ISOLATION, CHARACTERIZATION AND PROCESS OPTIMIZATION OF ETHANOL PRODUCING THERMOTOLERANT POTENTIAL YEAST STRAINS FROM SUGAR INDUSTRIAL WASTE
HTML Full TextISOLATION, CHARACTERIZATION AND PROCESS OPTIMIZATION OF ETHANOL PRODUCING THERMOTOLERANT POTENTIAL YEAST STRAINS FROM SUGAR INDUSTRIAL WASTE
Md. Ariful Islam 1, Md. Shozan Mondal *2, Md. Abdullah Al-Masum 2, Sharif Md. Abuzar 2, Md. Tauhidul Islam 2 and Md. Ashraful Islam 2
Department of Mathematics and Natural Science 1, BRAC University, 66 Mohakhali, Dhaka - 1212, Bangladesh.
Department of Pharmacy 2, University of Asia Pacific, Dhanmondi, Dhaka - 1209, Bangladesh.
ABSTRACT: The supply of non-renewable energy sources from fossil fuel is depleting day by day, and it has become one of the greatest challenges to find alternative sources. Bioethanol as alternative biofuels has stimulated the worldwide interest. The use of bioethanol as fuel will minimize the amounts of fossil-derived carbon dioxide (CO2) to the Earth’s atmosphere. Yeast is the most acceptable of all organisms for ethanol production because of its diverse substrate specificity and ease of production of ethanol. Temperature is one of the most important environmental factors affecting microbial activity and production cost. The main objective of this research work was to isolation, characterization, and process optimization of ethanol-producing thermotolerant yeast strains. In total, 5 yeast isolates have been characterized based on morphological and physicochemical characters. Most of the strains were thermotolerant, ethanol tolerant, pH tolerant, as well as osmotolerant. All the strains were formed pseudo mycelium under stress condition. They were resistant to chloramphenicol, but growth was inhibited in the presence of 1% acetic Acid. The strains C and E showed good invertase activity, and all the strains were capable of fermenting glucose, fructose, sucrose, maltose, and amylose among 10 carbohydrates. Ethanol producing capability of the strains was studied using sugarcane molasses as substrate. Productivity was observed at different incubation temperature, pH, and reducing sugar concentration. After fermentation at different condition, at temperature 35 °C, pH 6.0, reducing sugar concentration 6.0% and shaking condition (115 rpm) found to be optimum for ethanol production by both C and E strains. The C and E strains produced maximum 11.25 % and 13.20% respectively at 48 hours under optimum condition. Pilot-scale (5 L) production by both strains was found 9.55% and 11.50% under optimum condition. These strains could be potential for ethanol production from cane molasses in a commercial scale.
Keywords: Bioethanol, Molasses, Thermotolerant, Characterization, Process optimization
INTRODUCTION: The supply of non-renewable energy sources from fossil fuel such as petroleum is expected to be limited one day since it is depleting day by day.
So it has become one of the greatest challenges for society today to find alternative sources to overcome this problem 1.
In this case, the utilization of renewable carbohydrate sources for the production of bioethanol has stimulated the worldwide interest 2. Bioethanol is renewable and help to reduce greenhouse gas emissions from fossil fuels 3. Yeast is the organisms capable of producing ethanol through fermentation of sugar. It is the most used of all organisms for ethanol production because of its diverse substrate specificity and ease of production of ethanol under anaerobic condition. Saccharomyces strains were used widely and traditionally for industrial ethanol production because of its ability to produce high concentrations of ethanol from hexoses and its high tolerance to ethanol and other inhibitory compounds 4. Carbohydrate-rich raw materials suitable for ethanol production can be classified into three groups of agricultural products. The first raw material group, sugar refers to sugar-beet as well as sugarcane and molasses. The second group, starch from cassava, cereals, and potatoes. The last group, lignocellulose, covers waste materials from the harvesting of crops such as rice straw, corn cob, and sugarcane bagasse 5. Molasses is the byproduct of the sugar industry most widely used carbon sources for bioethanol production by yeast.
Several factors like high temperature, low ethanol, and sugar tolerance of the yeast limit the industrial production of ethanol at low production costs. Physico-chemical and environmental factors such as inoculums type, moisture and water activity, pH, temperature, substrate, particle size, aeration and agitation, nutritional factors, oxygen, and carbon dioxide are affecting fermentation.6 The objective of this study was to isolate some thermotolerant yeast strains to evaluate their ethanol production capability using molasses as a substrate.
MATERIALS AND METHODS:
Isolation of Strain: The yeast strains were isolated from some sugar industries of Bangladesh. The sample of the sugar industry was soaked in YMM broth and incubated at 37 °C for 3 days then spread the ½ loop full of the liquid sample in YPD agar plate and incubate at 37 °C for 48 h. After observing under a microscope, the isolate strains were coded as “A, B, C, D and E.
Identification of Yeast Strains: The yeast strains were identified based on their culture characteristics (Colony color, shape, and surface appearance). According to 7 morphological and physicochemical characterization of the isolated yeasts was performed.
Pseudomycelium Formation: According to 8, the formation of pseudomycelium was investigated by slide culture technique.
Carbohydrate Fermentation: YMM broth base with Durham tube was used for testing of yeasts for carbohydrate fermentation. The carbohydrates were Glucose, Fructose, Sucrose, Lactose, Galactose, maltose, trehalose, raffinose, ribose, and xylose. The carbohydrates were dissolved, and indicator added; each yeast strains were inoculated into the medium then incubate for 48 h. The color of the medium changed from blue to yellow due to the formation of acids and gas 9.
Thermotolerance: Studied 10 the effects of temperature on fermentation capacity of three strains 19G, 78I and baker’s yeast in complete medium and sugarcane juice broth containing 15% total sugar. Complete conversion of total sugar to ethanol was observed after 12 h of fermentation at 39-40 °C. Above 40 °C, a strong inhibitory effect of temperature on ethanol production in all classes was observed. In this study, the YPD liquid medium was prepared and added 15 ml media in each McCartney tube for detecting thermotolerance.
After inoculating half loopful of Yeast cell in the media, the initial optical density of each tube was recorded on a spectrophotometer at 600 nm against the medium as blank. All cultures were incubated at 35°C, 37°C, 39°C, 41°C, 43°C, 45°C, 47°C and 49°C for 48 h and final optical density was recorded for observing thermotolerance of yeast strain. The increase in optical density in a tube was recorded as evidence of growth 11.
Ethanol Tolerance: A limitation of ethanol fermentation is the capacity of yeast to tolerate ethanol concentration. Ethanol, which is produced during fermentation, is inhibitory to cell growth than that from an exogenous source.12 YPD liquid medium was prepared and added 15 ml media in each McCartney tube for detecting ethanol tolerance. An ethanol concentration of absolute ethanol was varied from 5 to 23% (v/v) in the media. The effect of 5%, 8%, 11%, 14%, 17%, 20% and 23% were observed. The initial optical density of each tube was recorded on a spectrophotometer at 600 nm against the medium as blank. All the cultures were incubated at 30 °C for 48 h. The increase in optical density in a tube was recorded as evidence of growth.
pH Tolerance: YEPD media was prepared at different pH and added 15 ml media in each McCartney tube. Then each tube was inoculated by half loopful of the yeast cell and measured the initial optical density at 600 nm and incubated at 30°c for 48 h. After 48 h, cell density was further recorded at 600 nm for growth. The increase in optical density in a tube was recorded as evidence of growth 13.
Sugar Tolerance: The profitability of ethanol production is dependent on favorable sugar cane molasses price and the quality of molasses (sugar %) 14. Use of concentrated sugar substrate is one of the ways to obtain high ethanol yield during fermentation. However, high substrate concentration is inhibitory to fermentation due to osmotic stress 15. According to 16 sugar tolerance of the yeast strain was observed. YEPD broth was prepared containing 10%, 20%, 30%, 40%, 50% and 60% of sugar. Each McCartney tube contained 15 ml of YEPD liquid media with appropriate concentration of sugar, and blank media was used as a control. Then each was inoculated by half loopful of the Yeast cell and measured the initial optical density at 600 nm and incubated at 30°C for 48 h. After 48 h, cell density was further recorded at 600 nm. The increase in optical density in a flask was recorded as evidence of growth.
Osmotolerance: YPD broth was prepared containing 6%, 8%, 10%, 12%, 14%, 16%, 18 and 20% of NaCl. 15 ml of YPD liquid media was taken in each McCartney tube. Then each tube was inoculated by half loopful of the yeast cell and measured the initial optical density at 600 nm and incubated at 30°C for 48 h. After 48 h, cell density was further recorded at 600 nm.
Acetic Acid Tolerance: 1% of acetic acid containing YEPD broth was prepared. 15 ml of YEPD liquid media was taken in each McCartney tube. YEPD broth without Acetic acid was used as blank. Then half loopful of each Yeast strain was inoculated in the YEPD media, and the initial optical density at 600 nm was measured and incubated at 30 °C for 48 h. After 48 h, cell density was further recorded at 600 nm 17.
Chloramphenicol Resistance Test: Sensitivity to Chloramphenicol was evaluated by growing isolates in MEA in the presence of 30 µg/ml chloramphenicol discs, sample collection using the method of Kirby 18.
In this study, the YPD agar medium was used for detecting yeasts for Chloramphenicol resistance. Chloramphenicol antibiotic disc (30 µ/L) was placed into the center of the already inoculated Petri dish. Then the plate kept at 30°C for growing. The zone of inhibition by the disc was recorded as evidence of Chloramphenicol sensitivity.
Invertase Activities: Yeast strains grown on the agar slants were harvested by pouring sterile distilled water into the slants and gently scraping with a wire loop. The cells were washed, centrifuged, and 0.1 g wet weight of each was re-suspended in 10 ml of acetate buffer, pH 5.0 19, sucrose solution (4% w/v, 2 ml) in the same acetate buffer was inoculated with 1 ml of cell suspension for 5 min at 30 °C. The amount of reducing sugar released was determined by dinitro-salicylic acid method 20. The amount of enzyme which liberates 1 µmole reducing sugar per minute was defined as one unit of invertase activity.
Molasses Pretreatment: Molasses has been pretreated to remove dirt, particles and kill unwanted organisms, etc. 1kg Molasses is diluted with 0.5 L water, and 0.001% sulfuric acid was added 21. In this study Molasses 250 gm/L, Urea 0.10 gm/L and concentrated (H2SO4) 0 .30 ml/L was taken in a beaker and filter. It was then heated to the boiling and kept standing for a couple of hours before use. The reducing sugar was adjusted by boiling, and pH was adjusted by using Hydrochloric acid or Sodium Hydroxide.
Ethanol Production from Fermentation of Molasses: Fermentation was carried out in Erlenmeyer conical flasks. 250 ml of pre-treated fermentation media was taken into 500 ml Erlenmeyer flasks and then added the homogenous suspension of yeast was inoculated into the media in an aseptic condition. The flask was incubated at 35°C, 38°C, 41°C and 44°C with different reducing sugar concentration, pH and shaking condition (115rpm) 22.
Reducing Sugar estimation: The reducing sugar concentration due to the enzymatic reaction was determined by the DNS method 23.
Alcohol Estimation: According to 24 ethanol percentage in the fermentation broth was measured.
RESULTS:
Identification of Isolated Yeast: After observing the colony characteristics (white and creamy texture), presence of budding pattern Fig. 1 under a compound microscope, it was found that all the strains were yeast.
Pseudomycelium Formation: All the yeast isolates (A, B, C, D, and E) were produced pseudomycelium and showed in a filamentous form under a microscope Fig. 2. Filamentous pseudomycelium formation is a characteristic of Saccharomyces cerevisiae, which is dimorphic that results elongated chains of cells that remain, attached to one another 24.
Fermentation of Carbohydrates: In this study, the yeast isolates "A, B, C, D and E” utilized 5 sugars among ten. They were utilized glucose, fructose, sucrose, maltose and amylose but failed to ferment lactose, xylose, rhamnose, Raffinose and arabinose 25.
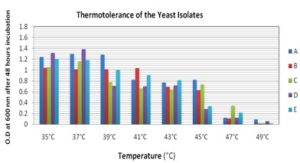
Thermotolerance: The isolated all the strains were thermotolerant, and some of them can tolerate high temperature.
FIG. 3: THERMOTOLERANCE OF THE YEAST ISOLATES
The isolates C and E can tolerate up to 47°C but A, B, and D can tolerate up to 45 °C. The growth was completely inhibited above 47°C in case of all strains. A C and D strains have grown maximum at 37°C, but B was grown at 41°C and E at 35°C Fig. 3. These thermotolerant yeast could produce a higher percentage of ethanol at a high temperature, which could reduce the production cost.
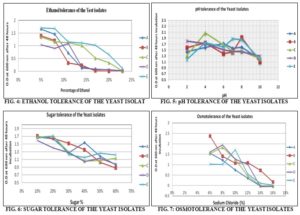
Ethanol Tolerance: A limitation of ethanol fermentation is the capacity of yeast to tolerate ethanol concentration. The strain A, B, C and D able to tolerate up to 14% (v/v) ethanol but the strain E can tolerate up to 20% (v/V) ethanol concentration. All the strain showed maximum growth at 8 % (v/v) ethanol, and none of the strains were grown tolerate above 20% (v/v) ethanol Fig. 4.
pH Tolerance: The strain A, B, C, D, and E were found to be grown at a wide range of pH (2 to 10). At highly acidic and basic pH, some of the strain showed low growth pattern, but most of the strains showed maximum growth at pH 6 Fig. 5.
Sugar Tolerance: All the strains found to tolerate up to 60% sugar concentration, but maximum growth was seen 20% sugar concentration Fig. 6.
Osmotolerance: None of the strain could successfully tolerate above 10% sodium chloride salt concentration, but at higher concentration growth was reduced Fig. 7. Maximum growth was seen at 6% sodium chloride salt concentration in case of all strains.
Acetic Acid Tolerance: None of the yeast isolates found to be tolerating 1%. 1% Acetic acid has inhibited the growth of all the 5 yeast strains in liquid YPED media after 48 h incubation Table 1.
TABLET 1: ACETIC ACID TOLERANCE OF THE YEAST ISOLATES
| Name of the Strains | Initial OD in 1% Acetic Acid containing YEPD liquid media | Final OD in 1% Acetic Acid containing YEPD liquid media | Concordant
value |
| A | 0.153 | 0.014 | -0.139 |
| B | 0.308 | 0.007 | -0.301 |
| C | 0.184 | 0.029 | -0.155 |
| D | 0.106 | 0.016 | -0.091 |
| E | 0.111 | 0.043 | -0.068 |
Chloramphenicol Resistance: Chloramphenicol 30µg/ml disk in YEPD plate has not inhibited the growth any of the strains Fig. 8.
Invertase Activity: Among all the strains A, C, and E found to be shown very good invert activities rather than strain B and D.
Ethanol Production from Fermentation of Molasses:
Primary Screening of Ethanol Producing Yeast: According to Conway method, alcohol production of all the characterized strains were investigated in a defined condition to isolate maximum ethanol-producing strain. In shaking condition at pH 5.0, sugar concentration 5.0% and temperature 30°C after 60 h following results were obtained: From the above-accumulated data, it was found that the C and E strains were the maximum ethanol producing strains among 5. So the strains C and E have been selected for further study. The effect of temperature, pH, and reducing sugar condition will be observed for ethanol production by both C and E strains.
FIG. 8: GROWTH ON CHLORAMPHENICOL CONTAINING SOLID YEPD AGAR MEDIA AT 30 °C
TABLE 2: INVERTAGE ACTIVITY OF THE SELECTED YEAST STRAINS
| Name of the Strains | Invertase activity (µmol/min) |
| A | 11.53 |
| B | 10.46 |
| C | 14.39 |
| D | 9.84 |
| E | 16.98 |
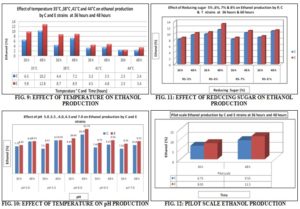
Effect of Temperature: It was found that all the strains were produced maximum ethanol at 35°C after 48 hours, whereas the pH of the media was 5.5 and reducing sugar concentration was 5.5%. But production rate was gradually decreased at a temperature 38°C, 41°C and 44°C. It was also found that E strain was maximum ethanol producing Yeast in all temperature. So the optimum temperature for both strains (C and E) was 35°C.
Effect of pH: pH has a marked influence in the production of ethanol by the yeast strains. It was found that both of the strains were produced maximum ethanol at pH 6.0 when the initial reducing sugar concentration was maintained at 5.5% and incubated at temperature 35°C. It was also found that C strain produced maximum 11.15% and E strain 12.74 at pH 6.0 Fig. 9. So the optimum pH found to be 6.00 for both strains.
Effect of Reducing Sugar: The sugar concentration in the media has a great influence on ethanol production by the yeast strains. It was found that both of the strains were produced maximum ethanol at initial reducing sugar concentration 6.0% when the pH of the media was maintained 6.0 % and incubated at temperature 35°C Fig. 10.
TABLE 3: ETHANOL PRODUCTION (%) OF SELECTED YEAST STRAINS TABULAR PRESENTATION
| Name of the Strains | Ethanol production (%) after 48 h | Ethanol production (%) after 60 h |
| A | 4.55 | 5.10 |
| B | 4.24 | 4.22 |
| C | 8.23 | 9.45 |
| D | 6.28 | 6.50 |
| E | 9.56 | 10.13 |
As the production rate at 6%, reducing sugar concentration was higher than other concentration. So the optimum reducing sugar percentage for both strains (C and E) found to be 6.0%.
Pilot-Scale Ethanol Production: The strain C and E were produced sufficient ethanol at lab scale bathes by using cost-effective method and materials. To identify the capability of the yeast strains on large scale; pilot scale production capacity was observed. Both strains were produced a satisfactory amount of ethanol in pilot scale batch (5 L). The C and E strain were produced 9.55% and 11.50% respectively after 48 h at 35°C temperature; where the initial reducing concentration of the media was maintained 6.0% and pH 6.0.
DISCUSSION: The fermentation process is always accompanied by the evolution of heat that raises the temperature of the fermenter. As a result, it becomes necessary to cool the large fomenters in the distilleries. This necessity often becomes a major operation and a cost factor in the production of ethanol. Temperature exerts a profound effect on the growth, metabolism, and survival of the fermenting organism. Fermentation in industries is usually carried out at an ambient temperature of 25- 35 ºC but temperature exceeds 40 ºC during fermentation, which decreases the cell viability and productivity. Maintenance of high cell viability is a major characteristic of fermentation to get high ethanol yield. Fermentation at 35- 40°C or above has advantages such as ethanol recovery and significant savings in operational costs of refrigeration control in distilleries for alcohol production 26. Samples were collected from industrial sugar waste. Based on some morphological & physiological characterization, presumptive yeast isolates has been selected. Based on the colony characteristics (white and creamy texture) ovoid microscope shape and budding pattern (multipolar), the selected isolate was found to belong Saccharomyces unicellular type ascomycete according to 27 Fig. 1. The identified strains can also produce pseudomycelium Fig. 2. All the strains were tested for fermentation of carbohydrates, and they were capable of fermenting five sugars among 10 sugars used as substrates. They fermented Glucose, fructose, sucrose, maltose and amylose but they couldn’t ferment lactose, xylose, rhamnose, raffinose, and arabinose.
According to 28, there are several potential benefits of thermotolerant yeast to be used in the production of industrial alcohol. Thermotolerant yeast exhibits rapid metabolic activity, and a high fermentation rate with high product output minimized contamination. Cooling costs during the process of ethanol production are expensive; hence, by using thermotolerant yeasts cooling and distillation costs can be reduced. 29 All the selected strains were thermotolerant. The isolates C and E can tolerate up to 47°C, but A, B, and D can tolerate up to 45°C. The growth was inhibited drastically above 47°C in case of all strains. A C and D strains have grown maximum at 37°C, but B was grown at 410C and E at 35°C Fig. 3.
A limitation of ethanol fermentation is the capacity of yeast to tolerate ethanol concentration because ethanol inhibits alcoholic fermentation 30. The strain A, B, C and D able to tolerate up to 14% (v/v) ethanol but the strain E can tolerate up to 20% (v/V) ethanol concentration. Among all the strains C and E strains have tolerated all concentration of ethanol. The strain C and E can tolerate up to 20% ethanol on the other hand A, B, and D strains were tolerated up to 14% ethanol. From this study, it could be concluded that strain C and E are more ethanol-tolerant than all other strains. Strain E showed extreme ethanol tolerance at 20% ethanol. The rate of ethanol production by yeast cells is highly affected by the pH of the fermentation medium. Most of the yeasts generally showed maximum growth under acidic conditions. Both acidic and basic conditions retard the yeast metabolic pathways and hence the growth of cells. 31 In this study, it was found that all the selected strains were able to grow a wide range of pH. They were able to grow spontaneously from pH 2 to pH 10. Maximum growth was seen at pH 4.
The profitability of ethanol production is dependent on the availability of sugar cane molasses, price, and the quality of molasses (sugar %). 32 Use of concentrated sugar substrate is one of the ways to obtain high ethanol yield during fermentation. However, high substrate concentrations are inhibitory to fermentation (Jones et al., 1981) due to osmotic stress. All the yeast strains were able to grow up to 60% sugar (sucrose) containing liquid YEPD media. Maximum growth was seen in 20% sugar containing media for all the strains.
None of the strain could successfully tolerate above 14% sodium chloride salt concentration, but only B strain can tolerate up to 14% Fig. 7. Maximum growth was found at 6% sodium chloride salt concentration in case of all strains. The strain A and B were found to maximum growth at 6% salt concentration; C and D at 8% and E at 10% salt concentration Fig. 7. 1% Acetic acid has inhibited the growth of all the 5 yeast strains Table 1. The selected strains were resistant to chloramphenicol. No zone of inhibition was observed against chloramphenicol disk (30µg/ml concentration) in case of all the strains Fig. 8.
Invertase is a yeast-derived enzyme. Invertase splits sucrose into glucose and fructose that are easily fermentable by yeast. A wide range of microorganisms produce invertase; thus, utilize sucrose as a nutrient. Commercially, invertase is biosynthesized chiefly by yeast strains of Saccharomyces cerevisiae or Saccharomyces carlsbergensis. In this study, good invertase activity was observed for most of the strains. Among all the strains A, B, and D found to be shown very good invert activities rather than strain C and E Table 2.
So they were able to breakdown rapidly sucrose into glucose and fructose that is readily usable. However, after all the characterization, the main target was to find the ethanol production capacity of the selected strains. The optimal pH range for growth of yeast can vary from pH 4.0 to 6.0, depending on temperature, the presence of oxygen, and the strain of yeast. This likely is due to the optimum pH value for the activity of plasma membrane-bound proteins, including enzymes and transport proteins. After all characterization, an optimum condition was selected for all the strains to see the ethanol production capacity. At shaking condition, temperature 300C, pH- 5.0, 5% Reducing Sugar concentration was used for all the strains. Among all 5 strains only C and D strains produced 9.15% and 10.13%, but A produced 5.10% B 4.225 and D 6.50% of ethanol after 60 hours incubation Fig. 8. So the strains C and E have been selected for further study. The effect of temperature, pH, and reducing sugar condition was observed for ethanol production by both C and E strains.
Incubation temperature is one of the vital factors for ethanol production, and the temperature has the vast impact on the ethanol production cost. To find the optimum temperature for ethanol fermentation, the yeast inoculated media was kept at temperature 35 °C, 38°C, 41°C, and 44°C in shaking condition. To determine the optimum temperature for ethanol fermentation, the inoculated media were kept at temperature 35 °C, 38°C, 41°C, and 44°C with fixed pH (5.5), reducing sugar concentration (5.5%) and shaking condition (115 rpm). At temperature 35°C the C strain was produced 6.5% of ethanol within 36 h and 10.20% at 48 h but E strain produced within 36 h 9.80% and 12.80% after 60 h. But production rate was gradually decrease with increasing the temperature.
At temperature 38°C strain C produced a maximum 7.2% and E 8.9% after 48 h incubation. At temperature 41°C strain C produced a maximum 3.5% and E 4.8% after 48 h, but at temperature 44°C ethanol production decreased more. So the optimum temperature for ethanol production found to be 35°C.
pH has a marked influence in the production of ethanol by the yeast strains. To determine the optimum pH for ethanol production, a series of fermentation batches were run with fixed temperature 35°C, initial reducing sugar concentration 5.5% in shaking condition (115 rpm). At pH 5.0, the strain C produced maximum 9.15 % but E 9.25% of ethanol after 48 hours incubation Fig. 9. At 5.5 pH the strain C and E produced an almost similar percentage of ethanol as pH 5.0 but ethanol production markedly increased at pH 6. At pH 6.0 the strain C produced maximum 11.15 % but E 12.74% of ethanol after 48 h incubation Fig. 9. On the other hand, ethanol production gradually decreased with increasing pH. At pH 6.5, C produced maximum 8.25 % but E produced 8.15% at pH 7.0, E produced maximum 9.54 % at pH 6.5 but 9.25% at pH 7.0. So the optimum pH found to be for both strain was 6.00.
The sugar concentration in the media has a great influence on the ethanol production by the yeast strains. To determine the optimum sugar concentration for ethanol production series of fermentation batches were conducted with fixed temperature 35 °C, pH 6.0 in shaking (115 rpm) condition. At 5.0% initial reducing sugar concentration, the strain C produced 9.58% and E produced 10.24 % after 48 hours incubation, but production rate gradually increased at 6% reducing sugar concentration. It was found that both of the strains were produced maximum ethanol C strain 11.25% and E strain 13.20% at initial reducing sugar concentration 6.0% when the pH of the media was maintained 6.0 and incubated at temperature 35 °C for 48 h Fig. 10. The ethanol production rate was almost similar percentage at 7% and 8% initial reducing sugar concentration. C produced at 7% reducing sugar 10.25% and at 8% reducing sugar it was 10.85 on the other hand, E produced 10.95% and 11.15% respectively. Considering all the sugar concentration and ethanol production capacity, 6% sugar concentration found to be optimum.
Pilot-scale (5 Liters) production by both strains was almost similar to that produced at a small scale (Shake flask 250 ml). The yeast strains were inculcated media containing reducing concentration of 6.0% and pH 6.0. Then incubated at 35 °C temperature, after 36 h C strain was produced 6.75% and E 8.03% ethanol. After 48 hours, the C and E strain were produced 9.55% and 11.50% respectively Fig. 11. These strains could be suitable for industrial scale.
CONCLUSION: The cell morphology of the yeast cells under a microscope is ovoidal to elongate, single or in pairs. Budding cells are present and ascospores, pseudo mycelium are developed. All the physiological and biochemical characters suggested that all the strains were yeasts. The fermentation of molasses using Strains C and E under different conditions showed that reducing sugar concentration 6.0%, temperature 35°C and pH 6.0 are suitable for ethanol production by free cells at 36 hrs and 48 hours in shaking condition.
Pilot-scale (5 Liters) production is almost similar to that observed with small scale (shake flasks 250 ml) production. These strains could be potential for ethanol production from cane molasses in a commercial scale. Productivity can also be improved by mutation through radiation or genetic manipulation. Metabolic pathway engineering to direct ethanol production may a promising way to improve productivity.
ACKNOWLEDGEMENT: The authors are indebted to the Institute of Food Science and Technology (IFST), Bangladesh Council of Scientific and Industrial Research (BCSIR), BRAC University and Incepta Pharmaceutical Ltd for necessary supports.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Hanh-Hagerdal B, Galbe M, Gorwa-Grauslund MF, Lidén, G and Zacchi G: Bio-ethanol -the Fuel of Tomorrow from the Residues of Today. Trends in Biotechnology 2006; 24(12): 549-556.
- Bandaru VVR, Somlanka SR, Mendu DR, Madicherla NR and Chityala A: Optimization of fermentation conditions for the production of ethanol from sago starch by co-immobilized amyloglucosidase and cells of Zymomonas mobilis using response surface methodology. Enzyme and Microbial Technology 2006; 38: 209-214.
- Ibeto CN, Ofoefule AU and Agbo KE: A global overview of biomass potentials for bioethanol production: An alternative renewable fuel. Trends App Sci Res 2011; 6(5): 410-425.
- Somda MK, Savadogo A, Ouattara CAT, Ouattara AS and Traore AS: Improvement of Bioethanol production using amylases properties from Bacillus licheniformis and yeast strains fermentation for Biomass valorization. Asian J Biotechnol 2011a; 3(3): 254-261.
- Mogg R: Biofuels in Asia: Thailand relaunches gasohol for automotive use. Refocus 2004; 5(3): 44-47.
- Krishna C: Solid-state fermentation systems-an overview. Crit Rev Biotechnol 2005; 25(12): 1-30.
- Boboye B and Dayo-Owoyemi I: Evaluation of dough sensory properties impacted by yeasts isolated from Cassava. J Appl Sci 2009; 9(4): 771-776.
- Kreger-Van Rij NJW: The yeast a taxonomic study. Elsevier Science Publishing Company, New York, 1984: 1082.
- Warren P and Shadomy L: Yeast fermentation broth base with carbohydrate and Durham tube. In: Manual of Clinical Microbiology. Washington, D.C., Edition 5th, 1991.
- Laluce C, Abud CJ, Greenhalf W and Sanches Peres MF: Thermotolerance behavior in sugarcane syrup fermentations of wild type yeast strains selected under pressures of temperature, high sugar and added ethanol. Biotechnol Lett 1993; 15: 609-14.
- Osman ME, Khattab OH, Hammad IA and El-Hussieny NI: Optimization of bio-fuel production by Saccharomyces cerevisiae isolated from sugar cane bagasse. J Am Sci 2011; 7(5): 485-492
- Wayman M and Parekh SR: Microbiology of fermentation catalysts. In Biotechnology of Biomass Conversion. Milton Keynes: Open University Press 1990; 75-100.
- Willaert R and Viktor AN: Primary beer fermentation by immobilized yeast - a review on flavor formation and control strategies. J Chem Technol Biotechnol 2006; 81: 1353-1367.
- Arshad M, Zia MA, Asghar M and Bhatti H: Improving bioethanol yield: Using virginiamycin and sodium fluoride at a Pakistani distillery. Afr J Biotechnol 2011; 10(53): 11071-11074.
- Borzani W, Garab A, Pires MH, Piplovic R, DeIa and Higuera GA: Batch ethanol fermentation of molasses: a correlation between the time necessary to complete the fermentation and the initial concentrations of sugar and yeast cells. World J Microbiol Biotechnol 1993; 9: 265-68.
- Ekunsanmi TJ and Odunfa SA: Ethanol tolerance, sugar tolerance and invertase activities of some yeasts strains isolated from steep water of fermenting cassava tubers. J App Bacteriol 1990; 69: 672-675.
- Fakruddin M, Islam MA, Quayum MA, Ahmed MM and Choudhury N: Characterization of stress tolerant high potential ethanol-producing yeast from agro-industrial waste. American Journal of BioScience 2013; 1(2): 24-34.
- Kirby WWM, Bauer AW and Sherris JC: Antibiotic susceptibility testing by a standardized single disk method. Am J Clin Pathol 1996; 45: 493-496.
- Jimenez J and Benitez T: Characterization of wine yeasts for ethanol production. App Microbiol Biotechnol 1986; 25: 150-154.
- Bernfield P: Enzymes of starch degradation and synthesis. Adv Enzymol 1951; 12: 379-481.
- Fakruddin M, Islam MA, Quayum MA, Ahmed MM and Choudhury N: Characterization of stress tolerant high potential ethanol-producing yeast from agro-industrial waste. American Journal of Bio Science 2013; 1(2): 24-34.
- Miller GL: Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 1959; 31: 426-428.
- Conway EJ: Micro-diffusion analysis and volumetric error. Crosby Lockwood and Son, London, 1939.
- Gimeno CJ, Ljungdahl PO, Styles CA and Fink GR: Unipolar cell divisions in the yeast S. cerevisiae lead to filamentous growth: regulation by starvation and RAS. Cell 1992; 68: 1077-1090.
- Bowknot T and Kurtzman CP: Principles and methods used in yeast classification, and an overview of currently accepted yeast genera. In Wolf, K. Nonconventional Yeasts in Biotechnology: A Handbook. Springer-Verlag: Heidelberg, 1996: 1-99.
- Osman ME, Khattab OH, Hammad IA and El-Hussieny NI: Optimization of bio-fuel production by Saccharomyces cerevisiae isolated from Sugar Cane Bagasse. J Am Sci 2011; 7(5): 485-492
- Lodder J: The yeasts: A Taxonomic study. North Holl and Publishing, Amsterdam, 1971.
- Roehr M: The Biotechnology of Ethanol: Classical and Future Applications. Chichester: Wiley-VCH 2001: 232.
- Sree NK, Sridhar M, Rao LV and Pandey A: Ethanol production in solid substrate fermentation using thermo-tolerant yeast. Process Biochemistry 1999; 34: 115-119.
- Fakruddin M, Islam MA, Quayum MA, Ahmed MM and Choudhury N: Characterization of stress tolerant high potential ethanol-producing yeast from agro-industrial waste. American Journal of Bio Science 2013; 1(2): 24-34.
- Willaert R and Viktor AN: Primary beer fermentation by immobilized yeast - a review on flavor formation and control strategies. J Chem Technol Biotechnol 2006; 81: 1353-1367.
- Arshad M, Zia MA, Asghar M and Bhatti H: Improving bioethanol yield: Using virginiamycin and sodium fluoride at a Pakistani distillery. Afr J Biotechnol 2011; 10(53): 11071-11074.
How to cite this article:
Islam MA, Mondal MS, Al-Masum MA, Abuzar S M, Islam MT and Islam MA: Isolation, Characterization and Process Optimization of Ethanol Producing Thermotolerant Potential Yeast Strains from Sugar Industrial Waste. Int J Life Sci & Rev 2015; 1(6): 227-37. doi: 10.13040/IJPSR.0975-8232.IJLSR.1(6).227-37.
All © 2015 are reserved by International Journal of Life Sciences and Review. This Journal licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.
Article Information
5
227-237
946
1864
English
IJLSR
M. A. Islam, M. S. Mondal*, M. A. Al-Masum, S. M. Abuzar, M. T. Islam and M. A. Islam
Department of Pharmacy, University of Asia Pacific, Dhanmondi, Dhaka, Bangladesh
shozan_uap15@yahoo.com
13 May 2015
23 June 2015
28 June 2015
10.13040/IJPSR.0975-8232.IJLSR.1(6).227-37
30 June 2015