it was dissolved in 10 ml of methanol. The form that 1 ml was pipetted out and it was made up to 10 ml using methanol, and then its absorbance was calculated using UV-Visible spectrophotometer at 243 nm of wavelength.
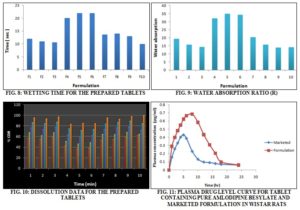
Wetting Time: Wetting time of the tablets was dignified using a piece of tissue paper (12 cm × 10.75 cm) folded twice, placed in a small Petri dish having a diameter of 6.5 cm and containing 6 ml of Saliva buffer (pH 6.8). A tablet was placed on the double folded tissue paper, and the time for the complete wetting was measured.
Water Absorption Ratio: It was tested by using double folded tissue paper, and the petri dish contains 6 ml of saliva buffer pH 6.8. Firstly randomly taken tablets to form the all formulations weight was calculated it was denoted as Wb, and then the tablets were allowed to place on the tissue paper. After completely wet of the tablet weight was calculated, and it was denoted as Wa. And by using the following formula water absorption ratio (R) was measured.
R = 100 × {(Wa – Wb) / Wb}
Where, Wb = Weight of tablet before absorption, Wa = Weight of tablet after absorption.
TABLE 4: PRECOMPRESSION PARAMETERS
| Formulations code | Bulk density
± S.D* |
Tapped density
± S.D* |
Angle of Repose ± S.D* | Hausner’s ratio ± S.D* | Carr’s index ± S.D* |
| F1
F2 F3 F4 F5 F6 F7 F8 F9 F10 |
0.379 ± 0.007
0.392 ± 0.004 0.368 ± 0.002 0.266 ± 0.08 0.371 ± 0.003 0.370 ± 0.25 0.392 ± 0.003 0.368 ± 0.002 0.376 ± 0.007 0.396 ± 0.002 |
0.443 ± 0.004
0.465 ± 0.004 0.413 ± 0.003 0.443 ± 0.004 0.442 ± 0.005 0.394 ± 0.006 0.442 ± 0.006 0.431 ± 0.003 0.456 ± 0.006 0.439 ± 0.003 |
39.82 ± 0.280
32 ± 0.095 32.53 ± 0.110 32.06 ± 0.051 30.18 ± 0.264 32.42 ± 0.03 32.51 ± 0.036 31.88 ± 0.448 32.12 ± 0.12 31.46 ± 0.04 |
1.169 ± 0.032
1.186 ± 0.019 1.122 ± 0.012 1.406 ± 0.017 1.192 ± 0.024 1.066 ± 0.020 1.129 ± 0.014 1.172 ± 0.003 1.212 ± 0.04 1.652 ± 0.027 |
14.42 ± 2.370
15.67 ± 1.380 10.89 ± 1.025 28.85 ± 0.867 16.1 ± 1.685 6.146 ± 1.806 11.44 ± 1.113 14.68 ± 0.235 17.44 ± 2.827 14.6 ± 0.045 |
TABLE 5: EVALUATION PARAMETERS FOR THE PREPARED TABLETS CHARACTERISTICS
| Characteristics | F1 | F2 | F3 | F4 | F5 | F6 | F7 | F8 | F9 | F10 |
| Colour | White | White | White | White | White | White | White | White | White | White |
| Odour | Odourless | Odourless | Odourless | Odourless | Odourless | Odourless | Odourless | Odourless | Odourless | Odourless |
| Taste | Acrid | Acrid | Acrid | Acrid | Acrid | Acrid | Acrid | Acrid | Acrid | Acrid |
| Shape | Flat | Flat | Flat | Flat | Flat | Flat | Flat | Flat | Flat | Flat |
TABLE 6A: EVALUATION PARAMETERS FOR THE PREPARED TABLETS
| Evaluation parameters | F1 | F2 | F3 | F4 | F5 |
| Hardness S.D* (kg/cm2) (N=20) | 3.56 ± 0.115 | 3.6 ± 0.2 | 3.56 ± 0.28 | 3.76 ± 0.25 | 3.63 ± 0.15 |
| Friability± S.D*(%) (N=20) | 0.65 ± 0.03 | 0.65 ± 0.03 | 0.62 ± 0.03 | 0.63 ± 0.02 | 0.03 ± 0.02 |
| Thickness ± S.D*(mm) (N=20) | 3.46 ± 0.11 | 3.48 ± 0.02 | 3.52 ± 0.02 | 3.51 ± 0.05 | 3.47 ± 0.14 |
| Weight variation ± S.D* (mm) (N=20) | 150.1 ± 0.11 | 150.2 ± 0.01 | 150.3 ± 0.43 | 151.5 ± 1.28 | 154.7 ± 2.51 |
| Drug content ± S.D* (N=20) | 99.13 ± 0.32 | 98 ± 1 | 99.0 ± 0.05 | 96.9 ± 0.65 | 95.8 ± 0.76 |
| Wetting time ± S.D* (Sec.) (N=6) | 12 ± 2 | 11 ± 1 | 10.6 ± 1.15 | 20 ± 2 | 22 ± 2 |
| Water absorption Ratio ± S.D* | 19.33 ± 1.52 | 15.67 ± 0.57 | 14.33 ± 0.57 | 32 ± 2 | 35 ± 1 |
| Dispersion time ± S.D* (Sec) (N=6) | 5.33 ± 0.57 | 5.33 ± 0.57 | 4.66 ± 0.57 | 9.66 ± 2.08 | 9.33 ± 1.15 |
TABLE 6B: EVALUATION PARAMETERS FOR THE PREPARED TABLETS
| Evaluation parameters | F6 | F7 | F8 | F9 | F10 |
| Hardness ± S.D* (kg/cm2) (N=20) | 3.86 ± 0.11 | 3.73 ± 0.20 | 3.93 ± 0.11 | 4 ± 2 | 4.99 ± 0.90 |
| Friability ± S.D*(%) (N=20) | 0.65 ± 0.03 | 0.64 ± 0.02 | 0.58 ± 0.01 | 0.63 ± 0.02 | 0.62 ± 0.07 |
| Thickness ± S.D*(mm) (N=20) | 3.47 ± 0.14 | 3.50 ± 0.05 | 3.52 ± 0.02 | 3.53 ± 0.11 | 3.83 ± 0.04 |
| Weight variation ± S.D* (mm) (N=20) | 150 ± 0.01 | 150.4 ± 0.5 | 149 ± 1 | 150.3 ± 1.5 | 150.5 ± 0.2 |
| Drug content ± S.D* (N=20) | 97.5 ± 0.5 | 98.2 ± 0.64 | 98.63 ± 0.5 | 99.23 ± 0.5 | 99.4 ± 0.45 |
| Wetting time ± S.D* (Sec.) (N=6) | 22 ± 2 | 13.67 ± 1.52 | 14 ± 2 | 13 ± 1 | 12.3 ± 0.97 |
| Water absorption ratio ± S.D* | 34.33 ± 0.57 | 20.33 ± 0.57 | 15.67 ± 0.57 | 14 ± 0 | 32 ± 0.42 |
| Dispersion time ± S.D* (Sec) (N=6) | 9.33 ± 0.57 | 6.66 ± 0.57 | 5.33 ± 0.57 | 4.66 ± 0.57 | 8.76 ± 0.22 |
In-vitro Dissolution Test: In-vitro dissolution studies for all the prepared formulations were carried by randomly picking the tablets from each formulation. This test was carried out using USP type II paddle apparatus at 50rpm using 300ml of Saliva phosphate buffer pH 6.8 as dissolution media, maintained at 37 ± 0.5 ºC. 1 ml aliquots were withdrawn at the specified time intervals, filtered through Whatmann filter paper and absorbance was calculated using UV-Visible spectrophotometer at 243 nm. An equal volume of fresh medium, which was pre-warmed at 37 ± 0.5 ºC, was replaced into the dissolution media after each sampling to maintain the constant volume thought the test 16.
TABLE 7A: DISSOLUTION DATA FOR THE PREPARED TABLETS
| % CDR | |||||
| Time (min) | F1 | F2 | F3 | F4 | F5 |
| 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 |
| 3 | 56.33 ± 0.5774 | 55 ± 2 | 59.67 ± 1.55 | 41.67 ± 1.528 | 36.67 ± 1.528 |
| 6 | 67 ± 1 | 62 ± 2 | 67 ± 2.646 | 51 ± 3 | 46 ± 3 |
| 9 | 76.33 ± 2.309 | 73.33 ± 4.163 | 77 ± 6.083 | 63 ± 3.606 | 59 ± 4 |
| 12 | 86 ± 3 | 84 ± 4.583 | 87 ± 4.933 | 74.67 ± 4.509 | 71.67 ± 3.055 |
| 15 | 95 ± 2.646 | 93 ± 3.606 | 96.67 ± 3.215 | 83 ± 2.646 | 85 ± 4.583 |
TABLE 7B: DISSOLUTION DATA FOR THE PREPARED TABLETS
| % CDR | |||||
| Time (min) | F6 | F7 | F8 | F9 | F10 |
| 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 |
| 3 | 38 ± 2.646 | 53 ± 2 | 54.33 ± 1.155 | 59 ± 3.1 | 62 ± 1.22 |
| 6 | 48.33 ± 4.512 | 64 ± 3.606 | 63.33 ± 2.08 | 67.67 ± 3.055 | 64 ± 3 |
| 9 | 61.67 ± 3.055 | 73 ± 1.732 | 76.33 ± 3.055 | 78.33 ± 3.055 | 72.22 ± 2.2 |
| 12 | 73 ± 6 | 83.67 ± 3.215 | 86.67 ± 4.933 | 89 ± 2.646 | 84 ± 2.602 |
| 15 | 84 ± 4.583 | 95 ± 2.646 | 92.67 ± 2.517 | 97.33 ± 1.528 | 99.33 ± 2.306 |
In-vivo Studies: The bioavailability studies for tablets with pure Amlodipine besylate, solid dispersion of Amlodipine besylate, were carried out using male Wistar rats (200-250 g). The animals were maintained in a clean room at a temperature between 20-25 °C with 12-h light and dark cycles and controlled relative humidity. The animals were fasted for 12 h before commencement of the study as well as during the study and had access to water ad libitum.
The Institutional Animal Ethical Clearance (BU/Pharm/IAEC/a/17/06) was obtained before conducting the studies. They were divided into three groups (two in each group); group I served as a control group whereas other two groups were treated with tablet formulation containing pure drug Amlodipine besylate, and tablet formulation containing solid dispersion of Amlodipine besylate, respectively. Tablets with a dose of 0.04 mg/kg body weight of rats were administered by dispersing in distilled water through oral feeding pipe. Blood samples were collected through the lateral tail vein of rats at 0, 5, 10, 15, 20, 30, 40, 50, and 60 min followed by 3, 8, 12, and 24 h after administration. The blood samples were centrifuged at 10 000 rpm for 10 min. After centrifugation, plasma was transferred into clean, fresh Eppendorf tubes and frozen at –20°C until assayed. The plasma concentration of drug was determined by High-Performance Liquid Chromatography (HPLC).
Stability Studies: The Stability studies indicated that there was no significant change observed for In vitro dissolution studies after three months. Fast-dissolving tablets F3 were found to be stable for 3 months at 40 ºC ± 2 ºC/75% Rh ± 5% Rh 17.
RESULTS AND DISCUSSION:
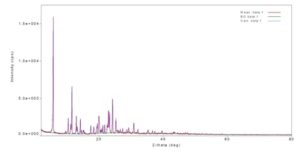
X-ray Powder Diffractometry Studies (XRPD): X-ray powder diffractometry studies (XRPD) is a powerful technique for the identification of the crystalline solid phase has a unique (XRPD). Every crystalline solid phase has a unique XRPD pattern which can form the basis for its identification 18.
FIG. 3: X-RAY POWDER DIFFRACTOMETRY (XRPD) OF PURE AMLODIPINE BESYLATE
CONCLUSION: The study showed that the dissolution rate of Amlodipine besylate was enhanced greater extent by a solid dispersion technique using the solvent evaporation method. The F3 batch is having the highest drug release and less disintegration time, which was optimized one. The prepared optimized tablet showed rapid disintegration as well as rapid dissolution as compared to a marketed tablet.
Furthermore, the fast dissolving tablet of Amlodipine besylate was successfully prepared by using different super disintegrants and the usage of this carrier for solid dispersion of Amlodipine besylates like EC, HPMC, and croscarmellose to enhance the dissolution profile. The in-vivo studies indicated that the solid dispersion approach could be adopted for the formulation of Amlodipine besylate tablets to achieve a faster onset of action.
ACKNOWLEDGEMENT: The authors thankful to Professor Dr. S. K. Prajapati for his valuable guidance.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Enin HAA: Development and evaluation of fast disintegrating extended-release tablets containing anti-hypertensive drug. Int J Pharm Pharm Sci 2014; 6: 165-74.
- Kumare MM, Marathe RP and Kawade RM: Design of fast dissolving tablet of Atenolol using novel co-processed super disintegrant. Asi J Pharm Clin Res 2013; 6(3): 81-85.
- Bhardwaj V, Bansal M and Sharma PK: Formulation and evaluation of fast dissolving tablets of Amlodipine besylate using different super disintegrants and camphor as a sublimating agent. Am-Euras J Sci Res 2010; 5(4): 264-69.
- Mohanachandran PS, Krishna Mohan PR, Saju F and Bini KB: Formulation and evaluation of mouth dispersible tablets of Amlodipine besylate. Int J Appl Pharma 2010; 2(3): 1-6.
- Pandit V, Pai RS, Devi K and Suresh S: In-vitro in-vivo evaluation of fast dissolving tablets containing solid dispersion of Pioglitazone hydrochloride. J Adv Pharm Technol Res 2012; 3(3): 160-170.
- Roy H, Parida KR, Nandi S, Panda SK and Mohapatra DK: Design of fast dissolving Amlodipine besylate tablet formulations. Asian Journal of Pharmaceutics 2012; 6: 51-59
- Mathews BR: Regulatory aspects of stability testing in Europe. Drug Dev Ind Pharm 1999; 831-56. (PubMed).
- Thakre A, Bhople A, Jaiswal S, Chandewa A, Ghuge N, Wagh N, Thakre S and Bari M: Formulation and development of oral fast dissolving tablet of Etoricoxib. Der Pharmacia Lettre 2012; 4 (4): 1169-1182.
- Kalia A, Khurana S and Bedi N: Formulation and evaluation of mouth dissolving tablets of Oxcarbazepine. Int J Pharn 2009; 1(S-1): 12-23.
- http://www.pharmpedia.com/Tablet:Formulation of tablets/Diluents.
- Najib NM, Suleiman M and Malakh A: Characteristics of the in-vitro release of ibuprofen from polyvinylpyrrolidone solid dispersions. Int J Pharm 1986; 32: 229-236.
- Ramu A, Vidyadhara S, Devanna N, Anusha C and Keerthi J: Formulation and evaluation of Rosuvastatin fast dissolving tablets. 2013; 25(10): 5340-5346.
- Banker GS: Sodium Starch Glycolate. Ainely Wade and Paul, London 1994.
- Lachman L and Liberman HA: Theory and Practice of Ind. Pharmacy, Varghese Publishing House 1998: 293.
- Siddiqui MN, Garg G and Sharma PK: Int Journal of Pharmaceutical Sciences Rev and Res 2010; 4(2): 87-96.
- Available at http;//www.drugs.com/pdr/amlodipine-besylate.html.
- Indian Pharmacopoeia, Ministry of Health and Family Welfare, Govt. of India. The controller of publications, New Delhi, Edition 4th, 1996: A-54.
- Gosai AR, Patil SB and Sawant KK: Formulation and evaluation of orodispersible tablets of ondansetron hydrochloride by direct compression using super disintegrants. Int J Pharm Sci Nanotech 2008; 1: 106-11.
How to cite this article:
Verma J, Prajapati SK, Chanchal DK, Bijauliya RK and Agnihotri N: Formulation and evaluation of fast dissolving tablet of Amlodipine besylate using different superdisintegrants containing solid dispersion by direct compression method. Int J Life Sci & Rev 2018; 4(4): 63-70. doi: 10.13040/IJPSR.0975-8232.IJLSR.4(4).63-70.
All © 2015 are reserved by International Journal of Life Sciences and Review. This Journal licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.
Article Information
2
63-70
835
1259
English
IJLSR
J. Verma *, S. K. Prajapati, D. K. Chanchal, R. K. Bijauliya and N. Agnihotri
Department of Pharmaceutics, Institute of Pharmacy, Bundelkhand University, Jhansi, Uttar Pradesh, India.
jyotivermaj924@gmail.com
08 February 2018
25 March 2018
15 April 2018
10.13040/IJPSR.0975-8232.IJLSR.4(4).63-70
30 April 2018