IN VITRO ANTI HAEMOLYTIC, ANTI BACTERIAL AND PHYTO CONSTITUENTS INVESTIGATION OF ANDROGRAPHIS PANICULATA METHANOLIC LEAF EXTRACT
HTML Full TextIN-VITRO ANTI-HAEMOLYTIC, ANTI-BACTERIAL AND PHYTO-CONSTITUENTS INVESTIGATION OF ANDROGRAPHIS PANICULATA METHANOLIC LEAF EXTRACT
Venkata Narasimha Kadali * 1, Tadi Ramesh 2, Sudhakara Rao Pola 1 and B. V. Sandeep 1
Department of Biotechnology 1, Andhra University, Visakhapatnam - 530003, Andhra Pradesh, India.
Department of Biotechnology 2, S V K P & Dr. K.S. Raju Arts and Science College, Penugonda - 534320, Andhra Pradesh, India.
ABSTRACT: The bitter plant Andrographis paniculata has been selected to evaluate the anti haemolytic activity, antibacterial activity, and various phytochemicals present in the methanolic leaf extract. The leaf extract of Andrographis paniculata shown to have significant anti-haemolytic activity. The extract showed anti-haemolytic activity in the range from 42% to 80%. At varying concentration of plant extracts 20, 40, 60, 80 and 100 mg/ml the percent of inhibition of haemolysis recorded were 42%, 56%, 62%, 70%, 80% respectively. The methanolic leaf extract of Andrographis paniculata showed significant inhibitory effect on all of the five bacterial species such as Pseudomonas aeruginosa, Staphylococcus aureus, Escherichia coli, Bacillus subtilis, and Streptococcus mutans. The inhibition zones for S. aureus were 12 mm, 14 mm and 19 mm (including well 4 mm) at concentrations of 15 µl, 20 µl, and 25 µl respectively. The inhibition zones P. aeruginosa were 11 mm, 13 mm and 20 mm, 8 mm, 10 mm and 15 mm for Streptococcus mutans. The zones for E. coli were 15 mm, 16 mm and 18 mm and 12 mm, 13 mm and 19 mm for Bacillus subtilis. The phytochemical investigation of methanolic extract of Andrographis paniculata leaves showed the presence of carbohydrates, flavonoids, alkaloids, tannins, phenols, and saponins.
Keywords: Andrographis paniculata, Phytochemicals, Anti-haemolytic, Anti-bacterial
INTRODUCTION: Despite the remarkable progress in the preparation of synthetic drugs, over 25% of prescribed medicines in industrialized countries are derived directly from plants 1. The World Health Organization (WHO) also considered phytotherapy in its health programs and suggested basic procedures for validation of drugs from plant origin in developing countries 2.
Indigenous plants are reservoirs of various metabolites and provide an unlimited source of important chemicals that have diverse biological properties 3. Medicinal plants are gaining a lot of importance nowadays because of efficacy they have been showing in the traditional healing 4. Herbs are the source of magnificent inhibitors that could act on a wide variety of diseases. One of the great aspects of herbs is they show 100% results when it comes to the healing. Herbs have all sorts of answers against various diseases 5.
The best source of drugs without lethal effects to human systems could be the plant source, and this has been proved by the Traditional healing system and the recent studies conducted on the experimental animals 6. Even expansion of modern medicines throughout the India people in rural areas still uses this wonder herbal medications for various sorts of diseases 7. The use of medicinal plants in developing countries is increasing, which offer a new source of antibacterial, antifungal, and antioxidants agents 8. The development of drug resistance, as well as the appearance of undesirable side effects of certain drugs, has led to the search of new antimicrobial agents in particular from medicinal plants 8. This medicinal herb A. paniculata otherwise called as king of bitters because it has extremely bitter where it is used to treat various infections and diseases 9.
Occurs quite commonly in the entire India subcontinent, which accounts for its widespread use since ancient times against a variety of disorders, in both Ayurvedic and Chinese Medicine 10. It has blood purifying property, so it is recommended for use in leprosy, gonorrhea, scabies, boils, skin eruptions, and chronic and seasonal fevers 9. The leaf extract of Andrographis paniculata was used to evaluate the enzyme inhibiting the activity of protease, phosphomono-esterase, phosphodiesterase, acetylcholinesterase, phospholipase A2, hyaluronidase and L-amino acid oxidase toxic enzymes present in snake venom 11.
The purified active compounds through TLC and crude plant extract were subjected to analyze the anti-inflammatory activity and shows potent inhibitory activity against the adverse effects of the inflammation such as tissue damage and protein denaturation 10. A. paniculata shows excellent anticancer activities against different cancer cell lines; it is alternatives medicines for cancer would replace side effect causing chemotherapeutic agent12.
Andrographolide active component of A. paniculata shown analgesic, antipyretic, and anti-ulcerogenic effect 13. Anti-hyperglycemic effect in streptozotocin-induced diabetic rats 14, it has anti-tumor activity via activation of cytotoxic T lymphocyte and attenuation of tumor growth in- vivo 15. A. paniculata is known to possess anti-HIV 16, cardioprotective 17 and hepatoprotective 18, properties. In this present study, the bitter plant Andrographis paniculata has been selected to evaluate the anti haemolytic activity, antibacterial activity, and various phytochemicals present in the methanolic leaf extract.
MATERIALS AND METHODS:
Materials: Leaves of Andrographis paniculata were collected from the campus S V K P & Dr. K S Raju Arts and Science College, Penugonda. The plant was authenticated by Dr. Suryanayana Raju, Department of Botany, S V K P & Dr. K S Raju Arts and Science College, Penugonda. Leaves were dried in sunlight for a week and then powdered using a blender to get a coarse powder. Chicken blood was collected from the local market in EDTA bottle.
Test Microorganisms: The methanolic leaf extract of Andrographis paniculata was tested against five pathogenic bacteria. The test organisms include Pseudomonas aeruginosa, Staphylococcus aureus, Escherichia coli, Bacillus subtilis, and Streptococcus mutans.
Extraction Process: Preparation of extract was done according to the procedure done by Sharmila et al., (2011) 19. 25g of leaf powder was packed in a Soxhlet extraction unit and exhaustively extracted using 100 ml of methanol at 60 ºC for 12 h. The extract was completely dried in a water bath at 40 ºC and subsequently stored at 4 ºC.
Phyto-chemical Analysis: A preliminary phyto-chemical investigation was conducted for the detection of steroids, terpenoids, flavonoids, saponins, tannins, carbohydrates, and phenols by using standard procedures 20, 21.
Determination of Antibacterial Activity: Antibacterial activity was measured by well diffusion method 22. Nutrient agar (Hi-media) was prepared and poured into the Petri plates. After solidification of media, overnight bacterial cultures were inoculated on the surface of media. By using a sterile gel puncher, 4 mm of wells were made in each Petri plates.
Then 15 µl, 20 µl, 25 µl of methanolic leaf extract Andrographis paniculata were added into the three wells respectively. The plates were incubated in the incubator at 37°C for optimum bacterial growth. In the next day, the diameter of the zone of inhibition was measured.
Assessment of Anti-haemolytic Activity: Anti-haemolytic activity against H2O2 induced haemolysis in chicken RBC determined by in-vitro method described by Tavazzi et al., 2001, Thagriki Dluya et al., 2015 23, 24. The chicken erythrocytes were separated by centrifugation at 200 rpm and washed with saline or isotonic sodium phosphate buffer (pH 7.4) until the supernatant is colorless. The erythrocytes were then diluted with saline or phosphate buffer to give a 4% suspension. Varying amounts of the plant extracts (20, 40, 60, 80 and 100 mg /ml) with saline or buffer was added to 2 ml of the suspension of erythrocytes, and the volume was made up to 3.5 ml with saline or buffer. This mixture was pre-incubated for 120 min and then 0.5 ml H2O2 solutions of appropriate concentration in saline or buffer was added. The concentration of H2O2 in the reaction mixture was adjusted to bring 90% haemolysis of blood cells after 120 min incubation. After 120 min of incubation, tubes were centrifuged, and the amount of haemolysis was determined by measurement of the absorbance at 540 nm corresponding to haemoglobin liberation. The anti-haemolytic activity was expressed as the inhibition percentage and was calculated using the following formula:
Anti-haemolytic activity (%) = Control 540 nm – Sample 540 nm / Control 540 nm × 100
Where, Sample 540 nm was the absorbance of the sample and Control 540 nm was the absorbance of the control.
RESULTS:
Phytochemical Analysis: The phytochemical investigation of methanolic extract of Andrographis paniculata leaves showed the presence of carbohydrates, flavonoids, alkaloids, tannins, phenols, and saponins Table 1.
TABLE 1: PRESENCE OF PHYTOCHEMICALS IN METHANOLIC EXTRACT OF ANDROGRAPHIS PANICULATA LEAVES
| S. no. | Phytoconstituent | Presence of phytochemical in methanolic extract |
| 1 | Carbohydrates | + |
| 2 | Flavonoids | + |
| 3 | Alkaloids | + |
| 4 | Tannins | + |
| 5 | Phenols | + |
| 6 | Proteins | - |
| 7 | Saponins | + |
| 8 | Sterols | - |
+ indicates presence; - indicates absence.
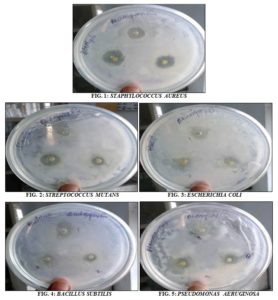
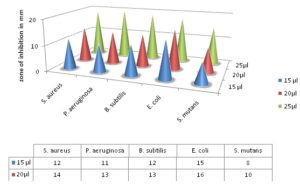
Antibacterial Activity: The methanolic leaf extract of Andrographis paniculata showed significant inhibitory effect on all of the five bacterial species such as Pseudomonas aeruginosa, Staphylococcus aureus, Escherichia coli, Bacillus subtilis, and Streptococcus mutans. The inhibition zones for S. aureus were 12 mm, 14 mm and 19 mm (including well 4 mm) at concentrations of 15 µl, 20 µl, and 25 µl respectively. The inhibition zones P. aeruginosa were 11 mm, 13 mm and 20 mm, 8 mm, 10 mm and 15 mm for Streptococcus mutans. The zones for E. coli were 15 mm, 16 mm and 18 mm and 12 mm, 13 mm and 19 mm for Bacillus subtilis. The antibacterial results of methanolic extract were given below Table 2 and Fig. 1-6.
TABLE 2: INHIBITION ZONES OF METHANOLIC EXTRACT OF ANDROGRAPHIS PANICULATA AGAINST TEST PATHOGENIC BACTERIA
| Test bacterial species | Inhibition zones in mm (including well 4 mm) | |||
| 15 µl (methanolic extract) | 20 µl (methanolic extract) | 25 µl (methanolic extract) | ||
| Staphylococcus aureus | 12 mm | 14 mm | 19 mm | |
| Pseudomonas aeruginosa | 11 mm | 13 mm | 20 mm | |
| Bacillus subtilis | 12 mm | 13 mm | 19mm | |
| Escherichia coli | 15 mm | 16 mm | 18 mm | |
| Streptococcus mutans | 8 mm | 10 mm | 15 mm | |
FIG. 6: ZONES OF INHIBITION OF METHANOLIC EXTRACT ON DIFFERENT BACTERIAL SPECIES
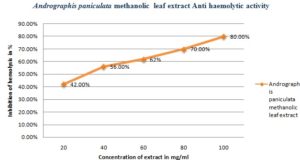
Anti-haemolytic Activity: The methanolic leaf extract of Andrographis paniculata shown to have significant anti-haemolytic activity. The methanolic extract showed anti-haemolytic activity in the range from 42% to 80%. At varying concentration of plant extracts 20, 40, 60, 80 and 100 mg/ml the percent of inhibition of haemolysis recorded were 42%, 56%, 62%, 70%, 80% respectively Fig. 7.
FIG. 7: ANTI-HAEMOLYTIC ACTIVITY OF METHANOLIC LEAF EXTRACT OF ANDROGRAPHIS PANICULATA
DISCUSSION: The preliminary phytochemical investigation of methanolic extract of Andrographis paniculata leaves showed the presence of carbohydrates, flavonoids, alkaloids, Tannins, phenols, and saponins. The methanolic leaf extract of Andrographis paniculata shown to have significant anti-haemolytic activity. In this study, the inhibition of haemolysis found to be increased with increase in the concentration of extract. When red blood cells were treated with H2O2 (toxicant), % haemolysis was found to be increased. This may be because of the oxidizing nature of H2O2 concerning cell membrane degradation and release of haemoglobin from the cell 25. H2O2 also cause mobilization of Fe2+ by Ca2+ via Fenton reduction stimulating the production of OH- radicals 26.
All these factors combinedly cause destabilization of the cell membrane, which is probably the key event of the lysis of the cell 25. Polyphenols possess many biological effects, mainly attributed to their antioxidant activities in scavenging free radicals, inhibition of peroxidation and chelation of transition metals 27, 28. Chidambaram et al., (2011) 29 proved that Andrographis paniculata has significant ability to stabilize the Red blood cell membrane. These findings are by them. The methanolic leaf extract of Andrographis paniculata showed inhibitory effect on all of the five bacterial species such as Pseudomonas aeruginosa, Staphylococcus aureus, Escherichia coli, Bacillus subtilis, and Streptococcus mutans. It is shown that methanol extract had a broad spectrum of activity. The extract has shown inhibitory zones ranging from 8 mm to 20 mm.
In all the bacteria used in this study, Pseudomonas aeruginosa proved to be more susceptible and exhibited maximum inhibitory zone 20 mm. The minimum inhibitory zone was observed for Streptococcus mutans 15 mm. As modern antibiotics have various anarchic toxic effects, these plant extracts could serve as an alternative antibacterial agent. In single plant many active secondary metabolites are present and medicinal effect can be attributed to either to a single compound or synergistic effect of many compounds and present antibacterial activity can either be due to the presence of some specific bioactive molecule or due to the synergistic effect of different phytoconstituents 30.
CONCLUSION: Through this present study, it can be concluded that the methanolic extract of A. paniculata leaves showed significant antibacterial activity and membrane stabilizing activity. These effects are due to the phyto inhibitors present in the leaf extract. The feasibility of making efficient drugs from plant sources is very much near. Further research should be done to isolate inhibitors and necessarily formulated.
ACKNOWLEDGEMENT: The authors wish to thank the secretary and correspondent of S.V.K.P & Dr. K. S. Raju Arts & Science College, Penugonda for providing the research laboratory and also chemicals which enabled us to complete this work. The authors are grateful to the expertise available in the campus that helped a lot in species identification.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Newman DJ, Cragg GM and Snader KM: The influence of natural products upon drug discovery. Natural Product Reports 2000; 17(3): 215-234.
- Anushia C, Sampathkumar P and Ramkumar L: Antibacterial and antioxidant activities in Cassia auriculata. Global Journal of Pharmacology 2009; 3(3): 127-130.
- Tomoko N, Takashi A, Hiromi T, Yuka I, Hiroko M, Munekazu I, Totshiyuki T, Tetsuro I, Fujio A, Iriya I, Tsotomu N and Kazuhito W: Antibacterial activity of extracts prepared from tropical and subtropical plants on methicillin-resistant Staphylococcus aureus. Journal of Health Sciences 2002; 48: 273-276.
- Kadali VN and Sandeep BV: Anti-hyperglycemic plants used by the traditional healer of west Godavari District, Andhra Pradesh, India. Int J Pharmacognosy 2015; 2(9): 473-77. doi link: http://dx.doi.org/10.13040/IJPSR. 0975-8232.IJP.2(9).473-77.
- Kadali VN, Kindangi KR, Rao PS and Sandeep BV: Wonder herbs having anti asthmatic activity present in West Godavari District, Andhra Pradesh, India- A Mini Review. Advances in Biology, Biotechnology and Genetics 2016; 3(1): 01-06.
- Kadali VN, Kindangi KR, Peter AE, Sudhakara Rao P, Bindiya P and Sandeep BV: Hepato-Protective Herbs- Present in West Godavari District of Andhra Pradesh, India- A mini review. International Journal of Medical and Health Research 2015; 1(1): 15-18.
- Kadali VN, Kindangi KR, Pola SR, Ramesh T and Sandeep BV: Cardio-protective plants present in West Godavari District of Andhra Pradesh, India: A Short Review. Int J Pharmacognosy 2016; 3(1): 100-07. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.3(1).100-07.
- Premanath R and Devi NL: Antibacterial, antifungal and antioxidant activities of Andrographis paniculata Leaves. IJPSR 2011; 2(8): 2091-2099.
- Rajeshkumar S: Antimicrobial effect of King of bitter Andrographis paniculata and traditional herb Aegle marmelos against clinical pathogens. Int J Pharm Tech Res 2014-2015; 7(2): 325-329.
- Sundar S, Sudevan S, Paramasivam R and Vijayaraghavan R: Evaluation of in-vitro anti-inflammatory activity of aqueous extract of Andrographis paniculata. Global J Pharmacol 2015; 9(4): 289-295.
- Sivakumar A, Manikandan A, Rajini Raja M and Jayaraman G: Andrographis paniculata leaf extracts as potential naja anti-snake venom. World J of Pharmacy & Pharmaceutical Sciences 2015; 4(12): 1036-50.
- Rajeshkumar S, Nagalingam M, Ponnanikajamideen M, Vanaja M and Malarkodi C: Anticancer activity of Andrographis paniculata leaves extract against neuroblastima (Imr-32) and human colon (Ht-29) cancer cell line. World Journal of Pharmacy and Pharmaceutical Sciences 2015; 4(6): 1667-1675.
- Madav S, Tripathi HC, Tandan SK and Mishra S: Analgesic, the antipyretic and antiulcerogenic effect of andrographolide. Ind J Pharm Sci 1995; 57: 121-125.
- Bu-Chin Y, Chen-Road H, Wang-Chuan C and Juei-Tang C: Antihyperglycemic effect of andrographolide in streptozotocin-induced diabetic rats. Planta Medica 2003; 69: 1075-1079.
- Sheeja K and Kuttan G: Activation of cytotoxic T lymphocyte responses and attenuation of tumor growth in- vivo by paniculata extract and andrographolide. Immunopharmacol Immunotoxicol 2007; 29: 81‐93.
- Calabrese C, Berman SH, Babish JG, MaX, Shinto L, Dorr M, Wells K, Wenner GA and Standish LJ: A phase I trial of andrographolide in HIV positive patients and normal volunteers. Phytother Res 2000; 14: 333‐338.
- Yoopan N, Thisoda P, Rangkadilok N, Sahasitiwat S, Pholphana N, Ruchirawat S and Satayavivad J: Cardiovascular effects of 14‐deoxy‐11, 12‐didehydro andrographolide and Andrographis paniculata Planta Med 2007; 73: 503‐511.
- Trivedi NP, Rawal UM and Patel BP: Hepatoprotective effect of andrographolide against hexachloro cyclohexane ‐induced oxidative injury. Integr Cancer Ther 2007; 6: 271‐280.
- Sharmila N and Gomathi N: Antibacterial, antioxidant activity and phytochemical studies of Crossandra infundibuliformis leaf extracts. International Journal of Phytomedicine 2011; 3: 151-156.
- Harborne JB: Phytochemical methods. In Chapman and Hall, New York, Edition 2nd, 1984: 21-26.
- Gayathri V and Kiruba D: Preliminary phytochemical analysis of leaf powder extracts of Psidium guajava International Journal of Pharmacognosy and Phytochemical Research 2014; 6(2): 332-334.
- Perez C, Pauli M and Bazerque P: An antibiotic assay by agar-well diffusion method. Acta Biologiae et Medecine Experimental 1990; 15: 113-115.
- Tavazzi B, Amorini AM, Fazzina G, Di-Pierro D, Tuttobene M and Giardina B: Oxidative stress induces impairment of human erythrocyte energy metabolism through the oxygen radical-mediated direct activation of AMP-deaminase. Journal of Biology and Chemistry 2001; 276: 48083-48092.
- Dluya T, Daniel D and Yaduma Gaiuson W: Comparative biochemical evaluation of leaf extracts of Ficus sycomorus and Piliostigma thonningii plant. Journal of Medicinal Plants Studies 2015; 3(5): 32-37.
- Chakraborty D and Shah B: Antimicrobial, antioxidative and antihaemolytic activity of Piper betel leaf extracts. Int J Pharm Pharm Sci 2011; 3(3): 192-199.
- Kupier‐Goodman T and Scott PM: Risk assessment of mycotoxin ochratoxin A. Biomedical and Environmental Sciences 1989; 2: 179‐248.
- Nickavar B, Kamalinejad M, Haj-Yahya M and Shafaghi B: Comparison of the free radical scavenging activity of six Iranian Achillea species. Pharmaceutical Biology 2006; 44: 208-12.
- James O and Alewo IM: In-vitro, antihaemolytic activity of Gymnema sylvestre extracts against hydrogen peroxide (H2O2) induced haemolysis in human erythrocytes. American Journal of Phytomedicine and Clinical Therapeutics 2014; 2(7): 861-869.
- Kumarapppan C, Jaswanth A and Kumarasunderi K: Antihaemolytic and snake venom are neutralizing effect of some Indian medicinal plants. Asian Pacific Journal of Tropical Medicine 2011; 743-747.
- Sati SC, Takuli P, Kuma P and Khulbe K: Antibacterial activity of three medicinal plants of Kumaun Himalaya against some pathogenic bacteria. International Journal of Pharma Sciences and Research 2015; 6(11): 1361-68.
How to cite this article:
Kadali VN, Ramesh T, Pola SR and Sandeep BV: In-vitro anti-haemolytic, anti-bacterial and phytoconstituents investigation of Andrographis paniculata methanolic leaf extract. Int J Life Sci & Rev 2016; 2(5): 102-08. doi: 10.13040/IJPSR.0975-8232.IJLSR.2(5).102-08.
All © 2015 are reserved by International Journal of Life Sciences and Review. This Journal licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.
Article Information
2
102-108
654
1504
English
IJLSR
V. N. Kadali *, T. Ramesh, S. R. Pola and B. V. Sandee
Department of Biotechnology, Andhra University, Visakhapatnam, Andhra Pradesh, India.
vnsimhakadalibiotech@gmail.com
15 April 2016
16 May 2016
19 May 2016
10.13040/IJPSR.0975-8232.IJLSR.2(5).102-08
31 May 2016