FORMULATION AND EVALUATION OF MOUTH DISSOLVING TABLET WITH TASTE MASKING RESIN
HTML Full TextFORMULATION AND EVALUATION OF MOUTH DISSOLVING TABLET WITH TASTE MASKING RESIN
G. R. Godge *, A. V. Misal and P. Y. Pawar
P.D.V.V.P. Foundation’s College of Pharmacy, Vilad Ghat, Ahmednagar - 414111, Maharashtra, India.
ABSTRACT: Mouth dissolving tablet disintegrates within a minute without drinking water. The purpose of present work was to develop a patient-friendly tablet for antihypertensive treatment using Benazepril HCl was taste masked by complexing with indion 204 and formulate as mouth dissolving tablet (MDT). The formulated drug-resin complex was characterized by IR, DSC, etc. Overall nine mouth dissolving formulations were prepared by direct compression method using three different superdisintegrants viz. croscarmellose sodium, sodium starch glycolate, and crospovidone. The tablets disintegrated in-vitro within 20 s, and the complex drug was released from tablet within 3 min. Formulation F1 to F3 were prepared by direct compression method using sodium starch glycolate as superdisintegrants was found to release the drug in the range of 88.20 to 91.98%. Similarly, formulation F4 to F6, which were prepared by using crospovidone as superdisintegrants showed maximum percent drug release, i.e. 88.27 to 92.68%. Further, F7 to F9 were prepared by using croscarmellose sodium as superdisintegrants and showed percent drug release between 87.72 to 89.18% range. Studies indicated that F4 was found to be best among all other formulations because it has exhibited faster wetting time, good taste, and faster disintegration time when compared to all other formulations.
Keywords: Disintegrate, Taste masking, Antihypertensive, Direct compression, Wetting time
INTRODUCTION: In recent years, a variety of pharmaceutical research has been conducted to develop new dosage forms. Most of the efforts have been focused on ease of medication 1. Among the dosage forms developed to facilitate ease of medication, the fast dissolving tablet (FDT) is one of the most widely employed dosage form 2, 3.
Difficulty in swallowing (dysphagia) is a common problem of all age groups, especially elderly and pediatrics, because of physiological changes associated with these groups of patients. Solid dosage forms that can be disintegrated dissolved or suspended saliva in the mouth resulting in easy swallowing can provide significant benefits to the pediatric and geriatric population, as well as other patients who prefer the convenience of easily swallowable dosage forms.
This tablet disintegrates instantaneously when placed on the tongue, releasing the drug that dissolves or disperses in the saliva. In case of taste masking the general aim is minimum free drug release in oral cavity 4. Taste is the ability to detect the flavor of substances like food, drugs, etc. Taste has now evolved as an important factor which governs the patient compliance. It gained importance as most of the drugs are administered through the oral route. Administration of unpalatable drugs is hampered by their unpleasant taste, particularly in case of pediatrics and geriatrics. Various methods like coating, inclusion complexes, microencapsulation, granulation, adsorption, prodrug approach, the addition of flavors and sweeteners, ion exchange resins are used for masking the taste of obnoxious drugs. However, there is no universal method for taste masking. Each method offers specific advantages and applications.
One method is not suitable for taste masking all the obnoxious drugs. Several parameters like the extent of the bitter taste, dose, dosage form, and type of the patient influence the method to be used for masking the taste of the bitter drugs. Evaluation of taste masking by the electronic tongue is a recent innovation. Advatab, Microcaps, Liquitard, Kleptose, Fourplex, and Formulcoat are the new taste masking technologies which are found to be better than existing ODT technologies like Zydis, Orasolv, and Quicksolv, etc. In addition to oral drug delivery, the taste masked drug delivery research is gaining importance for improving the quality of the treatment for pediatrics and geriatrics.
Benazepril hydrochloride is an angiotensin-converting enzyme inhibitor used in the treatment of hypertension. It is very bitter. To overcome this problem of the bitter taste, there is a need for taste masking. Conventional taste masking techniques such as the use of sweeteners, amino acids, flavoring agent are often ineffective in masking the taste of highly bitter drugs. Ion exchange resins are water-insoluble, crosslinked polymers containing salt-forming groups in repeating positions on the polymer chain. It can be used in the drug formulations to stabilize the sensitive components and to mask the taste of the bitter drug.
In the present study, an attempt has been made to form the drug, and Indion 204 complex is such that the average cation concentration of about 40 meq / l and pH of 6.7 in saliva was unable to break the complex. The complex was weak enough to be broken down by the hydrochloric acid present in the stomach with a good mouthfeel to prepare a “patient-friendly dosage form.” Thus the complex is tasteless and stable with no after taste, but at the same time, its bio-availability was found to be unaffected.
MATERIALS AND METHODS: Benazepril HCl was obtained as a gift sample from Aurobindo Pharmaceuticals, Hyderabad. Resin 204 was obtained as a gift sample from by Ion Exchange (India) Ltd. Mumbai. Sodium starch glycolate, cross-povidone, and croscarmellose sodium were purchased from S. D. Fine Chemicals, Mumbai. All other materials used were of pharmaceutical grade.
Methods:
Determination of the UV Spectrum in Phosphate Buffer 6.8: The stock solution of Benazepril HCl (100 µg/ml) was prepared by dissolving it in phosphate buffer 6.8. A dilution of 10 µg/ml was kept in the cuvette of path length 10 mm. The UV spectrum was recorded using double beam UV-VIS spectrophotometer (JASCO) in the wavelength range 200 nm- 400nm with Phosphate buffer 6.8 as blank.
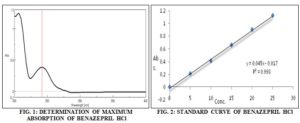
Preparation of Standard Curve in Phosphate Buffer 6.8: A stock solution of phosphate buffer pH 6.8 (100 µg/ml) was prepared by dissolving 10 mg of drug in Phosphate buffer pH 6.8, and the final volume was made to 100 ml. The solutions in the concentration range of 5-30 µg/ml were prepared by appropriate dilutions of stock solution. The UV absorbance of these solutions was determined spectrophotometrically at λ max 240 nm. The maximum wavelength of Benazepril HCl (λ max) was observed at 240 nm, which match with reported wavelength. Standard curve of Benazepril HCl was prepared in Phosphate buffer 6.8. When absorption platted against concentration Fig.1, 2, the linear curve developed Table 1. From above it concludes that drug follows Beer’s Lambert’s law and hence authentic one.
Taste Masking using Drug Resinate Complex (DRC): Formulation of DRC was done by the batch process; 100 mg of resin Indion 204 was placed in a beaker containing 100 ml of deionized water and allowed to swell for a definite period of time. Accurately weighed amount of (drug: resin ratio) was added and stirred for desired period of time. The mixture was filtered and residue was washed with deionised water. The filtrate was then analyzed using UV spectrophotometer at 240 nm for the calculation of unbound drug and percentage drug loading capacity.
TABLE 1: STANDARD CURVE OF BENAZEPRIL HCl
| Concentration ( μg/ml) | Absorbance (nm) |
| 5 | 0.2074 |
| 10 | 0.4094 |
| 15 | 0.6576 |
| 20 | 0.9018 |
| 25 | 1.1219 |
Effect of Concentration of Resin on Drug Loading: An accurately weighed amount of Benazepril HCl was added to the different concentration of Indion 204 for the determination of optimized ratio with maximum drug loading. Amount of maximum bound drug was determined at 240 nm using UV spectroscopy.
Characterization of DPC:
Infrared (IR) Study: The infrared spectrum of the pure Benazepril HCl sample was recorded, and the spectral analysis was done. The dry sample of drug with IR grade KBr in the ratio of 1:1. This mixture was compressed in the form of a pellet. The pellets were scanned over a wave number range of 4000 to 400 cm-1 in FTIR instrument (Shimadzu,) and spectral analysis was done.
Differential Scanning Calorimetry (DSC) Study: The differential calorimetric scanning of benazepril hydrochloride and formulation was carried out using Mettler Toledo 821e Differential Scanning Calorimeter. Samples of the drug were placed in aluminium crucibles, and DSC thermograms were recorded at the heating rate of 10 °C/min in the range of 0 °C to 300 °C. Nitrogen gas was purged at the rate of 30 ml/min to maintain the inert atmosphere.
Determination of Drug Content: 5 DPC equivalent to 20 mg of drug was stirred by using magnetic stirrer with 100 ml of 0.1 N HCl for 60 min, till the entire drug leached out from complex, then the solution was filtered through Whatman filter paper. Further, solution diluted with 0.1 N HCl and the drug content was determined spectrophotometrically at 240 nm.
Taste Evaluation of Drug-Resin Complex (DRC): Volunteers performed taste evaluation of DRC in the age group of 19 to 22 years. The study protocol was explained, and written consent was obtained from volunteers. DRC equivalent to 10 mg Benazepril hydrochloride was held in the mouth for 30 sec by each volunteer. Bitterness levels were recorded instantly and then after 30 sec, 60 sec. The bitterness level was recorded against pure drug using a numerical scale.
A numerical scale was used with the following values: 0 = tasteless, 1= acceptable bitterness, 2= slight bitterness, 3= moderately bitterness and 4= strong bitterness
Formulation Development:
Formulation Composition: Fast dissolving tablets of Benazepril HCl were prepared by direct compression method after incorporating different concentrations indion-204 as taste masking resin. Mannitol, microcrystalline cellulose, magnesium stearate were used as directly compressible diluents along with different conc. of superdisintegrants viz. sodium starch glycolate, cross-carmellose sodium, cross-povidone. Composition of tablets is mentioned in Table 2. All ingredients were passed through mesh #60. The required quantity of each was taken for a particular formulation, and the blend was mixed by using the poly bag for 15 min. Blend was compressed on 7 mm (diameter) bi concave punches on a ‘CAP’ 8 station rotary compression mission. Each tablets weighing 300 mg, were obtained.
Pre-Compression Properties: 6, 7 Pre-compression properties of granules such as bulk density, tapped density, compressibility index, and angle of repose were determined.
Evaluation of Tablets: The prepared tablets hardness was measured by using Monsanto hardness tester. The hardness was measured in terms of kg/cm2. 8, 9 Thickness of prepared tablets was tested by vernier calipers, and the average was calculated. Twenty tablets were selected at random and weighed individually. The individual weights were compared with the average weight for determination of weight variation 10.
The percentage weight deviation was calculated and then compared with USP specifications. Five tablets were powdered, from the blend equivalent to 20 mg of Benazepril HCl was weighed and dissolved in a suitable quantity of pH 1.2 0.1 HCl. The solution was filtered and suitably diluted, and drug content was analyzed spectrophotometrically at 240 nm.
A piece of tissue paper folded twice was placed in a small Petri dish (internal diameter = 6.5cm) containing 5 ml of distilled water. A tablet was placed on the paper, and the time for complete wetting of the tablet was measured 11, 12, 13.
Disintegration Time: 14 Tablets were taken and introduced in each tube of disintegration apparatus, and the tablet rack of the disintegration apparatus was positioned into a beaker containing 900 ml of distilled water and the disintegration time was recorded. To discriminate between the formulations disintegration was done at room temperature.
TABLE 2: COMPOSITION OF BENAZEPRIL HCl MOUTH DISSOLVING TABLETS
| S. no. | Tablet ingredients(mg/tab) | F1 | F2 | F3 | F4 | F5 | F6 | F7 | F8 | F9 |
| 1. | DRC equivalent to 20 mg of drug | 168 | 168 | 168 | 168 | 168 | 168 | 168 | 168 | 168 |
| 2. | Sodium starch glycolate | 5 | 4 | 3 | - | - | - | - | - | - |
| 3. | Crosscarmellose | - | - | - | - | - | - | 5 | 4 | 3 |
| 4. | Crosspovidone | - | - | - | 5 | 4 | 3 | - | - | - |
| 5. | Sodium saccharin | 6 | 6 | 6 | 6 | 6 | 6 | 6 | 6 | 6 |
| 6. | Mannitol | 20 | 20 | 20 | 20 | 20 | 20 | 20 | 20 | 20 |
| 7. | Microcrystalline cellulose | 96 | 96.5 | 97 | 96 | 96.5 | 97 | 96 | 96.5 | 97 |
| 8. | Magnesium stearate | 1 | 1.5 | 2 | 1 | 1.5 | 2 | 1 | 1.5 | 2 |
| 9. | Talc | 4 | 4 | 4 | 4 | 4 | 4 | 4 | 4 | 4 |
In-vitro Drug Release Studies: The in-vitro dissolution study was carried out in the USP dissolution test apparatus type 2 (paddle). 900 ml of the dissolution medium (0.1 N HCl solution) was taken in, and the temperature was maintained at 37 ± 0.5 °C. The speed of the paddle was maintained at 100 rpm. Sampling was done every one min interval. For each sample, 5 ml of the dissolution medium was withdrawn, and the same amount of dissolution medium was replenished to maintain sink condition. The samples withdrawn were analyzed in the UV spectrophotometer at 240 nm 15, 16, 17.
RESULTS AND DISCUSSION:
Effect of Concentration of Resin on Drug Loading: While studying the effect of concentration of resin on drug loading, maximum drug loading was found in ratio1:4 (drug: Indion-204) Table 3.
TABLE 3: EFFECT OF CONCENTRATION OF RESIN ON DRUG LOADING
| Drug: Resin Ratio | % Drug bound |
| 1:2 | 82.94 |
| 1:3 | 88.35 |
| 1:4 | 90.78 |
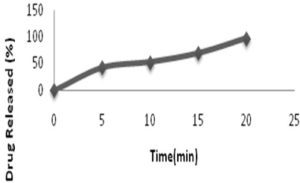
In-vitro Drug Release Study from Resonate: Samples were withdrawn, analyzed at 240 nm and %drug release was determined. In vitro drug release study was performed in 0.1 N HCl Fig. 3. The drug released from Indion 204 was found to be more than 97% within 20 minutes in 0.1 N HCl which shown in Fig. 5.
Taste Evaluation: Taste evaluation revealed that Indiaon-204 masks the bitter taste of the drug.
FIG. 3: IN-VITRO DRUG RELEASE STUDY FROM DRUG-RESIN COMPLEX
TABLE 4: TASTE EVALUATION OF DRC
| Volunteers | 30 sec | 1 min | 2 min |
| 1 | 1 | 1 | 1 |
| 2 | 2 | 1 | 1 |
| 3 | 1 | 1 | 1 |
0=tasteless, 1=acceptable bitterness, 2=slightly bitterness, 3=strongly bitterness
Evaluation of Tablets: The values of bulk density and tapped density were found in the range from 0.8221 to 0.867 g/ml and from 0.9121 to 0.9916 g/ml, respectively. The Carr’s compressibility indices were in the range of 5.30 to 12.48 % and angle of repose (θ) was in the range of 24.67 to 28.08°. This indicates that formulations have good flow property.
TABLE 5: PRECOMPRESSION PARAMETERS OF BENAZEPRIL HCl
| Batch | Angle of
repose(θ) |
Bulk Density (gm/cm2) | Tapped Density (gm/cm2) | Carr’s
Index |
| F1 | 26.06±0.04 | 0.8221±0.03 | 0.9247±0.02 | 12.48 |
| F2 | 24.67±0.01 | 0.864±0.02 | 0.9234±0.04 | 11.73 |
| F3 | 28.08±0.02 | 0.845±0.02 | 0.9916±0.01 | 9.67 |
| F4 | 25.55±0.02 | 0.867±0.02 | 0.9135±0.02 | 5.30 |
| F5 | 23.67±0.02 | 0.8284±0.03 | 0.9286±0.04 | 11.96 |
| F6 | 27.08±0.01 | 0.8321±0.02 | 0.9221±0.02 | 10.81 |
| F7 | 28.08±0.01 | 0.8439±0.04 | 0.9121±0.02 | 8.49 |
| F8 | 26.06±0.04 | 0.8294±0.02 | 0.9296±0.03 | 12.08 |
| F9 | 24.85±0.01 | 0.8221±0.03 | 0.9247±0.02 | 12.48 |
TABLE 6: POST- COMPRESSION PARAMETERS OF BENAZEPRIL HCl
| Parameter | F1 | F2 | F3 | F4 | F5 | F6 | F7 | F8 | F9 |
| Dispersion time (Sec) | 80 | 65 | 82 | 52 | 55 | 60 | 65 | 55 | 55 |
| Wetting time (Sec) | 82 | 80 | 52 | 40 | 42 | 42 | 55 | 52 | 50 |
TABLE 7: POST -COMPRESSION PARAMETERS OF BENAZEPRIL HCl
| Batch | Weight Variation (%)
±SD, n=20 |
Friability (%)
± SD, n=20 |
Hardness
(Kg/cm2) n=3 |
Thickness
(mm) n=3 |
Drug content
(%) |
| F1 | 299.33±0.28 | 0.88±.001 | 3.5 | 4 | 88 |
| F2 | 299.33±0.57 | 0.66±0.03 | 3.5 | 4.2 | 93 |
| F3 | 299.66±0.57 | 0.48±0.03 | 3 | 4 | 95.30 |
| F4 | 299.16±0.28 | 0.41±0.01 | 3.5 | 4.1 | 97.47 |
| F5 | 299.33±0.57 | 0.54±0.01 | 3 | 4 | 94 |
| F6 | 299.33±0.28 | 0.66±0.03 | 4 | 4 | 95.3 |
| F7 | 299.66±0.57 | 0.8±0.04 | 3.5 | 4.1 | 89 |
| F8 | 299.33±0.28 | 0.60±0.02 | 4 | 4.1 | 88.5 |
| F9 | 299.66±0.57 | 0.54±0.01 | 4 | 4 | 90 |
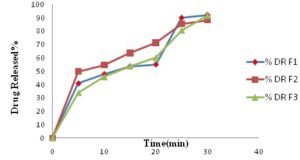
Dissolution Profile: 18-22 All the nine formulations were subjected for the in-vitro dissolution studies using tablet dissolution tester USP II. The samples were withdrawn at different time intervals and analyzed at 240 nm. Cumulative drug release and cumulative percent drug retained were calculated based on the mean amount of Benazepril HCl present in the respective tablet. Formulation F1 to F3 were prepared by direct compression method using sodium starch glycolate as superdisintegrants was found to be drug release in the range of 91.46% to 91.98.
TABLE 8: PERCENT DRUG RELEASED OF BENAZEPRIL HCl-RESIN BASED COMPLEX FORMULATIONS USING SODIUM STARCH GLYCOLATE AS SUPERDISINTEGRANTS
| Time
(min) |
% Drug Released | ||
| F1 | F2 | F3 | |
| 0 | 0 | 0 | 0 |
| 5 | 40.85 | 49.8 | 33.81 |
| 10 | 47.82 | 54.7 | 45.62 |
| 15 | 53.67 | 63.6 | 53.65 |
| 20 | 54.9 | 71.3 | 59.91 |
| 25 | 89.9 | 85.5 | 80.14 |
| 30 | 91.98 | 88.2 | 91.46 |
FIG. 4: DISSOLUTION PROFILES OF BENAZEPRIL HCl-RESIN BASED COMPLEX FORMULATIONS USING SODIUM STARCH GLYCOLATE AS SUPERDISINTEGRANTS
TABLE 9: PERCENT DRUG RELEASED OF BENAZEPRIL HCl-RESIN BASED COMPLEX FORMULATIONS USING CROSSPOVIDONE AS SUPERDISINTEGRANTS
| Time
(min) |
% Drug Released | ||
| F4 | F5 | F6 | |
| 0 | 0 | 0 | 0 |
| 5 | 32.92 | 34.80 | 28.27 |
| 10 | 57.51 | 43.70 | 43.33 |
| 15 | 63.63 | 49.88 | 53.04 |
| 20 | 74.31 | 56.28 | 65.21 |
| 25 | 81.66 | 79.83 | 83.85 |
| 30 | 92.68 | 88.27 | 90.42 |
FIG. 5: DISSOLUTION PROFILES OF BENAZEPRIL HCl-RESIN BASED COMPLEX FORMULATIONS USING CROSSPOVIDONE AS SUPERDISINTEGRANTS
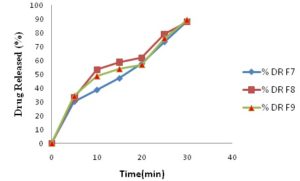
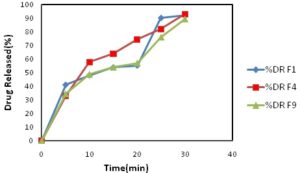
Here F1 shows maximum percent drug released as shown in Fig. 4 & Table 8. F4 to F6 were prepared by using Crosspovidone as superdisintegrants, F4 shows maximum %DR, i.e., 92.68% Fig. 5 & Table 9. Further, F7 to F9 were prepared by using croscarmellose sodium as superdisintegrants; Moreover, F9 showed maximum %DR, i.e., 89.18% Fig. 6 & Table 10. When comparing the dissolution profile of three super disintegrants, F4 shows maximum drug released 92.68 % within 30 min. F4 prepared by direct compression using Crosspovidone showed good drug release (92.68%) than other formulation Fig. 7.
TABLE 10: PERCENT DRUG RELEASED OF BENAZEPRIL HCl-RESIN BASED COMPLEX FORMULATIONS USING CROSSCARMELLOSE AS SUPERDISINTEGRANTS
| Time
(min) |
%Drug Released | ||
| F7 | F8 | F9 | |
| 0 | 0 | 0 | 0 |
| 5 | 30.25 | 32.87 | 34.21 |
| 10 | 38.73 | 53.16 | 48.55 |
| 15 | 47.09 | 58.66 | 53.71 |
| 20 | 57.74 | 61.76 | 56.80 |
| 25 | 73.42 | 78.93 | 75.84 |
| 30 | 88.43 | 87.72 | 89.18 |
FIG. 6: DISSOLUTION PROFILES OF BENAZEPRIL HCl-RESIN BASED COMPLEX FORMULATIONS USING CROSSCARMELLOSE AS SUPERDISINTEGRANTS
FIG.7: COMPARISON OF DISSOLUTION PROFILES OF BENAZEPRIL HCl-RESIN BASED COMPLEX FORMULATIONS USING DIFFERENT SUPERDISINTEGRANTS
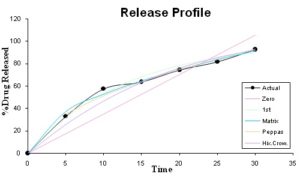
TABLE 11: IN-VITRO DRUG RELEASE KINETICS OF F4 FORMULATION
| Models | R2 value | K value |
| Zero order | 0.9071 | 3.5 |
| 1st order | 0.9768 | -0.07 |
| Matrix | 0.9961 | 16.6 |
| Korsmeyer- Peppas | 0.9860 | 14.5 |
| Hixon- Crowel | 0.9854 | -0.01 |
FIG. 8: IN-VITRO DRUG RELEASED FROM F4 FORMULATION
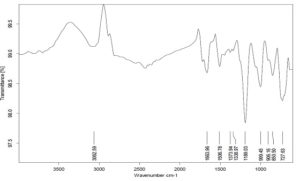
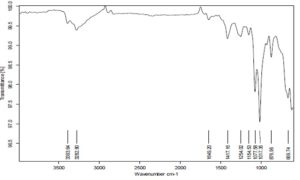
FTIR Studies: 23-27 The IR spectra of pure drug shows characteristic functional peaks at 3062.59 cm-1, 1663.96 cm-1, 1506.78 cm-1, 1189.03 cm-1 and 727.63 cm-1 Fig. 9-10. The IR spectrum of the complex shows characteristic functional peaks at 3393.64 cm-1, 1649.20 cm-1, 1417.16 cm-1, 1077.58 cm-1 and 669.74 cm-1. The similarity in the peaks when compared to pure drug and physical mixtures indicated that the compatibility of the drug with excipients.
DSC Analysis: DSC analysis of the prepared complex was carried out to check any changes in a thermal property of drug due to complexation. Pure benazepril HCl thermogram was a single, sharp melting endotherm at 187.90 °C Fig. 11.
In case of prepared complex shortened endothermic peak appears at 162.78 °C Fig. 12 due to ethanol dissolution state.
FIG. 9: IR SPECTRA OF BENAZEPRIL HYDROCHLORIDE
FIG. 10: IR SPECTRA OF BENAZEPRIL HYDROCHLORIDE + EXCIPIENTS
FIG. 11: DSC OF BENAZEPRIL HYDROCHLORIDE
FIG. 12: DSC OF BENAZEPRIL HYDROCHLORIDE + EXCIPIENTS
Thus it was proved that there was no major difference in thermograms; hence, the excipients were compatible with the drug is chosen and so could be safely used to formulate FDT.
CONCLUSION: The study was concluded that taste masking of Benazepril HCl and rapid disintegration and dissolution of MDT was attained successfully. Taste masking and rapid disintegration of tablets formulated in this study was novel formulation. Thus, the patient-friendly dosage form of bitter drug Benazepril HCl can be successfully formulated with the use of cation exchange resin offers good method for preparing taste-masked substrate of Benazepril HCl. The study was concluded that the complete taste masking of Benazepril HCl after the addition of sodium saccharin.
Thus, complexation of Benazepril HCl with Indion 204 increases acceptability and palatability of formulated rapid disintegrating tablets. Taste masking with resin and rapid disintegration of tablets formulated in this study was novel formulation. Thus, the patient-friendly dosage form of bitter drug Benazepril HCl can be successfully formulated.
ACKNOWLEDGEMENT: Authors want to express their gratitude for continuous support and encouragement by the principal, P.D.V.V.P. Foundation's college of pharmacy, Ahmednagar. We also thank Prof. A. N. Merekar and Dr. R.S. Godge for giving his valuable guidance & support during the preparation of the manuscript.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Hanawa T, Watanabe A, Tsuchiya T and Ikoma R: New oral dosage form for elderly patients: Preparation and characterization of silk fibroin gel. Chem Pharm Bull 1995; 43: 284-88. http://dx.doi.org/10.1248/cpb.43.284
- Watanable Y, Ishikawa Y, Utoguchi N and Matsumoto M: Preparation and evaluation of tablets rapidly disintegrating in salive containing bitter-taste-masked granules by the compression method. Chem Pharm Bull 1999; 47(10): 1451-54. http://dx.doi.org/10.1248/cpb.47.1451
- Kaushik D, Dureja H and Saini T: Mouth dissolving tablets: A review. Indian Drugs-Bombay 2004; 41: 187-93.
- Patel AR and Vavia PR: Preparation and evaluation of taste masked famotidine formulation using drug/β-cyclodextrin/ polymer ternary complexation approach. AAPS Pharm Sci Tech 2008; 9(2): 544-50.
- Aurora J and Pathak V: Oral Disintegrating Technologies: oral disintegrating dosage forms: An overview. Drug Delivery Technology 2005; 5(3): 50-54.
- Jhonson B, Roache J and Javors M: Ondansetron for reduction of drinking among biologically predisposed alcoholic patients: A randomized controlled trial. JAMA 2000; 284: 963-71.
- Arwood R: Man Chem 196; 61: 36-7.
- Ghuge N, Bhopal A, Thakare A, Bakade B, Bari M and Deshmukh M: Der Pharmacia Letter 2012; 4(6) 1721-36.
- Makwana S, Patel L and Patel T: J Pharm Sci & Res 2010; 2(4): 231-239.
- Jagadale S, Gattani D, Bhavsar D, Chabukswar A and Kuchekar B: Formulation and evaluation of taste masked orally disintegrating tablet of tinidazole. Asian J Pharm Clin Res 2011; 4(4): 25-28.
- Dahima R and Sharma R: Formulation and in-vitro evaluation of taste masked orodispersible tablet of metoclopramide hydrochloride using indion 204. Int J Chem Tech Res 2010; 2(1): 447-453.
- Saroha K, Mathur P, Varma S, Syan N and Kumar A: Mouth dissolving tablets: An overview on future compaction in oral formulation technologies. Der Pharmacia Sinica 2010; 1(1): 179-187.
- Aly A, Semreen M and Qato M: Pharmaceutical Technology 2005; 68-78.
- Nautiyal U, Singh S, Singh R and Kakar S: Fast dissolving tablets as a novel boon: A review. J of Pharmaceutical, Chemical and Biological Sciences 2014; 2(1): 05-26.
- Bhanja S, Hardel D and Sudhakar M: Formulation and evaluation of mouth dissolving tablets of losartan potassium. Int J Curr Pharm Res 2012; 4(4): 15-23.
- Vummaneni V and Nagpal D: Int J of Res in Pharma and Biomed Scie 2012; 3(2): 511-524.
- Salunke T and Mayee R: International Journal of Pharmaceutical Innovations 2013; 5(3): 59-66.
- Sharma D, Kumar D, Singh M, Singh G and Singh R: Taste masking technologies: a novel approach for the improvement of organoleptic property of pharmaceutically active substance. IRJP 2012; 3(4): 108-116.
- Suthar A and Patel M: Int J Pharma Scie 2010; 1(2): 6-12.
- Biladi A and Mishra A: International Journal of Advanced Pharmaceutics 2014; 4(2): 134-145.
- Belal F, Abdine H and Abdullah A: (Benazepril HCl: Comprehensive Profile) 2014; 31, 121, 128, 129. http://dx.doi.org/10.1016/s0099-5428(04)31003-8
- Crowe R, Paul J, Sheskey S and Marian E: Handbook of excipients. 2009; 6: 208, 404, 118, 206.
- Godge G and Hiremat S: Development and evaluation of colon targeted drug delivery system by using natural Polysaccharides/Polymers. Dhaka University Journal of Pharmaceutical Sciences. 2014; 13(1): 105-113.
- Godge G and Hiremath S: Colonic delivery of film coated meloxicam tablets using natural polysaccharide polymer mixture. Int Current Pharma Journal 2012; 1(9): 264-271.
- Godge G and Hiremath S: Mahidol University Journal of Pharmaceutical Sciences 2014; 41(2): 7-21.
- Godge G and Hiremath S: Int J Pharm Dru Ana 2014; 2(1): 3.
- Hiremath S and Godge G: Preparation and in-vitro Evaluation of inclusion complexes of nelfinavir with chemically modified?-cyclodextrins. Dhaka University Journal of Pharmaceutical Sciences 2013; 11(2): 107-116.
How to cite this article:
Godge GR, Misal AV and Pawar PY: Formulation and evaluation of mouth dissolving tablet with taste masking resin. Int J Life Sci & Rev 2015; 1(7): 253-63. doi: 10.13040/IJPSR.0975-8232.IJLSR.1(7).253-63.
All © 2015 are reserved by International Journal of Life Sciences and Review. This Journal licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.
Article Information
3
253-263
920
2244
English
IJLSR
G. R. Godge *, A. V. Misal and P. Y. Pawar
P.D.V.V.P. Foundation’s College of Pharmacy, Vilad Ghat, Ahmednagar, Maharashtra, India.
grgodge@yahoo.com
28 June 2015
28 July 2015
29 July 2015
10.13040/IJPSR.0975-8232.IJLSR.1(7).253-63
31 July 2015