LURASIDONE: A REVIEW OF ANALYTICAL METHODS FOR ESTIMATION IN PHARMACEUTICAL FORMULATION
HTML Full TextLURASIDONE: A REVIEW OF ANALYTICAL METHODS FOR ESTIMATION IN PHARMACEUTICAL FORMULATION
Zamir G. Khan, S. B. Bari and D. D. Patil *
Department of Pharmaceutical Chemistry, H. R. Patel Institute of Pharmaceutical Education & Research, Shirpur, Dhule - 425405, Maharashtra, India.
ABSTRACT: Lurasidone is an atypical antipsychotic approved by the FDA for the treatment of schizophrenic disorder and depressive episodes linked through bipolar I disorder in adults when used exclusively or in combination with lithium or valproate. Lurasidone is a benzisothiazole antipsychotic, an antagonist at D2, 5-HT2A and 5-HT7 receptors and a partial agonist at 5-HT1A receptors. As with other antipsychotics, the exact mechanism of action of lurasidone is unknown, although its effects in schizophrenia are supposed to be related to D2 and 5-HT2A receptor antagonism. This work includes published analytical methods so far reported in the literature, for estimation of lurasidone in biological samples and pharmaceutical formulations. Techniques like UV – Visible Spectrophotometry, high - performance liquid – chromatography (HPLC), high - performance thin - layer chromatography (HPTLC), and Liquid chromatography-mass spectroscopy (LC-MS) with ESI has been used for analysis, from which it can be witnessed that high-performance liquid chromatography methods have been applied most expansively.
Keywords: UV - Visible Spectrophotometry, HPTLC, RP-HPLC, LC-MS, Lurasidone, Validation
INTRODUCTION: Lurasidone is chemically (3aR, 4S, 7R, 7aS) - 2-hexahydro-4, 7-methano-2H-isoindole- 1, 3-dione hydrochloride. It was approved by the U.S. Food and Drug Administration (FDA) for treatment of schizophrenia on October 28, 2010 1. The chemical structure of lurasidone is shown in Fig. 1. Lurasidone hydrochloride is a benzisothiazole derivative and an atypical antipsychotic drug.
This drug has much high affinities for dopamine D2, serotonin 5-HT2A, 5-HT1A, 5-HT7 receptors, and α2c adrenoceptor, but only weak or negligible interactions with serotonin 5-HT2c, histamine H1, acetylcholine M1 receptors, and α 1 adrenoceptor. Lurasidone hydrochloride consists of six chiral centers, e.g. C1, C2, C11, C12, C15, and C16. Currently, the clinically used form is a single isomer 2.
Methods for pharmaceutical analysis are significantly much simpler comparatively with metabolites in biological samples as in urine, blood, and plasma. The estimation of a drug is, to a great extent, important in complex matrices because the pharmaceutical product quality is directly associated with patient wellbeing. In the drug development and pharmaceutical control, chemical analysis plays a key role to ensure high efficacy and safety for patients 3.
Lurasidone is practically insoluble in water, has poor bioavailability and slow onset of action and as a result could not be given in emergency clinical situations like schizophrenia. Bioavailability of poorly water-soluble drug can be enhanced through different techniques similar to solid dispersion and mixed hydrography. These techniques are useful in improving bioavailability as a result of increasing solubility. Solid dispersion is defined as a dispersion of drug in a matrix of the hydrophilic carrier. The solid dispersion technique is essentially used for BCS class II drugs. Drugs which come under BCS class II have low water solubility, and high permeability and these drugs have solubility as the rate-limiting step. So, if we promote solubility of the drug, subsequently bioavailability of drug will also increase. Hence there is a need to enhance the solubility of lurasidone 4.
Various analytical methods, including hyphenated techniques, can be performed for estimation of lurasidone in pharmaceutical formulations and biological samples. Pharmaceutical quality control provides several methods, and there is a need for fast, reliable, and definite analytical techniques. In the present work, we reviewed some of the recent quantitative published analytical techniques. These methods are mostly utilized in pharmaceutical analysis. We discussed the published paper mainly from 2010 to 2014. The selected papers were ordered according to the analytical technique employed. Several techniques discussed are the main techniques that have been used for the quantitative analysis of lurasidone in pharmaceutical formulation.
Analytical methods for estimation of Lurasidone:
UV – Visible Spectrophotometry: UV - VIS Spectrophotometric tests have been extensively developed to quantify lurasidone in the pharmaceutical dosage form. As most pharmaceuticals acquire chromophore groups, they can be indomitable straight in the ultraviolet region without the need for a derivatization reaction. The common accessibility of the instrumentation, the ease of procedures, economy, speed, precision, and precision of the technique still make spectrophotometric methods attractive 5.
However, direct UV/VIS spectrophotometric method is not suitable for the estimation of drugs with broad spectra. For this reason, derivative spectrophotometry offers a choice approach to the enhancement of sensitivity and specificity for analysis. This technique has been frequently used to extort information from bands of the analytes and pharmaceutical dosage forms. It consists of calculating and plotting first order and second order derivative of the mathematical expression of a spectral curve 6. UV – Visible Spectrophotometry methods have been developed for the estimation of lurasidone in bulk and pharmaceutical formulation summarised in Table 1.
Muvvala S Sudhir and Ratnakaram V Nadh 7 have been developed and validated a simple ultraviolet spectrophotometric method for estimation of lurasidone in bulk form. They have been reported lurasidone hydrochloride appears as a white to light yellow crystalline powder and is stable up to thirty-six months. In chloroform and acetonitrile, it is sparingly soluble; in ethanol, it is slightly soluble; in water and acetone, it is very slightly soluble, whereas in toluene and 0.1 N HCl it is insoluble. It has an aqueous solubility of 0.224 mg/ml in water with a maximum solubility of 0.349 mg/ml in pH 3.5 buffer. For analysis of lurasidone, the standard stock solution has been stored at + 4o C for better precision.
Mali Nikita et al., 8 have been developed and validated three spectrophotometric methods for the estimation of lurasidone HCl in bulk and pharmaceutical dosage forms. The first method (A) is zero order UV – Visible Spectrophotometric, second method (B) is First order derivative UV – Visible Spectrophotometric and third method (C) is zero order UV – Visible Spectrophotometric with Area Under Curve (AUC) technique.
Nirav K. Joshi et al., 9 have been reported the validation of a spectrophotometric method for estimation of lurasidone in bulk and pharmaceutical dosage form. Nirav also reported solvent suitability study of lurasidone HCl in methanol and have been proved that bulk drug is stable up to 48 h in a solvent.
TABLE 1: UV – VISIBLE SPECTROPHOTOMETRIC METHODS 7-9
| S. no. | Method | λ max
(nm) |
Solvent | Linearity
(µg/ml) |
r2 | %
recovery |
LOD, LOQ
(µg/ml) |
| 1 | Zero order 7 | 263 | ACN | 10 - 60 | 0.999 | 93.48-
100.88 |
1.25,
3.79 |
| 2 | Zero order 8 | 227 | Methanol | 5 - 30 | 0.999 | 99.40-
101.28 |
0.204,
0.619 |
| 3 | First order derivative 8 | 237 | Methanol | 5 - 30 | 0.999 | 100.33-
101.46 |
0.635,
1.925 |
| 4 | Zero order (AUC) 8 | 225 - 235 | Methanol | 5 - 30 | 0.999 | 99.83-
100.76 |
0.0831,
0.252 |
| 5 | Zero order 9 | 230 | Methanol | 10 - 50 | 0.996 | 98.5-
100.16 |
2.81,
8.43 |
* r2 – Correlation coefficient, ACN- Acetonitrile
Chromatographic Methods:
High Performance Liquid Chromatography (HPLC): HPLC is predominately used in the pharmaceutical industry for assessment of a large variety of samples. It is the method of preference for checking the purity of new chemical entities, monitoring changes in synthetic procedures or scale up, evaluating new formulations and carrying out quality control of the final drug product. HPLC technique utilizes the development of methods that would resolve unknown potential impurities and degradation products. It provides development of strategies for instrument qualification and validation to meet regulatory requirements. HPLC also offers rigorous validation of HPLC methods before they are utilized routinely.
Today the separation mode of alternative for the preponderance of High-Performance Liquid Chromatography (HPLC) analyses is Reversed-Phase Liquid Chromatography (RP–LC). Chromatographers within industrial setting habitually use RP - HPLC system of conventional size. The pharmaceutical industry repeatedly employees more than one chromatographic conditions with different selectivities for impurity profiling of drugs. HPLC cover various detectors like UV/Visible, conductivity detector, photodiode array detector, fluorescence detector, electrochemical detector, refractive index detector, evaporative light scattering detector, Mass spectrometer detector 10.
Nirav K. Joshi and Nehal J. Shah 11 have been reported development and validation of RP-HPLC method for estimation of lurasidone HCl, a novel antipsychotic drug in bulk and pharmaceutical dosage form.
P. Ravisankar et al., 12 have been described novel analytical method development and validation for the quantitative analysis of lurasidone HCl in bulk and pharmaceutical dosage forms by RP-HPLC.
Damle MC and Polawar AR 13 have been reported RP-HPLC method for estimation of lurasidone in bulk and pharmaceutical dosage form. They utilize methanol as a diluent for lurasidone; other methods use either mobile phase or ACN, which proves that this method is economic.
Pawanjeet J. Chhabda et al., 14 have been worked on development and validation of stability indicating a method for determination of lurasidone in bulk drug and pharmaceutical dosage form by RP-HPLC., is a first HPLC method which reported stability indicating a method for lurasidone. They reported that there was no significant degradation of lurasidone upon exposure to dry heat at 80 ºC for 24 h. Acid and photolysis treatment has been proved lurasidone to be stable, which indicated that the drug was stable against these three conditions. Significantly higher degradation observed in alkaline (16.19%) and oxidation condition (10.19%), however, a marked degradation is seen with 4% peroxide for a period of 5 h.
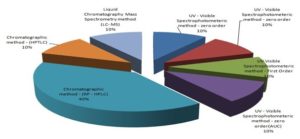
Many authors have been used C18 column with the changeability of different mobile phases for better separation and quantification of lurasidone in pharmaceutical dosage forms. It has been observed in all chromatographic methods, Welchrom C18 12 and Water X- Bridge C18 14 provide maximum sensitivity summarized in Table 2. HPLC is a method of choice has been chosen by most of the analysts for analysis of lurasidone in a variety of pharmaceutical dosage forms as depicted in Fig. 2.
TABLE 2: CHROMATOGRAPHIC METHODS (RP- HPLC) 11-14
| Column | Dimension (mm×mm×µ) | Mob
phase |
λmax (nm) | Linearity (µg/ml) | % Recovery | LOD, LOQ (µg/ml) | Detector | r2 | % Assay | Diluent |
| Inertsil C18 ODS-3V | 250×4.6×5.0 | ACN: MeOH: Acetic acid (30:45:25) v/v | 254 | 10-60 | 99.23-101.27 | 1.97,
5.96 |
UV Visible | 0.999 | 99.60 ±0.54 | MeOH |
| Welchrom C18 | 250×4.6×5.0 | 10mM phosphate buffer (pH-3.0): ACN (50:50) v/v | 235 | 10-50 | 99.61-99.86 | 0.065, 0.198 | UV Visible | 0.9999 | 99.90 | Mob phase |
| HiQsil C18 HS | 250×4.6×5.0 | MeOH:phospate buffer pH-3.0
(80:20) v/v |
231 | 5-25 | 98.53-100.19 | 0.62,
1.82 |
PDA | 0.996 | - | MeOH |
| Water X- Bridge C18 | 150×4.6×5.0 | 0.1% perchloric acid : ACN (50:50) v/v | 230 | 30-225 | 99.70- 99.96 | 0.07,
0.23 |
PDA | 0.9999 | - | MeOH |
* ODS- Octadecyl Silica, ACN- Acetonitrile, MeOH- Methanol, PDA- Photo Diode Array, Mob phase- Mobile phase
High Performance Thin Layer Chromatography (HPTLC): High-performance thin layer chromatography (HPTLC) is an instrumental sophisticated technique dependent on the inclusive capabilities of thin layer chromatography. The advantages of automation, full optimization, scanning, minimum sample preparation, selective detection principle, hyphenation, etc. facilitate it to be a powerful analytical tool for chromatographic information of complex mixtures of organic, inorganic, and biomolecules. Analytical chemists focus on new applications, new methods, and discoveries of analysis to boost up the specificity and sensitivity of a method. Many methods, just the once developed, are kept deliberately static so that data can be compared over long periods. HPTLC has strong potentials as a substitute to chromatographic model for estimating partitioning properties in support of environmental, combinatorial chemistry, and health effect studies 15.
M.C. Damle and A.R. Polawar 16 have been worked on development and validation of stability, indicating an HPTLC method for quantification lurasidone HCl. Developed method (range 200 – 1200 ng/spot) for the quantitative estimation of lurasidone in its single component tablet formulation, with LOD and LOQ, values 15.92 and 48.25 ng/band described. The sensitivity of the method, as summarized in Table 3. They have been observed that Lurasidone showed considerable degradation under acidic, alkali, oxidative and neutral hydrolytic conditions and results specify the suitability of the method to study the stability of lurasidone HCl under various forced degradation conditions like hydrolysis, photolytic degradation, and dry heat.
TABLE 3: HIGH- PERFORMANCE THIN- LAYER CHROMATOGRAPHY METHOD (HPTLC) 16
| Stationary phase | Diluent | Mobile phase | Scanning (nm) | Rf value | r2 | % Recovery |
| Silica Gel 60F254 | Methanol | Toluene: methanol (9.5:0.5) v/v | 231 | 0.48 ± 0.02 | 0.995 | 99.25 – 100.34 |
* r2 – correlation coefficient
Hyphenated Techniques:
Liquid Chromatography-Mass Spectrometry:
Kyeong-Ryoon Lee et al., have been reported Sensitive and Selective LC-MS method 17. They have been developed reproducible LC/MS assay using the ESI positive mode and validated for the estimation of lurasidone in rat plasma, bile, and urine.
Moreover based on the data has been reported in this, the assay appears to be pertinent for the determination of pharmacokinetic characteristics of lurasidone involving the administration of typical doses of the drug. Therefore, the analytical method may be useful in terms of characterizing the pharmacokinetic profiles of lurasidone in preclinical studies, summarized in Table 4.
TABLE 4: LIQUID CHROMATOGRAPHY – MASS SPECTROMETRY
| Stationary phase | Mobile phase | Scanning (m/z) lurasidone | Scanning (m/z) IS | Linearity (ng/mL) |
| Gemini C6-Phenyl column | ACN : 0.1% formic acid (80:20) v/v | 493 | 413 | 5-5000 |
FIG. 1: CHEMICAL STRUCTURE OF LURASIDONE
FIG. 2: 3D PIE DIAGRAM OF PERCENTAGE OF ANALYTICAL METHODS UTILISED FOR ESTIMATION OF LURASIDONE
CONCLUSION: In this paper, recent analytical methods employed for quantitative analysis of lurasidone in pharmaceutical formulations chiefly from 2010 to 2014 were reviewed. Several techniques like UV - Visible spectrophotometry, Chromatographic methods primarily (high - performance liquid - chromatography, high - performance thin - layer chromatography), Liquid chromatography-mass spectrometry with electrospray ionization are the major techniques that have been used. It is observed to use quicker techniques with cost savings and lessening in solvent consumption.
From this work, it has been observed that High-Performance Liquid Chromatography is extensively utilized for estimation of lurasidone in pharmaceutical formulation. It has been seen that for biological samples as rat plasma, bile, and urine, more sophisticated techniques like LC-MS have been used in the analysis of lurasidone.
ACKNOWLEDGEMENT: The authors would like to thank Department of Pharmaceutical Chemistry, H. R. Patel Institute of Pharmaceutical Education & Research, Shirpur, Dhule - 425405, Maharashtra, India.
CONFLICT OF INTEREST: The authors declare that there is no conflict of interests regarding the publication of this paper.
REFERENCES:
- Ishibashi T, Horisawa T, Tokuda K, Ishiyama T, Ogasa M, Tagashira R, Matsumoto K, Nishikawa H, Ueda Y and Toma S: Pharmacological profile of lurasidone, a novel antipsychotic agent with potent 5-hydroxytryptamine 7 (5-HT7) and 5-HT1A receptor activity. Journal of Pharmacology and Experimental Therapeutics 2010; 334(1): 171-81.
- Zhang H, Wang H, Zhu X, Yuan Z and Jiang H: Lurasidone hydrochloride. Acta Crystallographica Section E: Structure Reports Online 2012; 68(5): 1357-57.
- Pluym A, Van Ael W and De Smet M: Capillary electrophoresis in chemical/pharmaceutical quality control. TrAC Trends in Analytical Chemistry 1992; 11(1): 27-32.
- Madan JR, Pawar KT and Dua K: Solubility enhancement studies on lurasidone hydrochloride using mixed hydrotropy. International Journal of Pharmaceutical investigation 2015; 5(2): 114.
- Gorog, S: Ultraviolet-visible Spectrophotometry in Pharmaceutical Analysis: Application of UV-VIS Spectroscopy in Pharmaceutical CRC 1995.
- Talsky G, Mayring L and Kreuzer H: High‐Resolution, Higher‐Order UV/VIS Derivative Spectrophotometry. Angewandte Chemie International Edition in English 1978; 17(11): 785-99.
- Muvvala S and Ratnakaram V: Simple and validated ultraviolet spectrophotometric method for the estimation of lurasidone in bulk form. Research Journal of Pharmaceutical, Biological and Chemical Sciences 2013; 4(1): 609-17.
- Nikita M, Jignesh P and Mandev P: Validated spectrophotometric methods for the estimation of lurasidone hydrochloride in bulk and pharmaceutical dosage forms. International Journal of Research in Pharmacy & Science 2012; 2(2).
- Joshi NK, Shah N, Dumasiya M and Patel A: Development and validation of a spectrophotometric method for estimation of lurasidone hydrochloride: a novel antipsychotic drug in bulk and pharmaceutical dosage form. Pharma Science Monitor 2012; 3(4).
- Ahuja S and Rasmussen H: HPLC method development for pharmaceuticals. Academic Press 2011; 8.
- Joshi N and Shah N: Development and validation of RP-HPLC method for estimation of Lurasidone hydrochloride: a novel antipsychotic drug in bulk and pharmaceutical dosage form. Pharma Science Monitor 2012; 3(4): 2886-99.
- Bulk LHI: RP-HPLC, P. D. F. B. World Journal of Pharmaceutical Research 2014.
- Polawar A and Damle M: Development and validation of RP-HPLC method for estimation of Lurasidone hydrochloride in bulk and pharmaceutical dosage form. IJRPC 2014; 4: 327-32.
- Chhabda PJ: Development and validation of stability indicating a method for determination of Lurasidone in bulk drug and pharmaceutical dosage form by HPLC. IJPRD 2013; 5: 103-14.
- Srivastava M: High performance thin layer chromatography (HPTLC). Springer Science & Business Media 2010.
- Polawar A and Damle M: Development and validation of stability indicating HPTLC method for quantification of lurasidone HCL. Pharma Science Monitor 2014; 5(3).
- Chae YJ, Koo TS and Lee KR: A sensitive and selective LC-MS method for the determination of lurasidone in rat plasma, bile, and urine. Chromatographia 2012; 75(19-20): 1117-28.
How to cite this article:
Khan ZG, Bari SB and Patil DD: Lurasidone: A Review of Analytical Methods for Estimation in Pharmaceutical Formulation. Int J Life Sci & Rev 2016; 2(2): 17-22. doi: 10.13040/IJPSR.0975-8232.IJLSR.2(2).17-22.
All © 2015 are reserved by International Journal of Life Sciences and Review. This Journal licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.
Article Information
1
17-22
691
2103
English
IJLSR
Z. G. Khan, S. B. Bari and D. D. Patil
Department of Pharmaceutical Chemistry, H. R. Patel Institute of Pharmaceutical Education & Research, Shirpur, Dhule, Maharashtra, India.
dipakpatil888@rediffmail.com
08 January 2016
23 February 2016
25 February 2016
10.13040/IJPSR.0975-8232.IJLSR.2(2).17-22
29 February 2016