GROWTH AND NON-ENZYMATIC ANTIOXIDATIVE STUDIES IN IN-VITRO GROWN SAFFLOWER (CV A-1) SEEDLINGS UNDER COPPER STRESS
HTML Full TextGROWTH AND NON-ENZYMATIC ANTIOXIDATIVE STUDIES IN IN-VITRO GROWN SAFFLOWER (CV A-1) SEEDLINGS UNDER COPPER STRESS
S. Gautam 1, S. S. Bhagyawant 2 and N. Srivastava * 1
Department of Bioscience and Biotechnology 1, Banasthali University, Banasthali, Jaipur - 304022, Rajasthan, India
SOS in Biotechnology 2, Jiwaji University, Gwalior - 474011, Madhya Pradesh, India.
ABSTRACT: In the present study, we were interested in evaluating the effects of copper on morphological features and non- enzymatic antioxidants in the safflower (cv. A-1). The safflower seeds were grown in Hoagland’s solution supplied with the 0.5 µM (control), 25, 50 and 100 µM CuSO4 for 10th and 20th days. The seedlings were harvested after different days (10th and 20th days). The presence of 50 µM and 100 µM concentration of Cu stimulated impaired root growth after 10th and 20th days, while 25 µM Cu was found to be less in inhibiting the root growth. Also, the shoot growth was found to be adversely affected with increased copper concentration. The malondialdehyde content, nonenzymatic antioxidants (flavonoids and polyphenols), and proline were found to be accumulated in both days harvested leaves of safflower’s seedlings. Thus the conclusion reveals that the increase of copper above threshold point affects the plants in a negative manner.
Keywords: Safflower, Copper stress, Proline, Lipid peroxidation
INTRODUCTION: Safflower (C. tinctorius L.) also called Kusum in Hindi belongs to family Asteraceae 1. It is a multipurpose crop and has a long history of cultivation. India is the largest producer of Safflower in the world 2, producing around 206,000 tonnes of seeds annually 3. Safflower has been mainly grown for the dye which is commonly known as Carthamin until the cheaper dye aniline became available. Safflower is commercially grown for the seeds to produce high-quality edible oil 4.
Now a day this crop has attracted its uses in pharmaceuticals being rich in medicinal properties. It is widely known for its great natural antioxidant properties 5, 6. The heavy metal pollution in the environment affects the normal functioning of the soil ecosystem and plant growth 7. These metals are not degraded and hence persist in the environment for a long time and enter the food chain 8. Copper (Cu) is an essential micronutrient for the growth and development of the plants 9. The increased level of Cu in the environment arises from mining; the uncontrolled use of Cu containing fungicides, pesticides, and bactericides to control pests and diseases 10.
The great concern of our study is that safflower is susceptible to fungal attack, which mainly includes the wilt caused by Fusarium oxysporum f. sp. Carthami 11. So, it is very important to take a control measure which constitutes the treatment of safflower seeds with copper-containing fungicides (e.g., Copper oxychloride). But copper can be detrimental for the growth of plants when applied to the fields above the recommended amounts. The excess concentration of Cu leads to the generation of reactive oxygen species (ROS) via the Haber-Weiss and Fenton reactions 12. These ROS cause damage to plants by the peroxidation of lipids, DNA mutation, and disruption of chlorophyll content 13, 14.
To overcome the effects of ROS, plants possess the antioxidative defense mechanism, which includes enzymatic and non-enzymatic compounds. The enzymatic compounds include Catalase (CAT), superoxide dismutase (SOD), peroxidase (POD) 15, 16, while the non-enzymatic antioxidants includes the ascorbate, carotenoids, glutathione 17, 18, 19. To cope with the abiotic stress, plants produce various antioxidants such as phenolic compounds, flavonoids, proline 20, 21, 22. The objective of the current study was to find out the effect of Cu accumulated with A-1 (Spiny) variety of safflower for its morphological and biochemical response under threshold concentration (Cu stress) in the environment.
MATERIAL AND METHODS:
Plant Material: The seeds of safflower (variety A-1) were obtained from the Directorate of Oilseed Research Institute (ICAR), Hyderabad, A.P. (India).
Plant Culture and Copper Treatment: The experiment was conducted in the June to December 2015 in the Department of Bioscience and Biotechnology, Banasthali University, Banasthali (Rajasthan), India. The seeds were washed with running tap water for 15 min and subjected to surface sterilization with 0.1% Mercuric chloride for 3 min and then rinsed with sterile deionized water (each time for 3 min) for four times under sterilized conditions. The uniform seeds were inoculated in pots containing Hoagland’s medium (pH 6.8), which served as control as well as nutrient solutions supplemented with copper sulphate (CuSO4.5H2O) of various concentrations (25, 50 and 100µM) which served as treatment solutions. Pots were kept for growth of seedlings in a growth chamber at 25 ± 1°C under 60-80% relative humidity and 16 h photoperiod with 40-50 µmol-2s-1 light intensity in a controlled environment. For biochemical and morphological estimations, the seedlings were harvested after 10th and 20th days and washed thoroughly with distilled water. Harvested seedlings were deep freezed and stored at -80°C for further analysis. To minimize the experimental errors, the experiments were carried out at least three times.
Seedlings Growth Analysis: After the 10th and 20th days, the seedlings were removed smoothly from the pots and length of root and shoot length of seedlings were measured using a meter scale. Fresh weight of the seedlings was taken immediately after harvesting of the seedlings. Dry weight of seedlings was determined by placing the samples in hot air oven at 60°C till they dried to constant weight. While the tolerance index (TI%) was calculated as the ratio of dry weight of Cu treated seedlings and controlled seedlings according to the formulae given by 23, 24.
Chlorophyll Content Determination: 0.2 gm leaves were homogenized in 10 ml of 80% acetone. The extract was centrifuged at 5000g for 10 min. The upper phase was transferred into a new tube, and its absorbance was taken at 663, 646, 470 nm (UV-VIS spectrophotometer, Shimadzu, Singapore) respectively with 80% acetone as a blank. The chlorophyll a, b and carotenoid content were measured according to the method of 25, 26.
Total Polyphenol Determination: The total polyphenol content was determined according to the method of the using Folin- Ciocalteu reagent 27. The absorbance of the sample was taken at 725 nm against a blank by spectrophotometer. Gallic acid was used as the standard for preparing the standard curve. The results were expressed as mg of gallic acid equivalent per gram of fresh weight.
Flavonoid Determination: The aluminum chloride method was used for flavonoid determination 28. The absorbance of the sample was taken at 510 nm against a blank by spectrophotometer. Quercetin was used as a standard curve for preparing the standard curve. The results were expressed as mg of quercetin equivalent per gram of fresh weight.
Proline Determination: The accumulation of proline was determined according to the ninhydrin method 29. The absorbance of the sample was monitored at 520 nm by spectrophotometer using toluene as blank. The known concentration of proline was used for preparing the standard curve. The results were expressed as µM of proline per gram of fresh weight.
Lipid Peroxidation Determination: The level of lipid peroxidation in leaves was analyzed by the formation of malondialdehyde (MDA) content after the reaction of TBA (Thiobarbituric acid) 30 and amount was calculated with a coefficient of 155 mM-1 cm-1.
Statistical Analysis: Each value was represented as Mean ± SE from three triplicates. All of the data were subjected to one-way analysis of variance (ANOVA), taking p<0.05, p<0.01 and p<0.001 as significant according to Tukey’s multiple range test to detect the significance using SPSS software (version 16.0).
RESULTS AND DISCUSSION:
Effect of Copper on Growth Parameters: The seedlings of Indian safflower (Carthamus tinctorius L. cv A-1) seeds were used for the current research study to understand their ability to tolerate the excess concentration of Cu through the morphological and biochemical parameters. At the end of the experiment (10th and 20th day), the seedlings were harvested to determine the root, shoot length, fresh, and dry weight. The previous research study also revealed that excess Cu affects the seed germination and growth of seedlings in the presence of Cu 32, 33.
Also, the inhibited growth and reduction in biomass are very common responses to heavy metal toxicity generally observed in higher plants. The decline in biomass may be due to the interference of heavy metal in cell division and elongation 34. Some Scientists 35 also reported that heavy metal affects the ultrastructure of meristematic cells and varying the ribosomal RNA precursor biosynthesis, ultimately affecting the plant growth. It has been proved that the effect of Cu toxicity is principally on root morphology 36.
FIG. 1: (A) AND (B) REPRESENTS THE EFFECT OF COPPER ON MORPHOLOGY OF SAFFLOWER SEEDLINGS HARVESTED AFTER 10th AND 20th DAYS RESPECTIVELY
The Cu tends to accumulate in the root tissue with little translocation to shoots because Cu is sparingly movable in plants 10. Root systems, though genetically determined 37, are very plastic and can be affected by several environmental factors, including metals 38. Our plants did not show any symptoms of chlorosis. Our results have similar findings with the research work 39, in which they observe the stunted root growth and absence of root hairs. Our results are also supported by the findings of 40, 41. According to Fig. 2E, the metal tolerance index was found to be increased by 21% and 41.74% in 10th and 20th days seedlings respectively at 25µM, while it was found to be decreased by (4.8% and 12% ) (9.09% and 20%) at 50 µM and 100 µM respectively in 20th days seedlings respectively. The seed germination was affected in the presence of increased Cu concentration (data not shown). The results showed that shoot length was found to be less affected as compared to the roots both in 10th and 20th days Fig. 2A, B. We evaluated that there was an increase in 1.12% of root length in 25µM while it was found to be significantly reduced by 21% and 47% with increased concentration of Cu (50 µM and 100 µM) as compared to non-treated (100%) safflower seedlings in 10th days harvested seedlings. While in 20th day’s harvested seedlings, root length was found to be significantly reduced (29%, 37%, and 63%) with an increased concentration of Cu (25, 50 and 100 µM), respectively as compared to control. There was a net reduction in the fresh and dry weight Fig. 2C, D which have been used as an indicator of heavy metal toxicity in various studies. The fresh weight was found to be increased by 6% and 10.4% in 25µM at 10th and 20th days harvested seedlings respectively along with the control. It was found to be reduced by 9.6% and 18.8% in the 10th day while 7.4% and 38% in 20th days harvested seedlings concerning control in 50 µM and 100 µM Cu concentration respectively.
FIG. 2: EFFECT OF DIFFERENT CONCENTRATION OF COPPER ON ROOT AND SHOOT LENGTH (A, B), FRESH AND DRY WEIGHT (C, D) AND TOLERANCE INDEX (TI%) (E) ON SAFFLOWER SEEDLINGS COLLECTED AFTER 10TH AND 20TH DAYS OF GROWTH. Values indicates the mean ± SE (n=3). Data with different letters were significant at (*= p<0.05, ** = p<0.01, ***= p<0.001) these levels.
The dry weight was found to be 21.4%, and 43.18% increased at 25 µM Cu concentration along with control at 10th and 20th days harvested seedlings. While it was found to be significantly reduced by 3.57% and 14.2% in 10th days and 9.09% and 20.45% in 20th days harvested seedlings at 50 µM and 100 µM Cu concentration respectively.
Effect of Copper on Chlorophyll Content: All Cu concentrations in the nutrient solutions caused a significant reduction in the chlorophyll a, chlorophyll b, total chlorophyll. The highest reduction in chlorophyll content was observed in 100 µM Cu treated safflower seedlings Table 1.
TABLE 1: THE CONTENT OF PHOTOSYNTHETIC PIGMENTS (CHLOROPHYLL a, b) IN DIFFERENT TREATMENTS OF Cu
| Days | Metal treatment
(µM) |
Chl a
(mg/g FW) |
Chl b
(mg/g FW) |
Total chlorophyll (mg/g FW) |
| 10th day | Control | 0.161±0.010 | 0.066±0.005 | 0.228± 0.015 |
| 25 | 0.147±0.013 | 0.054±0.001 | 0.201± 0.014 | |
| 50 | 0.077±0.001 | 0.039±0.004 | 0.116± 0.005 | |
| 100 | 0.072±0.002 | 0.036±0.001 | 0.109± 0.003 | |
| 20th day | Control | 0.856±0.041 | 0.350±0.025 | 1.206± 0.066 |
| 25 | 0.813±0.017 | 0.232±0.004 | 1.045± 0.021 | |
| 50 | 0.787±0.006 | 0.116±0.020 | 0.904± 0.026 | |
| 100 | 0.763±0.004 | 0.030±0.037 | 0.794± 0.041 |
In plants, chlorophyll content is determined to assess the alteration in pigment content, which is a visual symptom during stress 42, 43. In plant tissues, more than half of Cu is found in the chloroplasts 44. However, the photosynthetic pigments are sensitive to excess Cu. According to various reports, the effect of excess Cu is reported in terms of reduction in chlorophyll pigments in plants 45, 46, 47 A very common symptom to metal toxicity is observed by the inhibition in biosynthesis of chlorophyll 48 which may be due to result of inhibition of the enzymes such as δ-aminolevulinic acid dehydratase (ALA- dehydratase) 49, 8 and protochlorophyllide reductase 50. The other reasons for the decrease in pigment may be the destruction of chloroplast membrane by lipid peroxidation and replacement of Mg2+ with heavy metals in chlorophyll structure 51.
Effect of Copper on Flavonoid and Polyphenol Content: As depicted in Fig. 3A and B, we evaluated the changes in flavonoid and polyphenol content in safflower seedlings after 10th and 20th days. There was an increase in flavonoid content in 10th day (9.99%, 34.7% and 64.8% ) while in 20th days harvested seedling’s leaves (20.4%, 88.2% and 97.8%) with increase in Cu concentration (25, 50 and 100 µM) respectively as compared to control (100%).
Also the polyphenol content, which was found to be abruptly increase in 10th day (12.9%, 49.4%, and 70.7% ) and 20th day (39.2%, 67.5% and 118.6%) harvested seedling’s leaves with increase in Cu concentration (25, 50 and 100 µM) respectively as compared to control (100%).
FIG. 3: EFFECT OF COPPER ON FLAVONOID CONTENT AND POLYPHENOL (A AND B) RESPECTIVELY IN SAFFLOWER LEAVES COLLECTED AFTER 10th AND 20th DAYS OF GROWTH. Values represent mean ±S.E. (n = 3). Data with different letters were significant at (*=p<0.05 **= p<0.01 *** = p<0.001) these levels.
According to the previous studies, it has been suggested that the phenolic compounds are found to generally increase with the response to heavy metals stress. They play a magnificent role in antioxidation by metal chelation and scavenging the ROS 52. These compounds have noteworthy ability to chelate the transition metal ions and the inhibiting the superoxide-driven Fenton reaction 53. It has been reported that the polyphenols have the chelating potential for metals such as Cu, therefore making it unavailable to plants, and this property is beneficial in case of Cu toxicity. Flavonoids are the most widely and distributed group of plant phenolics 54. Flavonoids act as a suppressor of enzyme lipoxygenase, responsible for converting polyunsaturated fatty acids to oxygen-containing derivatives 55-56, thus serving in inhibiting the lipid peroxidation.
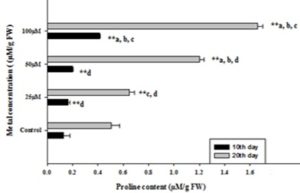
Effect of Copper on Proline Content: Fig. 4 shows a significant gradual increase in the accumulation of proline in both the day's treated safflower leaves. However, at 100 µM Cu concentration, the level of proline content was significantly higher (216%) as compared to other concentrations (25 µM and 50 µM), which were found to be increased by 26% and 52% in 10th-day safflower seedling’s leaves respectively. While in 20th days leaves, it was found to be appreciably elevated with increased concentration of Cu (28.3%, 138.4%, and 229.1%) respectively.
FIG. 4: EFFECT OF COPPER ON PROLINE CONTENT IN SAFFLOWER LEAVES COLLECTED AFTER 10th AND 20th DAYS OF GROWTH. Values represent mean ±S.E (n = 3). Data with different letters were significant at (**= p<0.01) these levels.
Proline is an amino acid which is found to be involved in protecting the plants from stress by absorption of OH radical. Proline has a remarkable affinity to form a complex with cupric ions, which is cooperative in reducing the Cu toxicity. In response to abiotic stress, the proline is accumulated due to increasing its denovo synthesis or decrease degradation 57. Proline helps in the survival of plants by acts as osmoregulant, protection of enzymes from denaturation 58, thus protecting the plant from stress 59 and stabilization of protein synthesis 60. In our study, the accumulation of proline in all treatments depicts that proline is involved in the detoxification of ROS. Our results are in agreement with the other researcher’s results 61, 62.
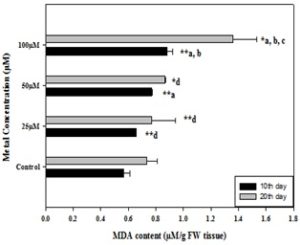
Effect of Copper on Lipid Peroxidation: The exposure of different concentration of Cu (25, 50 and 100µM), which leads to about 16%, 36% and 56% increase in MDA content in 10th days harvested leaves while in 20th days harvested seedling’s leaves it was found to be increased by 4%, 18% and 84% as compared to control (100%) respectively Fig. 5.
FIG. 5: EFFECT OF DIFFERENT CONCENTRATION OF COPPER ON THE LEVEL OF MDA CONTENT IN SAFFLOWER LEAVES COLLECTED AFTER 10th AND 20th DAYS OF GROWTH. Values represent mean ±S.E (n = 3). Data with different letters were significant at (*= p<0.05, **= p<0.01) these levels.
Due to the generation of free radicals or enzymatic activities, the process of lipid peroxidation occurs 63. Due to these reactions, mainly free poly-unsaturated fatty acids (linoleic and linolenic acids) are oxidized 64. These free radicals also cause damage to free and membrane-bound fatty acid, which ultimately leads to the degradation of membranes. Malondialdehyde (MDA) which is a product of lipid peroxidation 65, can be considered as an indicator of heavy metal stress. Therefore the production of MDA and other aldehydes during the Cu toxicity is a very common process. It is cleared that different Cu concentration induces the changes in the level of MDA content in both the day's treated leaves of safflower seedlings. The present study is supported by the other researchers who have observed an increased MDA content under Cu stress 66, 67.
CONCLUSION: Since safflower is highly susceptible to fungal infection, mainly Fusarium sp. Carthami, therefore, the usage of Cu containing fertilizers, pesticides, and fungicides has abruptly increased in the field. With our study, it can be concluded that the C. tinctorius cv. A-1 is sensitive to Cu toxicity. There was an adverse effect on the morphology of seedlings with an increased concentration of Cu due to increase in Cu toxicity, which leads to the production of ROS. The oxidative damage caused by the ROS generated due to excess concentration of Cu is evidenced by the increase of the level of malondialdehyde. However, in our research study, various non-enzymatic components were found to be involved in antioxidative defense mechanism. Proline was found to be accumulated, which is believed to play a significant role in tolerance during abiotic stress by scavenging the ROS. Flavonoid and polyphenols were accumulated to mitigate the harmful effects of ROS and helps the treated safflower seedlings to survive by keeping the detrimental reaction to the minimum extent in the medium.
ACKNOWLEDGEMENT: The authors are thankful to Banasthali University, Banasthali (Raj), for providing the facilities for conducting the research work in the department. Also, the authors are also thankful to Dr. Anjani Kaemmli, Principal Scientist, Directorate of Oilseeds Research Institute, Hyderabad for providing the safflower seeds to carry out the research study.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Al-Chami Z, Amer N, Al Bitar L and Cavoski I: Potential use of Sorghum bicolor and Carthamus tinctorius in phytoremediation of nickel, lead and zinc. Int J Environ Sci Technol 2015; 12: 3957-3970.
- Gautam S, Bhagyawant SS and Srivastava N: Detailed study on therapeutic properties, uses and pharmacological applications of safflower (Carthamus tinctorius). Int J Ayur Pharm Res 2014; 2(3): 1-12.
- Gautam, S, Anjani K and Srivastava N: In-vitro evaluation of excess copper affecting seedlings and their biochemical characteristics in Carthamus tinctorius (variety PBNS-12). Physiol Mol Biol Plants 2016; 1-9.
- Amini F, Saeid G and Arzani A: Study of genetic diversity in safflower genotypes using agro-morphological traits and RAPD markers. Euphytica 2008; 163: 21-30.
- Dwiedi SL, Upadhyaya HD and Hegde DM: Development of core collection in safflower (Carthamus tinctorius) germplasm. Genet Resour Crop Evol 2005; 52: 821-830.
- Golkar P, Arzani A, Rezaei, AM, Yarali Z and Yousefi M: Genetic variation of leaf antioxidants and chlorophyll content in safflower. Afr J Agri Res 2009; 4(12): 1475-82.
- Alia N, Sardar K, Said M, Salma K, Sadia A, Sadaf S, Toqeer A and Miklas S: Toxicity and Bioaccumulation of heavy metals in spinach (Spinacia oleracea) grown in a controlled environment. Int J Environ Res Public Health 2015; 12: 7400-7416.
- Rastgoo L and Alemzadeh A: Biochemical responses of Gouan (Aeluropus littoralis) to heavy metals stress. Aust J Crop Sci 2011; 5(4):375-383.
- Ali S, Shahbaz M, Shahzad AN, Khan HAA, Anees M, Haider MS and Fatima F: Impact of copper toxicity on stone-head cabbage (Brassica oleracea capitata) in hydroponics. Peer J 2015; 3: e1119.
- Adrees M, Ali S, Rizwan M, Ibrahim M, Abbas F, Farid M, Zia-ur-Rehman M, Irshad MK and Bharwana SA: The effect of excess copper on growth and physiology of important food crops: a review. Environ Sci Pollut Res 2015; 22: 8148-8162.
- Nimbkar N: Issues in safflower production in India. 7th International Safflower Conference Held on 3-6 Nov 2008.
- Li S, Zhang G, Gao W, Zhao X, Deng C and Lu L: Plant growth, development and change in gsh level in safflower (Carthamus tinctorius) exposed to copper and lead. Arch Biol Sci Belgrade 2015; 67(2): 385-396.
- Ruley AT, Sharma NC and Sahi SV: Antioxidant defense in a lead accumulating plant, Sesbania drummondii. Plant Physiol Biochem 2004; 42: 899-906.
- Semane B, Dupae J, Cuypers A, Noben JP, Tuomainen M, Tervahauta A, Kärenlampi S, Belleghem FV, Smeets K and Vangronsveld J: Leaf proteome responses of Arabidopsis thaliana exposed to mild cadmium stress. Plant Physiol 2010; 167: 247-254.
- Yang Y, Zhang Y, Wei X, You J, Wang W, Lu J and Shi R: Comparative antioxidative responses and proline metabolism in two wheat cultivars under short term lead stress. Ecotoxicol Environ Saf 2011; 74: 733-740.
- Saha D, Mandal S and Saha A: Copper-induced oxidative stress in tea (Camellia sinensis) leaves. J Environ Biol 2012; 33: 861-866.
- Hall JL: Cellular mechanisms for heavy metal detoxification and tolerance. J Exp Bot 2002; 53: 1-11.
- Shao HB, Chu LY, Lu ZH and Kang CM: Primary antioxidant free radical scavenging and redox signaling pathways in higher plant cells. Int J Biol Sci 2008; 4: 8-14.
- Sharma A and Singh G: Studies on the effect of Cu (II) ions on the antioxidant enzymes in chickpea ( arietinum L) cultivars. J Stress. Physiol Biochem 2013; 9(1): 5-13.
- Sroka Z and Cisowski W: Hydrogen peroxide scavenging, the antioxidant and anti-radical activity of some phenolic acids. Food Chem Toxicol 2003; 41:753-758.
- Górecka K, Cvikrová M, Kowalska U, Eder J, Szafrańska K, Górecki R and Janas KM: The impact of Cu treatment on phenolic and polyamine levels in plant material regenerated from embryos obtained in anther culture of carrot. Plant Physiol Biochem 2007; 45: 54-61.
- Kowalska U, Szafrańska K, Krzyżanowska D, Kiszczak W, Górecki R, Janas K and Górecka K: Effect of increased copper ion content in the medium on the regeneration of androgenetic embryos of carrot (Daucus carota). Acta Agrobot 2012; 65(2): 73-82.
- Balint AF, Kovács G and Sutka J: Copper tolerance of Aegilops, Triticum, Secale and triticale seedlings and copper and iron contents in their shoots. Acta Biol Szegediensis 2002; 46: 77-78.
- Azooz MM, Abou-Elhamd MF and Al-Fredan MA: Biphasic effect of copper on growth, proline, lipid peroxidation and antioxidant enzyme activities of wheat (Triticum aestivum Hasaawi) at the early growing stage. Aust J Crop Sci 2012; 6(4): 688-694.
- Lichtenthaler HK: Chlorophylls and carotenoids: Pigments of photosynthetic biomembranes. Methods Enzymol 1987 148: 350-382.
- Aly AA and Mohamed AA: The impact of copper ion on growth, thiol compounds and lipid peroxidation in two maize cultivars (Zea mays) grown in-vitro. Aust J Crop Sci 2012; 6(3): 541-549.
- Singleton VL and Rossi JA: Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J Enol Viticul 1965; 16(3): 144-158.
- Chang C, Yang M, Wen H and Chern J: Estimation of total flavonoid content in propolis by two complementary colorimetric methods. J Food Drug Anal 2002; 10: 178-182
- Bates LS, Waldren RP and Teare ID: Rapid determination of free proline for water-stress studies. Plant Soil 1973; 39: 205-207.
- Heath RL and Packer L: Photoperoxidation in isolated chloroplasts. Arch Biochem Biophy 1968; 125(1): 189-98.
- Li X, Ma H, Jia P, Wang J, Jia L, Zhang T, Yang Y, Chen H and Wei X: Response of seedling growth and antioxidant activity to excess iron and copper in Triticum aestivum Ecotoxicol Environ Safe 2012; 86: 47-53.
- Nenova V: Effect of iron supply on growth and photosystem II efficiency of pea plants. Gen Appl Plant Physiol 2006; 81-90.
- Posmyk MM and Kontek R: Antioxidant enzymes activity and phenolic compounds content in red cabbage seedlings exposed to copper stress. Ecotoxicol Environ Saf 2009 72: 596-602.
- Khatun S, Ali MB, Hahn EJ and Paek KY: Copper toxicity in Withania somnifera: Growth and antioxidant enzymes responses of in-vitro grown plants. Environ Exp Bot 2008; 64: 279-285.
- Ouzounidou G, Elefteriou EP and Karataglis S: Ecophysiological and ultrastructural effects of copper in ochroleucum (Cruciferae). Can J Bot 1992; 70: 947-957.
- Sheldon AR and Menzies NW: The effect of copper toxicity on the growth and root morphology of Rhodes grass (Chloris gayana) in resin buffered solution culture. Plant Soil 2005; 278:341–349
- Berta G, Trotta A, Fusconi A, Hooker JE, Munro M, Atkinson D, Giovannetti M, Morini S, Fortuna P, Tisserant B, Gianinazzi- Pearson V and Gianinazzi S: Arbuscular mycorrhizal induced changes to plant growth and root system morphology in Prunus cerasifera. Tree Physiol 1995; 15: 281-293.
- Chiatante D, Di Iorio A and Scippa GS: Root responses of Quercus ilex seedlings to drought and fire. Plant Biosystems 2005; 138: 124-139.
- Elisa B, Marsano F, Cavaletto M and Berta G: Copper stress in Cannabis sativa roots: a morphological and proteomic analysis. Caryologia 2007; 60(1-2): 96-101.
- Bouazizi H, Jouili H, Geitmann A and Ferjani EEI: Copper toxicity in expanding leaves of Phaseolus vulgaris: antioxidant enzyme response and nutrient element uptake. Ecotox Environ Safe 2010; 73: 1304-1308.
- Manivasagaperumal R, Vijayarengan P, Balamurugan S and Thiyagarajan G: Effect of copper on growth, dry matter yield and nutrient content of Vigna radiata (L.) Wilczek. J Phytol 2011; 3(3): 53-62.
- Parekh D, Puranlk RM and Srivastava HS: Inhibition of chlorophyll biosynthesis by cadmium in greening maize leaf segments. Biochem Physiol Pfl 1990; 186: 239-242.
- El-Beltagi HS and Mohamed HI: Reactive oxygen species, lipid peroxidation and antioxidative defense mechanism. Not Bot Hort Agrobot Cluj 2013; 41(1): 44-57.
- Hansch R and Mendel RR: Physiological functions of mineral micronutrients (Cu, Zn, Mn, Fe, Ni, Mo, B, Cl). Curr Opin Plant Biol 2009; 12: 259-266.
- Shakya K, Chettri MK and Sawidis T: Impact of heavy metals (copper, zinc, and lead) on the chlorophyll content of some mosses. Arch Environ Contam Toxicol 2008; 54: 412-421.
- Xing W and Huang W: Effect of excess iron and copper on physiology of aquatic plant Spirodela polyrrhiza (L.) Schleid. Environ Toxicol 2010; 25: 103-112.
- Brahim L and Mohamed M: Effects of copper stress on antioxidative enzymes, chlorophyll and protein content in Atriplex halimus. Afr J Biotechnol 2010; 10: 10143-10148.
- Sandalio LM, Dalurzo HC, Gomez M, Romero-Puertas MC and Del Rio LA: Cadmium-induced changes in the growth and oxidative metabolism of pea plants. J Exp Bot 2001; 52: 2115-2126.
- Padmaja K, Prasad DDK and Prasad ARK: Inhibition of chlorophyll synthesis in Phaseolus vulgaris seedlings by cadmium acetate. Photosynthetica 1990; 24: 399-405.
- Van Assche F and Clijsters H: Effects of meals on enzyme activity in plants. Plant Cell Environ 1990; 13: 195-206.
- Kupper H, Kupper F and Spiller M: Environmental relevance of heavy metal-substituted chlorophylls using the example of water plants. J Exp Bot 1996; 47: 259-266.
- Lavid N, Schwartz A, Yarden O and Tel-Or E: The involvement of polyphenols and peroxidase activities in heavy metal accumulation by epidermal glands of the waterlily (Nymphaeaceae). Planta 2001; 212(3): 323-331.
- Arora A, Nair MG and Strasburg GM: Structure-activity relationships for antioxidant activities of a series of flavonoids in a liposomal system. Free Radic Biol Med 1998; 24: 1355-1363.
- Bravo L: Polyphenols: chemistry, dietary sources, metabolism, and nutritional significance. Nutr Rev 1998; 56(11): 317-333.
- Nijveldt RJ, Nood EV, Hoom DEV, Boelens PG, Norren KV and Leeuwen PAV: Flavonoids: a review of probable mechanisms of action and potential applications. Am J Clin Nutr 2001; 201(74): 418-425.
- Ahmad P, Jaleel CA, Azooz MM and Nabi G: Generation of ROS and non-enzymatic antioxidants during abiotic stress in plants. Bot Res Int 2009; 2(1): 11-20.
- Kaur G and Asthir B: Proline: a key player in plant abiotic stress tolerance. Biol Plantarum 2015; 1-11.
- Gajewska E and Skłodowska M: Differential biochemical responses of wheat shoots and roots to nickel stress: antioxidative reactions and proline accumulation. Plant Growth Regul 2008; 54: 179-188.
- Farago ME and Mullen WA: Plants which accumulate metals. Part IV. A possible copper-proline complex from the roots of Armeria maritima. Inorg Chim Acta 1979; 32: 93-94.
- Kuznetsov VV and Shevyakova NI: Stress responses of tobacco cells to high temperature and salinity. Proline accumulation and phosphorylation of polypeptides. Physiol Plant 1997; 100: 320-326.
- Chen CT, Chen LM, Lin CC and Kao CH: Regulation of proline accumulation in detached rice leaves exposed to excess copper. Plant Sci 2001; 160: 283-290.
- Zengin FK and Kirbag S: Effects of copper on chlorophyll, proline, protein and an abscisic acid level of sunflower (Helianthus annuus) seedlings. J Environ Biol 2007; 28(3): 561-566.
- Shewfelt RL and Purvis AC: Toward a comprehensive model for lipid peroxidation in plant tissue disorders. Hort Sci 1995; 30: 213-218.
- Gardner HW: Biological roles and biochemistry of the lipoxygenase pathway. Hort Sci 1995; 30: 197-204.
- Koca H, Ozdemir F and Turkan I: Effect of salt stress on chlorophyll fluorescence, lipid peroxidation, superoxide dismutase and peroxidize activities of cultivated tomato ( esculentum) and its wild relative (L. pennellii). Environ Exp Bot 2007; 60: 344-351.
- Sinha S and Basant A: Iron-induced oxidative stress in a macrophyte: a chemometric approach. Ecotoxicol Environ Saf 2009; 72: 585-595.
- Zhao S and Liu Q: Responses of root growth and protective enzymes to copper stress in turfgrass. Acta Biol Cracov Ser Bot 2010; 52: 7-11.
How to cite this article:
Gautam S, Bhagyawant SS and Srivastava N: Growth and non-enzymatic antioxidative studies of in-vitro grown safflower (C.V A-1) seedlings under copper stress. Int J Life Sci & Rev 2016; 2(3): 52-60. doi: 10.13040/IJPSR.0975-8232.IJLSR.2(3).52-60.
All © 2015 are reserved by International Journal of Life Sciences and Review. This Journal licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.
Article Information
3
52-60
730
1701
English
IJLSR
S. Gautam, S. S. Bhagyawant and N. Srivastava *
Department of Bioscience and Biotechnology, Banasthali University, Banasthali, Jaipur, Rajasthan, India.
nidhiscientist@gmail.com
08 February 2016
10 March 2016
18 March 2016
10.13040/IJPSR.0975-8232.IJLSR.2(3).52-60
31 March 2016