FORMULATION, DEVELOPMENT AND CHARACTERISATION OF MICROEMULSION SYSTEM OF AMOXICILLIN
HTML Full TextFORMULATION, DEVELOPMENT AND CHARACTERISATION OF MICROEMULSION SYSTEM OF AMOXICILLIN
Ajay Rastogi * 1, Ram Narayan Prajapati 1, S. K. Prajapati 1, Rohit Kumar Bijauliya 2, Dilip Kumar Chanchal 2 and Santosh Kumar 3
Department of Pharmaceutics 1, Department of Pharmacognosy 2, Institute of Pharmacy, Bundelkhand University, Jhansi - 284128, Uttar Pradesh, India.
Hygia Insitute of Pharmaceutical Education and Research 3, Faizullahganj, Prabhandh Nagar, Ghaila Road, Lucknow - 226020, Uttar Pradesh, India.
ABSTRACT: Present study aimed to develop a microemulsion system for topical application of Amoxicillin to provide various advantages over conventional dosage forms. Amoxicillin is broad-spectrum semisynthetic antibiotic similar to ampicillin except that its resistance to gastric acid permits higher serum levels with oral administration. Amoxicillin is commonly prescribed with clavulanic acid (a beta-lactamase inhibitor) as it is susceptible to beta-lactamase degradation. Clinical studies show that it is highly effective for topical treatment of skin infections. The microemulsion was prepared using oleic acid as an oil phase, tween 20 as surfactant and propylene glycol as co-surfactant. The drug was evaluated for their micropolitical properties like the angle of repose, bulk density; tapped density, Carr's index, and Hausner's ratio were studied. In-vitro characterization of the microemulsion of amoxicillin was done through measurement of pH, conductivity, viscosity, drug stability.
Keywords: Amoxicillin, Microemulsion, Penetration, Topical
INTRODUCTION: UV irradiation causes oxidative stress to the skin by inducing the generation of reactive oxygen species exceeding the antioxidant defense ability of cells. Thus, the use of naturally occurring herbal antioxidants has gained considerable interest to protect the skin from adverse biological effects of solar UV irradiation.
Silymarin, flavonolignans isolated from milk thistle, is generally used for several liver disorder conditions such as cirrhosis, chronic hepatitis, and liver diseases associated with alcohol consumption 1, 2, 3. Recently, topical application of silymarin has received attention because of its antioxidant, anti-inflammatory, and immunomodulatory properties, which may prevent UV-induced skin disorders including erythema, photoaging, and skin cancer 4, 5. To achieve skin benefits, effective amounts of amoxicillin require to be solubilised and incorporated into the corresponding formulation.
The Modern system of medicines possesses many types of dosage form for treating dermatological diseases. The conventional dosage forms include pastes, ointments, creams, gels but due to their certain shortcomings new forms came into the picture for topical delivery to name a few, micro-emulsion, lipogels, organogels, micelle system, etc. these dosage forms offer improved drug diffusion, spreadability, and thermodynamic stability. Two immiscible liquids (e.g., water and oil) can be brought into a single phase with appropriate use of surfactant and co-surfactant(s). This thermo-dynamically stable optically clear system is called as ‘Microemulsion.’
In the present study microemulsion-gel system has been prepared with the help of an appropriate concentration of surfactant and co-surfactant. It is considered that the envelope formed by surfactant monolayer at the oil/water interface decides phase behavior and microstructure of final formulation hence tween 20 and polyethylene glycol 400 selected as surfactant and co-surfactant respectively as it gives oil-in-water (O/W) microemulsions 6, 7, 8. Present work was an attempt to develop a stable microemulsion system to provide greater loading capacity, good bioavailability, and penetrability of amoxicillin across the skin after topical application.
MATERIALS AND METHODS:
Materials: Amoxicillin is a gifted sample of Micro Labs Limited, Bangalore. Surfactant tween 20, tween 40, tween 60, tween 80 purchased from Himedia Laboratories Pvt. Ltd. Mumbai, India.
Methods:
Preformulation Study:
Micromeritics Study: The drug was evaluated for their micromeritics properties. Different parameters like angle of repose, bulk density, tapped density, Carr's index, and Hausner's ratio were studied. The bulk density of a powder is the ratio of the mass of an untapped powder sample and its volume including the contribution of the interparticulate void volume. It was determined by measuring the volume of a known mass of powder sample that has been passed through a sieve into a graduated cylinder, and the bulk density was calculated in g/mL by the formula m/V0 (mass/apparent volume). Tapped density is an increased bulk density attained after mechanically tapping a container containing the powder sample, and it was determined by tapping a measuring cylinder 100 times and reading the volume after tapping.
Carr’s Index: It indicates the ease with which a material can be induced to flow. It was calculated from the bulk and tapped densities by using the following formula
Carr’s index =Tapped density-Bulk density / Tapped density× 100
Hausner’s Ratio: The Hausner's ratio is related to the interparticle friction and is correlated to the flowability of powder material. It was calculated from bulk density and tapped density by using the following formula:
Hausner’s Ratio =Tapped density/Bulk density
The angle of Repose: Angle of repose is used to measure the flow property of powder materials. It was determined by the fixed funnel method. A funnel was fixed to a stand and bottom of the funnel was fixed at the height of 2 cm from the plane. Powder samples were placed in the funnel and allowed to flow freely. The height and radius of the heap were measured, and the angle of repose was calculated from the following formula:
Tan θ = h / r
Where, h = height of heap, r = radius of the heap.
Melting point: The melting point was determined by the capillary method. For the melting point measurement drug sample was placed in a thin-walled capillary tube 10-15 cm long, about 1 mm in inside diameter, and closed at one end. The capillary, which contains the sample, and a thermometer were then suspended in a beaker containing liquid paraffin so they can be heated slowly and evenly. The temperature range over which the sample started to melt was taken as the melting point.
Partition Coefficients: To determine the partition coefficients of Amoxicillin; n-octanol and pH 7.4 phosphate buffer were saturated with each other for the period of 24 h in 50 ml volumetric flask. 0.1% w/v of the drug was added to the mixture of the above saturated solution in 50 ml volumetric flask and shaken for 24 h on a rotary shaker at room temperature. After equilibrium for another 24 h, the system was centrifuged for 15 min at 3000 rpm in a centrifuge. The concentration of Amoxicillin in n-octanol and pH 7.4 phosphate buffer were analyzed by UV visible spectrophotometer at 381.4 nm after appropriate dilution with methanol. The partition coefficient was calculated according to the following equation. The study was repeated thrice to confirm the reproducibility of the result.
Partition coefficients = Drug concentration in n-octanol / Drug concentration in pH 7.4 phosphate buffer
Screening of Oils: The solubility of Amoxicillin in various oils, surfactants, co-surfactants, and various media was determined by using the shake flask method. Briefly, an excess amount of Amoxicillin was added to each 10 ml volumetric flask containing 2 ml of the selected vehicle, i.e., oil, surfactant, co-surfactants and various media and mixed for 10 min using a vortex mixer to facilitate proper mixing of Amoxicillin with the vehicles. Mixtures were shaken for 72 h in a rotary shaker at 37 °C. After equilibrium, mixtures were centrifuged at 3,000 rpm for 15 min, followed by filtration through Whatman filter which has pore size 125 mm. The concentration of Amoxicillin in the filtrate was determined by UV visible spectrophotometer at 381.4 nm after appropriate dilution with methanol.
Screening of Surfactant: The surfactant was evaluated based on their potential to emulsify the selected oil phase (oleic acid). Briefly, 10 ml of 10% v/v aqueous solution of surfactant (tween 20, tween 40, tween 60, tween 80, gelucire 50/13 and gelucire 44/14) was titrated individually by selected oil 20 µl at a time until it becomes hazy and notes down the total amount of oil consumed.
Screening of Co-surfactant: Pseudo-ternary phase diagrams were constructed by an oil titration method to optimize the co-surfactant. For the selection of co-surfactant different co-surfactant (PEG 200, PEG 400, PEG 600, propylene glycol, ethanol, butanol, isopropyl alcohol). For the study purpose surfactant (tween 80) to the co-surfactant ratio (Smix) was fixed to 1:1. For each phase diagram, water and specific Smix ratio were mixed thoroughly in different weight ratios from 1:9 to 7:3 in different glass vials. Fourteen different combinations of water and Smix, 1:9, 1:8, 1:7, 1:6, 1:5, 2:8 (1:4), 1:3.5, 1:3, 3:7 (1:2.3), 1:2, 4:6 (1:1.5), 5:5 (1:1), 6:4 (1:0.7), 7:3 (1:0.43), were made so that maximum ratios were covered for the study to delineate the boundaries of phases precisely formed in the phase diagrams. To a clear mixture of water and Smix mixture in different ratios, oil was added in steady increments of 20 µl, and the mixture was vortexed using vortex shaker until the addition of oil turned the mixture milky. No attempts were made to identify different phase systems, and the phase diagram shows only the existence of the micro-emulsion area.
The physical state of the microemulsion was marked on a pseudo-three-component phase diagram with one axis representing aqueous phase, the other representing oil and the third representing a mixture of surfactant and co-surfactant at fixed ratios (Smix ratio). At the end of the titration, the concentrations of the component were recorded, and pseudo-ternary phase diagram was constructed using Triplot 4.1.2 trial version. The area covered by the points was considered to be the microemulsion region.
% A (Oil) + % B (Smix) + % C (Water) = 100
Optimization of Surfactant and Co-surfactant Ratio (Smix): The surfactant to co-surfactant ratio was also optimized by constructing the pseudo-ternary diagram for various ratios (3:1, 2:1, 1:1. 1:2, 1:3) by water titration method as mentioned for co-surfactant selection (section 4.2.5.). Pseudo-ternary diagram involves oleic acid as an oil phase, tween 20 as the surfactant phase, isopropyl alcohol as cosurfactant phase and distilled water as aqueous phase and ratio with maximum microemulsion region was selected for further study.
FIG. 1: FORMULATION OF MICROEMULSION
Formulation of Amoxicillin Loaded Micro-emulsion: Amoxicillin loaded microemulsion was prepared by dissolving 0.4% w/v Amoxicillin to the mixtures of oil, surfactant, and co-surfactant by putting the mixture on vortex mixer and then an appropriate amount of water was added to the mixture drop by drop and the microemulsion containing Amoxicillin was obtained by simultaneously vortexing the mixtures on vortex mixer for 10 min at ambient temperature. All microemulsions were stored at ambient temperature.
Evaluation of Microemulsion:
Appearance: The Amoxicillin loaded micro-emulsions were visually inspected for homogeneity and optical clarity. Samples were analyzed for any precipitation or phase separation in the presence of Amoxicillin. The systems were also checked for the presence of an undissolved drug or another solid ingredient.
Centrifugation: Centrifugation study helps to determine the behavior of small particles in the gravitational field, i.e., their separation rate is quite simple and inexpensive providing a rapid full- proof identification of the system as micro-emulsion. All the batches of drug loaded micro-emulsions were subjected to centrifugation at 3000 rpm for 30 min and then examined for any phase separation.
pH: The pH values of drug loaded microemulsions were determined using digital pH meter, which was previously standardized using pH 4 and 7 standard buffers before use. The study was repeated three times.
Refractive Index: The refractive index which was used to evaluate the transparency of microemulsion and compare the microemulsions with and without Amoxicillin by Abbe’s refractometer calibrated with distilled water. The refractive index was measured by an Abbe by placing 1 drop of sample on the sample holder and measures the reflectance of light by a sample. The study was repeated three times.
Drug Content: Drug content was determined by adding 1 ml drug loaded microemulsion in 10 ml volumetric flask and diluted up to 10 ml. The sample was further diluted appropriately with methanol and analyzed spectrophotometrically at 381.4 nm against methanol as blank. The study was repeated three times.
In-vitro Skin Permeation Study:
Permeation Study: In-vitro skin permeation studies were carried out using Franz diffusion cell. The cell consists of two chambers, the donor and the receptor compartment with a diffusion area of 1.77 cm2. The donor compartment was open at the top and was exposed to the atmosphere. The excised mouse skin was mounted between the compartments of the diffusion cell with stratum corneum facing the donor compartment and clamped into position. The entire setup was placed over magnetic stirrer and stirred at 600 rpm. The receptor solution was filled with phosphate buffer pH 7.4 containing 1.5% v/v tween 20, and the skin surface was covered with 1 ml of the same solution. The cells were equilibrated in a water bath at 37 ± 0.5 ºC for 1 h. After this 2 ml of the microemulsion formulation was applied onto the surface of the skin. 1 ml of medium was collected from receptor compartment at predetermined intervals (0.083, 0.16, 0.25, 0.33, 0.42, 0.50, 0.75, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 24 h) and replaced with the same amount of fresh buffer containing 1.5% v/v tween 80. The amount of permeated drug was measured using UV-Visible spectrophotometer by measuring absorbance at Zmax 379 nm.
Analysis of Permeation Data: The cumulative amount of Amoxicillin permeated per unit area into the receptor compartment was plotted against time. The steady state flux, Jss (mg/cm2/h) was calculated from the slope of the linear portion of the cumulative amount permeated versus time and expressed as:
Jss = dM / dt
Where, M is the cumulative amount of Amoxicillin permeated through skin per unit area in (mg/cm2) in experimental time t (h).
Enhancement ratios (ER) were calculated according to the following expression
ER= The flux of microemulsion based gel/The flux of conventional gel
Drug Retained in Skin: At the end of the permeation experiments (after 24 h), the remaining formulation in the donor phase was scrapped off the skin. The effective diffusion area of the skin was separated, washed several times with distilled water, to remove formulation excess, and then cut into small piece. The receptor media was then replaced with fresh phosphate buffer pH 7.4 containing 1.5 % v/v tween 20. Pieces of skin were put in the receptor chamber and stirred at 600 rpm at 37 °C. Receptor contents were allowed to stir for the next 24 h to ensure effective extraction of the retained drug from the skin, before being subjected to analyzed sonication was done for 30 min. After this, the media was analyzed by UV visible spectrophotometer for the amount of drug retained in the skin.
Formulation of Microemulsion based Gel: Previously reported gelling polymer such as carbopol 940, carbopol 937, xanthan gum and sodium alginate were used to prepare the microemulsion based gel for improving the viscosity of optimized microemulsion. These polymers were slowly mixed with microemulsion under stirring. Xanthan gum and sodium alginate were tried in different concentration (1-4% w/v). 1 % w/v carbopol 937 and carbopol 940 were swelled in microemulsion separately after the uniform swelling system was neutralized by adding triethanolamine and a high viscosity gel was obtained.
Formulation of Conventional Gel of Amoxicillin: Amoxicillin conventional gel was prepared by dispersing 1 g of the carbopol- 940 in sufficient quantity of distilled water. After complete dispersion, carbopol-940 solution was kept in the dark for 24 h for the complete swelling of carbopol-940. Then 300 mg of Amoxicillin was dissolved in a mixture of 10 ml of polyethylene glycol-200 (PEG-200) and 10 ml polyethylene glycol-400 (PEG-400). This solution of the drug was added slowly in the aqueous dispersion of carbopol-940. Then other ingredients like 10 ml of isopropyl alcohol (IPA) and 0.5 ml of triethanolamine were added to get homogeneous dispersion of gel. The remaining quantity of distilled water was added to get 100 g of gel.
Evaluation of Microemulsion based Gel and Conventional gel of Amoxicillin:
Appearance: The microemulsion based gel and conventional gel were visually inspected for homogeneity, precipitation of drug if any particulate matter present and phase separation Samples were analyzed. Homogeneity test was performed by placing hundred milligrams of gel between the thumb and the index finger to notice the consistency of gel that any coarse particles being attached or detached on the finger.
pH: A 10% w/w aqueous solution of microemulsion based gel and conventional gel were prepared, and pH was measured using digital pH meter which was previously standardized using pH 4 and 7 standard buffers before use. The study was repeated three times.
Viscosity: Viscosity of microemulsion based gel and conventional gel were measured using Brookfield Viscometer. The sample (300 g) was placed in a beaker and was allowed to equilibrate for 5 min before measuring the dial reading using a spindle no. 63 at temperature 30 ± 1 ºC. At each speed, the corresponding dial reading on the viscometer was noted. The spindle speed was successively increased to achieve torque 100%, and the corresponding dial reading was noted. The measurements were carried in triplicate at ambient temperature.
Drug Content: Drug content of microemulsion based gel and conventional gel were determined by dissolving accurately weighed 1 gm of gels in few ml of methanol in 100 ml volumetric flask and diluted up to 100 ml with methanol. After the appropriate dilution of the above solution, absorbance was measured by using UV-visible spectrophotometer at 381.4 nm using methanol as blank. The study was repeated three times.
Fourier Transform Infrared Spectroscopy (FTIR): FTIR spectra of pure drug and optimized microemulsion based gel were recorded using Nicolat is 1 FTIR spectrophotometer. Few quantities of microemulsion based gel and Amoxicillin powder was placed on the diamond assembly. The scanning was performed between the range 4000-500 cm-1.
RESULTS AND DISCUSSION:
Pre-formulation Study:
Micromeritics Properties: The data of drug’s Micromeritics properties like bulk density, tapped density, angle of repose, Carr's index, and Hausner's ratio was shown in Table 1. Improper flow is due to particle-particle friction forces which are quantified by the high value of angle of repose and Carr's index. Compressibility is indirectly related to the relative flow rate, cohesiveness, and particle size distribution of powder. The value of carr’s index was found to be 35.821% reflects very poor flow properties of Amoxicillin, which was supported by Hausner's ratio of 1.559.
TABLE 1: RESULTS OF MICROMERITICS PROPERTIES
| Parameter | I | II | III | Average # |
| Bulk density (g/ml) | 0.348 | 0.359 | 0.364 | 0.357 ± 0.008 |
| Tapped density (g/ml) | 0.556 | 0.546 | 0.567 | 0.556 ± 0.010 |
| Carr's Index (%) | 37.410 | 34.249 | 35.802 | 35.821 ± 1.580 |
| Hausner's ratio | 1.597 | 1.520 | 1.557 | 1.559 ± 0.003 |
| Angle of repose | 45.78 | 46.88 | 49.45 | 47.37 ± 1.883 |
# All values are shown in mean ± SD (n=3).
FIG. 2: RESULTS OF MICROMERITICS PROPERTIES
Melting Point: Melting point determination is a simple and fast method to estimate the purity of a substance, small quantity of impurities my change the melting point or at least enlarge melting range. The melting point of Amoxicillin was found to be in the range 227-230 ºC Amoxicillin.
Partition Coefficients: Aqueous solubility and lipophilicity have been shown to influence membrane flux, therapeutic activity, and pharmacokinetics profile of the drug. A partition coefficient of Amoxicillin was found to be 2.008 ± 0.030, which was slightly higher to the reported value.
Screening of Oils: Usually, the oil, which has the maximum solubilizing potential for the selected drug candidates, was selected as an oily phase for the formulation of the microemulsion. This help to achieve the maximum drug loading in the microemulsion. At the same time, the ability of the selected oil to yield a system with larger microemulsion existed area is also important.
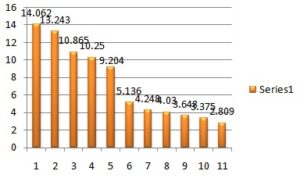
Accordingly, based on maximum solubility (14.062 ± 0.503 mg/ml), oleic acid was selected as an oil phase in the formulation of the microemulsion. The use of oleic acid is advantageous because it increases skin permeability by two mechanistic scenarios of the enhancer; (a) lipid fluidization, and (b) lipid phase separation, oleic acid is a model skin permeation enhancer. It was also reported that oleic acid act as a penetration enhancer for transdermal application so oleic acid was selected as the oil phase for further study. The solubility of Amoxicillin in various oils is presented in Table 2 and Fig. 3.
TABLE 2: SOLUBILITY OF AMOXICILLIN IN VARIOUS OILS
| Oils | Solubility# (mg/ml) |
| Oleic acid | 14.062 ± 0.502 |
| Castor oil | 13.243 ± 0.521 |
| Olive oil | 10.865 ± 0.540 |
| Acconon MC-8 | 10.250 ± 0.291 |
| Maisine 35-1 | 9.204 ± 0.377 |
| Captex 200 P | 5.136 ± 0.289 |
| Capmul PG-8 | 4.248 ± 0.264 |
| Capryol 90 | 4.03 ± 0.266 |
| Triacetin | 3.648 ± 0.308 |
| Labrafac lipophile WC 1349 | 3.375 ± 0.289 |
| IPM | 2.809 ± 0.226 |
#All values are shown in mean ± SD (n=3)
FIG. 3: SOLUBILITY OF AMOXICILLIN IN VARIOUS OILS
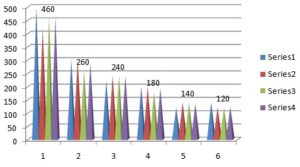
Screening of Surfactant: Nonionic surfactants are generally considered safer than the ionic surfactants and are usually accepted for transdermal application. For the o/w microemulsion, HLB value of surfactant should be 8-18. Different non-ionic surfactants having different HLB were screened based on both, their ability to solubilize the drug and to emulsify the selected oil phase. Solubility in surfactants was determined because the drug also solubilized in the surfactant and should have good solvent properties to allow the presentation of the drug in solution. The emulsification efficiency of surfactants like tween 20, tween 40, tween 60, and tween 80 depends on the chain length. Tween 20 (C-12) has highest emulsification efficiency, which is due to the shortest chain length, the highest hydrophilicity (HLB 16.7), and the greatest ability to incorporate water. Furthermore, tween 20 has the lowest dynamic viscosity (400 cp) compared to that of tween 40 (500 cp) and tween 80 (425 cp).
Selection of surfactants was done by their emulsification efficiency for the selected oil and their ability to solubilize Amoxicillin. Good solubility of Amoxicillin in the surfactant was considered as an additional advantage. Results inferred that oleic acid exhibited the highest emulsification efficiency with tween 20. Table 3 and Fig. 4 represent the solubility of Amoxicillin in various surfactants, and Table 4 represents the emulsification efficiency of surfactants.
TABLE 3: SOLUBILITY OF AMOXICILLIN IN VARIOUS SURFACTANTS
| Surfactants | Solubility# (mg/ml) |
| Tween 20 | 45.510 ± 2.015 |
| Tween 40 | 40.653 ± 2.853 |
| Tween 60 | 34.706 ± 2.529 |
| Tween 80 | 33.618 ± 2.474 |
| Gelucire 50/13 | 20.899 ± 0.856 |
| Gelucire 44/14 | 18.613 ± 0.822 |
# All values are shown in mean ± SD (n=3)
FIG. 4: SOLUBILITY OF AMOXICILLIN IN VARIOUS SURFACTANTS
TABLE 4: EMULSIFICATION STUDY BY SURFACTANTS
| Surfactants | Amount of oil (µl) | The average amount of oil
(µl) |
||
| I | II | III | ||
| Tween 20 | 500 | 420 | 460 | 460 ± 40 |
| Tween 40 | 300 | 300 | 260 | 286.66 ± 23.094 |
| Tween 60 | 220 | 240 | 240 | 233.33 ± 11.547 |
| Tween 80 | 200 | 200 | 180 | 193.33 ± 11.547 |
| Gelucire 50/13 | 120 | 140 | 140 | 133.33 ± 11.547 |
| Gelucire 44/14 | 140 | 120 | 120 | 126.66 ± 11.547 |
All values are shown in mean ± SD (n=3)
Screening of Co-surfactant: The main factor determining the range of formation of the micro-emulsion zone is the physicochemical properties of the oil phase, aqueous phase, and surfactant with some essential conditions required for microemulsion formation. These include the existence of very low surface tension at the oil-water interface, the presence of a highly fluid interfacial film of surfactant and the penetration and association of oil molecules with the interfacial surfactant film.
Introduction of cosurfactants provides a further reduction in the surface tension and fluidizes the interfacial surfactant film, which can increase the area of existence of the microemulsion system. Medium chain length alcohols also increase the mobility of the hydrocarbon tail and also allow greater penetration of the oil into this region.
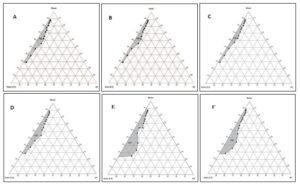
Furthermore, any alcohol present may also influence the solubility properties of the aqueous and oily phases due to its partitioning between these phases. At very low water content maximum oil was solubilized in the surfactant water blend. The amount of incorporated oil was increased progressively with decreasing water content and increasing Smix concentration. It should be noted that within the formed microemulsion zone, the fluidity of the microemulsion reduced with decreasing water content and increasing Smix concentration. Outside the microemulsion zone, especially at high oil and Smix concentration, the system changes into a gel and high viscosity system. Pseudo-ternary diagram of different cosurfactant like PEG 200, PEG 400, PEG 600, propylene glycol, ethanol, butanol, isopropyl alcohol were shown in Fig. 5A-G. For all of these pseudo-ternary diagrams surfactant (tween 20): co-surfactant ratio (Smix) was kept constant at 1:1. The shaded area in these pseudo-ternary diagrams indicates the microemulsion region. Pseudo-ternary diagram of isopropyl alcohol has largest microemulsion area than the other co-surfactant. Shaded area in these pseudo-ternary diagrams of isopropyl alcohol Fig. 5G covered 2-19% v/v oil, 10-58% v/v Smix and 25-88% v/v water.
FIG. 5: PSEUDO TERNARY PHASE DIAGRAM OF MICROEMULSION COMPOSED OF OIL PHASE, SURFACTANT, WATER AND DIFFERENT COSURFACTANT (A) PEG 200 (B) PEG 400, (B) PEG 600 (D) PROPYLENE GLYCOL (E) ETHANOL (F) BUTANOL (G) ISOPROPYL ALCOHOL
Evaluation of Microemulsion:
Appearance: The droplets of the microemulsions being smaller than 1/4th the wavelength of visible light, permit white light to pass through the dispersed system making it transparent or translucent. All the microemulsion formulations were clear, transparent and appeared like a homogeneous single-phase liquid when observed for visual clarity against strong light. After the drug loading, there was not any precipitation of drug and other ingredients. There was not found any traces of undissolved drug or another solid ingredient.
pH: For transdermal formulation, pH is very important in accordance with permeation of drug and skin irritation. Value of pH of microemulsion should be near to skin pH (7.4) to prevent the skin irritation. Value of pH of the microemulsion is not a major issue when incorporated into gelling polymer because gel causes change of microemulsion property after adding gelling polymer. The network structure formed and the increase of the viscosity decreased the contact chances between skins and microemulsion. The values of pH of all micro-emulsions were found to be in the range of 4.62 ± 0.06 to 5.42 ± 0.07.
Refractive Index: The values of the refractive index of all drug loaded microemulsions formulation were found in the range of 1.376 ± 0.001 to 1.405 ± 0.003. The values of the refractive index of all microemulsions formulation without drug were found in the range of 1.374 ± 0.002 to 1.406 ± 0.002. So, it indicated that there were no significant changes in the value of the refractive index of microemulsion containing drug, thus having no interaction between microemulsion components and drug. All values are near to the value of the refractive index of water; it’s indicated that all microemulsion formulations were transparent and clear.
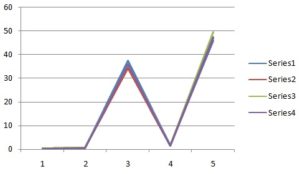
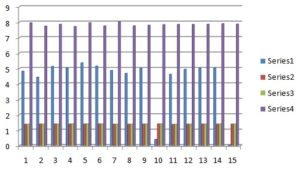
TABLE 5: EVALUATION PARAMETERS OF MICROEMULSION AND MICROEMULSION BASED GEL
| Batch | pH | Refractive Index without drug | Refractive Index with drug | Drug content |
| ME 1 | 4.81 ± 0.05 | 1.383 ± 0.002 | 1.383 ± 0.001 | 7.951 ± 0.176 |
| ME 2 | 4.73 ± 0.04 | 1.390 ± 0.001 | 1.391 ± 0.001 | 7.742 ± 0.202 |
| ME 3 | 5.13 ± 0.05 | 1.393 ± 0.003 | 1.394 ± 0.002 | 7.859 ± 0.153 |
| ME 4 | 5.04 ± 0.05 | 1.397 ± 0.001 | 1.398 ± 0.001 | 7.716 ± 0.192 |
| ME 5 | 5.35 ± 0.09 | 1.398 ± 0.004 | 1.401 ± 0.002 | 7.959 ± 0.115 |
| ME 6 | 5.15 ± 0.03 | 1.405 ± 0.003 | 1.405 ± 0.003 | 7.750 ± 0.239 |
| ME 7 | 4.86 ± 0.07 | 1.381 ± 0.002 | 1.382 ± 0.002 | 8.010 ± 0.206 |
| ME 8 | 4.69 ± 0.06 | 1.387 ± 0.002 | 1.388 ± 0.001 | 7.767 ± 0.258 |
| ME 9 | 5.00 ± 0.08 | 1.397 ± 0.001 | 1.398 ± 0.002 | 7.800 ± 0.167 |
| ME 10 | 5.41 ± 0.08 | 1.403 ± 0.002 | 1.405 ± 0.002 | 7.834 ± 0.133 |
| ME 11 | 4.61 ± 0.05 | 1.373 ± 0.002 | 1.376 ± 0.001 | 7.850 ± 0.151 |
| ME 12 | 4.96 ± 0.07 | 1.387 ± 0.002 | 1.389 ± 0.002 | 7.850 ± 0.126 |
| ME 13 | 5.01 ± 0.06 | 1.391 ± 0.002 | 1.393 ± 0.001 | 7.859 ± 0.165 |
| ME 14 | 5.02 ± 0.06 | 1.393 ± 0.002 | 1.393 ± 0.001 | 7.901 ± 0.129 |
| ME 15 | 5.02 ± 0.08 | 1.393 ± 0.002 | 1.393 ± 0.001 | 7.867 ± 0.129 |
All values are shown in mean ± SD (n=3)
FIG. 6: EVALUATION PARAMETER OF MICRO-EMULSION AND MICROEMULSION BASED GEL
Drug Content: The value of drug content of all the microemulsions formulation were found in the range of 95.463% w/v to 101.15% w/v.
In-vitro Skin Permeation Study: The effects of formulation component (oil, Smix, and water) of all the microemulsion batches on permeation through the skin were studied by in-vitro permeation study on rat skin. The detailed in-vitro skin permeation data and permeation profiles. Permeation profile of all batches showed that the microemulsion formulations were successfully increased permeation of drug increase with the time. ME 7 batch of microemulsion highest permeation flux which contains 5 ml oil, 50 ml Smix and 60 ml water. The permeation coefficient (Km) of ME 7 was 0.04599 ± 0.0012 cm/h.
The permeation of Amoxicillin was increased with a decrease in the amount of oil. Those results might be due to the increased in the size of the globules of internal (oil) phase of microemulsion as increased in the amount of oil. The permeation of Amoxicillin was increased with increased in the amount of Smix, but permeation decreased with further increase in the amount of Smix.
It was reported that oleic acid (oil phase) act as a permeation enhancer, but results indicate that oil increase than permeation decrease so oleic acid act as a permeation enhancer in the lowest amount. Microemulsions contain surfactant and cosurfactant, which also act as a permeation enhancer. From all batch ME 3 had the highest permeability coefficient, which was found to be 0.04658 ± 0.0011 cm/h. For all batches, the permeability coefficient was found to be in the range of 0.04234 ± 0.0008 cm/h to 0.04656 ± 0.0018 cm/h. The apparent diffusion coefficient (Dapp) was found to be in the range of 0.00122 ± 0.01 cm2/h to 0.00336 ± 0.00050 cm2/h. Dapp was inversely proportional to lag time. As increased in the amount of oil lag time also increased, and the apparent diffusion coefficient decreased.
The apparent partition coefficient (Kapp) was found to be in the range of 1.0307 ± 0.169 to 0.00338 ± 2.7775 ± 0.132. The Kapp value was directly proportional to permeability coefficients and thickness of skin barrier and inversely proportional to apparent diffusion coefficients.
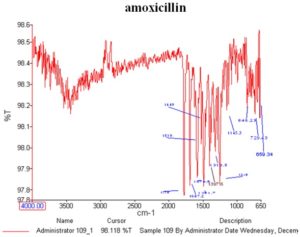
Fourier Transform Infrared Spectroscopy (FTIR): FTIR study was performed for identification and determination of purity of the drug and to check drug excipients compatibility. The FTIR spectrum of Amoxicillin and microemulsion based gel were shown. The characteristic functional group indicating the peak of Amoxicillin and microemulsion based gel were shown in Table 6. The peak value was indicated that there was not any characteristic functional group peak shifted in FTIR spectrum indicated that there was not any significant interaction in microemulsion based gel.
FIG. 7: FTIR SPECTRA OF DRUG
TABLE 6: IR INTERPRETATION OF AMOXICILLIN
| Frequency cm-1 Reference | Sample | Indication
Inferences |
| 1694.9-1639.3 | 1646.0 | C –C stretch (in ring) Aryl systems |
| 1612.4-1585.8 | 15.79.5 | C – C stretch Aryl systems |
| 1639.3-1538.4 | 1519.3 | C – C stretc Aryl systems |
| 1300-1370 | 1339.5 | N – O asymmetric stretch Amide |
| 900 – 675 | 846.23, 729.3, 846 | C – H Aromatics |
Stability Study: The stability of microemulsion based gel was carried out at 40 ± 2 ºC and 75 ± 5% RH and microemulsion based gel were evaluated via appearance, pH, spreadability, extrudability, viscosity, drug content, and in-vitro skin permeation.
TABLE 7: STABILITY STUDY DATA OF MICRO-EMULSION BASED GEL
| Parameters | Initial | After 1 month |
| pH | 6.886 ± 0.085 | 6.813 ± 0.062 |
| Spreadability (gm.cm/sec) | 46.64 ± 3.137 | 47.67 ± 2.043 |
| Extrudability (gm/cm2) | 14.97 ± 0.818 | 14.72 ± 0.70 |
| Drug content (mg) | 7.926 ± 0.066 | 7.926 ± 0.063 |
| Viscosity (cP) | 8069.67 ± 97.127 | 8026.66 ± 93.05 |
| Q24 (mg/cm2) | 3.9440 ± 0.037 | 3.9402 ± 0.035 |
| Flux (mg/cm2/h) | 0.1714 ± 0.003 | 0.1715 ± 0.0015 |
| Lag time (h) | 0.446 ± 0.046 | 0.446 ± 0.043 |
All values are shown in mean ± SD (n=3).
The results suggested that the formulation did not show any significant changes result in data. Table 7; Stability study data of microemulsion based gel
CONCLUSION: Transdermal drug delivery system is viable administration route because of its convenience, lowers total daily dose, avoidance of the first-pass metabolism, avoidance of gastrointestinal side effects, high level of patient compliance and many more. The present investigation was aimed to developed microemulsion based gel of Amoxicillin for transdermal administration. Amoxicillin is mainly prescribed for rheumatoid arthritis and Osteoarthritis in which long term treatment is required. Long term use causes serious gastric side effect. The main interest in such dosage form was to alter the route of drug administration so that gastrointestinal side effect could be avoided.
Skin irritation study of microemulsion based gel was carried out on albino Wistar rats. The primary irritation index (PII) was found to be 0.25 for optimized microemulsion based gel, indicating that formulation was non-irritating and safe for dermal application. FTIR spectroscopy of pure drug and microemulsion based gel was performed, results of that indicated not any changes shown in the characteristic functional group peak in the FTIR spectrum. So there was no interaction observed between drug and excipients.
Stability of microemulsion based gel was subjected to stability study as per ICH guidelines. The result suggested that the formulation did not shows any significant change in appearance, pH, spreadability, extrudability, viscosity, drug content, and in-vitro skin permeation after one month, indicating formulation is stable.
Hence, it can be concluded that with the help of microemulsion based gel, TDDS can provide greater patient compliance by providing easy administration and avoidance of side effect.
ACKNOWLEDGEMENT: The authors thanks to the authorities of Institute of Pharmacy, Bundelkhand University, Jhansi, Uttar Pradesh for providing all the support to study and all other necessary facilities like internet surfing, library and other technical support to write the research article.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Afaq F, Adhami VM, Ahmad N and Mukhtar H: Botanical antioxidants for chemoprevention of photo-carcinogenesis. Front Biosci 2002; 7: 784-92.
- Křena VB and Walterováb D: Silybin and silymarin-new effects and applications. Biomed Papers 2005; 149(1): 29-41.
- Svobodová A, Psotová J and Walterová D: Natural phenolics in the prevention of UV-induced skin damage. A review. Biomed Papers 2003; 147(2): 137-45.
- Katiyar SK: Treatment of silymarin, a plant flavonoid, prevents ultraviolet light-induced immune suppression and oxidative stress in mouse skin. Int J Oncol 2002; 21(6): 1213-22.
- Katiyar SK: Silymarin and skin cancer prevention: anti-inflammatory, antioxidant and immunomodulatory effects (review). Int J Oncol 2005; 26(1): 169-76.
- Paul BK and Moulik SP. Uses and applications of microemulsions. Current Science 2001; 80(8): 990-1001.
- Oricha BS: Cosmeceuticals: A review. African Journal of Pharmacy and Pharmacology 2010; 4(4): 127-129.
- Mandal S and Das S: Design and development of carbamazepine mucoadhesive microemulsion for intranasal delivery: An ex-vivo International Journal of Pharmaceutical Sciences Review and Research 2010; 3(1): 56-60.
How to cite this article:
Rastogi A, Prajapati RN, Prajapati SK, Bijauliya RK, Chanchal DK and Kumar S: Formulation, development and characterisation of microemulsion system of Amoxicillin. Int J Life Sci & Rev 2018; 4(9): 140-50. doi: 10.13040/IJPSR.0975-8232.IJLSR.4(9).140-50.
All © 2015 are reserved by International Journal of Life Sciences and Review. This Journal licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.
Article Information
2
140-150
896
1287
English
IJLSR
A. Rastogi *, R. N. Prajapati, S. K. Prajapati, R. K. Bijauliya, D. K. Chanchal and S. Kumar
Department of Pharmaceutics, Institute of Pharmacy, Bundelkhand University, Jhansi, Uttar Pradesh, India.
rastogiajay500@gmail.com
06 February 2018
20 August 2018
28 August 2018
10.13040/IJPSR.0975-8232.IJLSR.4(9).140-50
01 September 2018