MORPHOLOGICAL CHARACTERIZATION OF PYRICULARIA ORYZAE CAUSING BLAST DISEASE IN RICE (ORYZA SATIVA L.) FROM DIFFERENT ZONES OF KARNATAKA
HTML Full TextMORPHOLOGICAL CHARACTERIZATION OF PYRICULARIA ORYZAE CAUSING BLAST DISEASE IN RICE (ORYZA SATIVA L.) FROM DIFFERENT ZONES OF KARNATAKA
P. Manjunatha * and M. Krishnappa
Department of Applied Botany, Kuvempu University, Shankaraghatta, Shivamogga - 577451, Karnataka, India.
ABSTRACT: Study was conducted to describe the cultural and morphological characteristics such as color and texture of the leaf blast pathogen Pyricularia oryzae on different solid media viz., Host extract agar, Oatmeal agar, Potato dextrose agar, and Richard's agar medium. Among all the solid media the highest mean mycelial growth (diameter) of the fungus Pyricularia oryzae was recorded on Host extract agar (40.80 mm) followed by Oat meal agar (38.33 mm) and least mean mycelial growth of the P. oryzae onRichard's agar (28.4 mm). The highest mean dry mycelial weight(mg) Rechard’s agar (300.65mg) followed by Oat meal agar (234.67 mg) and least mean mycelial weight was recorded in Potato Dextrose agar (96.31 mg). In general, among all solid media, the Host extract agar media is more appropriate for cultural and morphological study of rice blast fungus P. oryzae.
Keywords: Mycelial, Richard’s agar, Blast, Pyricularia
INTRODUCTION: Rice blast is one of the most spread and the most damageable diseases of rice in India. Rice is a member of the grass family (Poaceae). There are more than 10,000 species of grasses distributed among 600 genera. Grasses occur worldwide in a variety of habitats. Rice is the most important food crop of India covering about one-fourth of the total cropped area and providing food to about half of the Indian population. Rice is unique because it can grow in wet environments that other crops cannot survive in. Such wet environments are abundant across Asia. Rice is life for thousands of millions of people.
In Asia alone, more than 2,000 million people obtain 60 to 70 percent of their calories from rice and its products. Recognizing the importance of this crop, the United Nations General Assembly declared 2004 as the “International Year of Rice” (IYR). The theme of IYR - “Rice is life” reflects the importance of rice as a primary food source, and is drawn from an understanding that rice-based systems are essential for food security, poverty alleviation and improved livelihood. Rice suffers from many diseases caused by fungi, bacteria, viruses, phytoplasma, nematodes, and other non-parasitic disorders.
Among the fungal diseases, the blast is considered as a major threat to rice production because of its widespread distribution and its destructiveness under favorable conditions. The Commonwealth Mycological Institute has recorded its presence from 85 countries throughout the world. Paddy blast is generally considered as the principal disease of rice and is caused by a fungus belonging to the Ascomycete Pyricularia oryzae Cavara (Teleomorph = Magnaporthe grisea (Hebert) Barr Combnov.). Losses due to the blast disease may range up to 90 percent depending upon the component of the plant infected. Pyricularia oryzae infects above ground parts of the plant, but neck blast and the panicle blast are the most damaging phases of the disease and have been shown to significantly reduce yield, grain weight, and milling quality. The pathogen may infect all the above-ground parts of a rice plant at different growth stages viz., leaf, collar, node, internodes, base or neck and other parts of the panicle and sometimes the leaf sheath. A typical blast lesion on a rice leaf is gray at the center, has a dark border, and it is spindle-shaped.
MATERIALS AND METHODS: Effect of different media on the growth of P. Oryzae Culture discs of the pathogen (5 mm) was inoculated separately on different media and incubated at 28 ± 1 ºC for 15 days. The cultural characters and the colony diameter (mm) on each medium were recorded. Fifteen ml of each medium were poured into each of the sterilized Petri plates. Inoculation was made by transferring the five mm disk of mycelia mat, taken from the periphery of ten days old culture of each fifteen isolates. Each treatment were replicated thrice. The plates were incubated at 28 ± 1 ºC. Observation of colony growth was taken when the maximum growth was attained in any one of the media tested. Other cultural characters viz., rate of growth, type of margin, colony color and sporulation were also recorded. The pathogen was multiplied by transferring a loop full of the stock culture to 250 ml of potato dextrose broth taken in a 1000 ml flask. The inoculated flask was incubated at 28 ± 10 ºC for fourteen days. The concentration of spore suspension was adjusted to 50 spores/microscopic field by adding sterilized distilled water.
Maintenance of Culture: All the fifteen isolates of P. oryzae were maintained on PDA slants at 40 ºC in the refrigerator and subculture periodically at an interval of 30 days during this study.
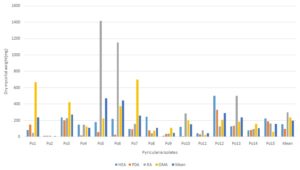
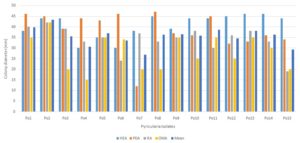
RESULTS AND DISCUSSION: Growth of P. oryzae on different solid media. The experiment was conducted as detailed in material and methods‟ to ascertain the period when the maximum growth of the fungus could occur among all the solid media the highest mean mycelial growth of the fungus Pyricularia oryzae was recorded Host extract agar (40.80 mm) followed by Oat meal agar (38.33 mm) and least mean mycelial growth of the P. oryzae on Richard's agar (28.4 mm) Table 3, Fig. 2. The highest mean dry mycelial weight(mg) Richard's agar (300.65mg) followed by Oatmeal agar (234.67 mg) and least mean mycelial weight was recorded in Potato Dextrose agar (96.31 mg). Table 2, Fig. 1. Morphological and cultural studies were carried to find out the variation for the growth and sporulation of isolates Table 1. Cultural characters of each of the isolates studied on four different solid media at room temperature 28 ºC showed the variation among the isolates of P. oryzae. Morphological characters viz., size and shape of conidia were studied for identification of the fungus.
FIG. 1: EFFECT OF DIFFERENT MEDIA ON MYCELIAL DRY WEIGHT (mg)
HEA: Host extract agar, OMA: Oat meal agar, PDA: Potato dextrose agar and RA: Richard's agar
FIG. 2: EFFECT OF DIFFERENT MEDIA ON COLONY GROWTH
HEA: Host extract agar, OMA: Oatmeal agar, PDA: Potato dextrose agar and RA: Richard's agar
Conidia were pyriform, almost greyish black, 2-septate and 3 celled. The shape, size, septation, and color characters are in agreement with those described by Nishikado (1926) 6 and Mijan Hossain (2000) 4.
Cultural characteristics studied on different media showed the variation among fifteen isolates of P. oryzae concerning colony characters like the type of growth, the color of colony and colony margin 8. Colour varied from grayish black to dark jet black color, smooth to irregular margin, medium to good growth of the pathogen were observed 7.
These fifteen isolates exhibited considerable variation in colony type and color when grown on different nutrient media Table 2.
TABLE 1: SOURCE OF PYRICULARIA ISOLATES COLLECTED FROM DIFFERENT REGION OF KARNATAKA
| S. no. | Isolate Number | Location | Plant part | Variety | Zone |
| 1 | Po1 | Gangavathi | leaf | BPT-5204 | Northern Dry Zone 3 |
| 2 | Po2 | Siraguppa | leaf | BPT-5204 | Northern Dry Zone 3 |
| 3 | Po3 | Mandya | leaf | Thanu | Southern Dry Zone 6 |
| 4 | Po4 | Shivamogga | leaf | Jyothi | Southern Transition Zone 7 |
| 5 | Po5 | Shivamogga | Neck | Jyothi | Southern Transition Zone 7 |
| 6 | Po6 | Haveri | leaf | Abhilash | Northern transition Zone 8 |
| 7 | Po7 | Mugad | leaf | Abhilash | Northern transition Zone 8 |
| 8 | Po8 | Mugad | Neck | Intan | Northern transition Zone 8 |
| 9 | Po9 | Sirsi | leaf | Intan | Hilly Zone 9 |
| 10 | Po10 | Malagi | leaf | Intan | Hilly Zone 9 |
| 11 | Po11 | Malagi | Neck | Intan | Hilly Zone 9 |
| 12 | Po12 | Mundogadu | leaf | Intan | Hilly Zone 9 |
| 13 | Po13 | Mudigere | leaf | Intan | Hilly Zone 9 |
| 14 | Po14 | Ponnampet | leaf | Intan | Hilly Zone 9 |
| 15 | Po15 | Ponnampet | Neck | Intan | Hilly Zone 9 |
Potato Dextrose Agar: Colonies were buff colored in isolates Po2, Po3, Po4, Po5, Po6, Po7, and Po8. However isolates Po1, Po9 and Po15 showed grayish black color, while isolates Po10, Po11, Po12, Po13, and Po14 showed black color colonies 1. The isolates Po1, Po4, and Po11 only, showed good to medium colony growth.
Richard’s Agar: Almost all isolates showed grayish color colonies with smooth and marginal good growth, except Po4, Po7, Po8, Po11, Po12, Po13, Po14, and Po15 showed irregular marginal growth.
Oat Meal Agar: Colonies were grayish colored in isolates Po1, Po4, Po5, Po7, Po9, Po10, Po12, and Po13. However isolates Po2, Po3, Po6, Po8, and Po15 showed black color while only isolate Po11 showed green-black color colonies 1. Except for Po3 and Po5, which showed irregular marginal growth, all other isolates showed smooth and good growth.
TABLE 2: COLONY CHARACTERISTICS OF DIFFERENT ISOLATE OF PYRICULARIA ORYZAE ON DIFFERENT MEDIA
| Media | Colony Characters | ||||||
| Po1 | Po2 | Po3 | Po4 | Po5 | Po6 | Po7 | |
| Host extract agar | Grayish black color smooth colony margin good growth | Grayish black color smooth colony margin good growth | Grayish black color smooth colony margin good growth | Grayish black color smooth colony margin good growth | Grayish black color smooth colony margin good growth | Grayish black color smooth colony margin good growth | Grayish black color smooth colony margin good growth |
| Oat meal agar | Grayish black color smooth colony margin medium growth | Black color smooth colony margin good growth | Black color irregular margin growth | Grayish black color margin medium growth | Grayish black color irregular margin medium growth | Black color smooth colony margin good growth | Grayish black color smooth colony margin medium growth |
| Potato dextrose agar | Grayish black color smooth colony margin good growth | buff color smooth margin risedmycelial good growth | buff color smooth margin risedmycelial good growth | buff color smooth margin risedmycelial good growth | buff color smooth margin risedmycelial good growth | buff color smooth margin risedmycelial good growth | buff color smooth margin risedmycelial good growth |
| Richard's agar | Grayish color smooth colony margin good growth | Grayish color smooth colony margin good growth | Grayish color smooth colony margin medium growth | Grayish color smooth colony margin irregular growth | Grayish color smooth colony margin good growth | Grayish color smooth colony margin good growth | Grayish color smooth colony margin irregular growth |
| Media | p8 | p9 | p10 | p12 | p13 | p14 | p15 |
| Host extract agar | Grayish black color smooth colony margin good growth | Grayish black color smooth colony margin good growth | Grayish black color smooth colony margin good growth | Grayish black color smooth colony margin good growth | Grayish black color smooth colony margin good growth | Grayish black color smooth colony margin good growth | Black color smooth colony margin good growth |
| Oat meal agar | Black color smooth colony margin good growth | Grayish black color smooth colony margin good growth | Grayish black color smooth colony margin good growth | Grayish black color smooth colony margin good growth | Grayish black color smooth colony margin good growth | Green, black color smooth colony margin good growth | Black color smooth colony margin good growth |
| Potato dextrose agar | Buff color smooth margin risedmycelial good growth | Grayish black color smooth colony margin good growth | Black color smooth colony margin good growth | Black color smooth colony margin good growth | Black color smooth colony margin good growth | Black color smooth colony margin good growth | Grayish black color smooth colony margin good growth |
| Richard's agar | Grayish color smooth colony margin irregular growth | Grayish color smooth colony margin good growth | Grayish color smooth colony margin good growth | Grayish color smooth colony margin irregular growth | Grayish color smooth colony margin irregular growth | Grayish color smooth colony margin irregular growth | Grayish color smooth colony margin irregular growth |
TABLE 3: COLONY DIAMETER OF ISOLATES IN DIFFERENT MEDIA
| Isolates | Colony diameter in different media (mm) | |||||
| HEA | OMA | PDA | RA | Mean | ||
| Po1 | 38 | 46 | 40 | 35 | 39.75 | |
| Po2 | 44 | 45 | 42 | 42 | 43.25 | |
| Po3 | 44 | 39 | 39 | 20 | 35.50 | |
| Po4 | 30 | 44 | 33 | 15 | 30.50 | |
| Po5 | 35 | 43 | 35 | 35 | 37.00 | |
| Po6 | 30 | 46 | 24 | 34 | 33.50 | |
| Po7 | 38 | 12 | 37 | 20 | 26.75 | |
| Po8 | 45 | 47 | 33 | 20 | 36.25 | |
| Po9 | 39 | 37 | 35 | 35 | 36.50 | |
| Po10 | 44 | 36 | 38 | 25 | 35.75 | |
| Po11 | 44 | 45 | 30 | 35 | 38.50 | |
| Po12 | 45 | 32 | 36 | 25 | 34.50 | |
| Po13 | 46 | 33 | 38 | 35 | 38.00 | |
| Po14 | 46 | 36 | 33 | 30 | 36.25 | |
| Po15 | 44 | 34 | 19 | 20 | 29.25 | |
| Mean | 40.80 | 38.33 | 34.13 | 28.40 | 35.42 | |
| SEM± | 1.07 | |||||
| CV (%) | 11.77 | |||||
HEA: Host extract agar, OMA: Oatmeal agar, PDA: Potato dextrose agar and RA: Richard's agar
Host Extract Agar: Except Po15 (Black colonies) all other isolates showed grayish black colonies. All isolates showed smooth and marginal good growth.
TABLE 4: DRY MYCELIA WEIGHT OF PYRICULARIA ISOLATES GROWN IN DIFFERENT MEDIA
| Isolates | Mycelia Dry weight (mg) in different media | Mean | ||||
| HEA | PDA | RA | OMA | |||
| Po1 | 84.6 | 150.4 | 49.6 | 665.5 | 237.53 | |
| Po2 | 11.5 | 12.6 | 14.6 | 2.9 | 10.40 | |
| Po3 | 237 | 200.34 | 227.4 | 420 | 271.19 | |
| Po4 | 150 | 18.2 | 150.8 | 124.6 | 110.90 | |
| Po5 | 180 | 56.3 | 1416.1 | 223.2 | 468.90 | |
| Po6 | 220 | 25.7 | 1150 | 373.3 | 442.25 | |
| Po7 | 96 | 90.4 | 160 | 699 | 261.35 | |
| Po8 | 245.8 | 78.3 | 44.6 | 74.6 | 110.83 | |
| Po9 | 15.3 | 35 | 38.1 | 109.7 | 49.53 | |
| Po10 | 123.5 | 10.45 | 287.6 | 199.1 | 155.16 | |
| Po11 | 44.5 | 32.6 | 80.7 | 20.9 | 44.68 | |
| Po12 | 500.45 | 330 | 127.4 | 204.2 | 290.51 | |
| Po13 | 125.5 | 134.56 | 500.87 | 185.3 | 236.56 | |
| Po14 | 80.4 | 82.9 | 98 | 155.8 | 104.28 | |
| Po15 | 222.7 | 186.9 | 164 | 62 | 158.90 | |
| Mean | 155.82 | 96.31 | 300.65 | 234.67 | 196.86 | |
| SEM± | 35.32 | |||||
| CV (%) | 69.50 | |||||
HEA: Host extract agar, OMA: Oatmeal agar, PDA: Potato dextrose agar and RA: Richard's agar
Growth Phase: The experiment was conducted as detailed in ‘Material and methods’ to ascertain the period when the maximum growth of the isolates could occur. PDA, HEA, OMA, and RA were used in the study as a basal media, and the dry mycelia weight of each isolated were taken on the 10th day of inoculation.
Dry mycelial weight of the isolates Po3, Po6, Po12, and po15 were maximum in HEA media. The dry mycelial weight of the isolates Po3, Po12 and Po15 were maximum in PDA media.
The dry mycelial weight of the isolates Po3, Po6, Po10, and Po13 were maximum in RA media and the dry mycelial weight of the isolates Po3, Po5, Po7, Po12 and Po14 were maximum in OMA media.
There have been several reports regarding such variation in growth rate among isolates of a pathogen. Simulated observations have been recorded by Kulkarni (1973) 3, Onofeghara, et al., (1973) 7 and Akhilesh et al. (2017) 1.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Kulmitra AK, Sahu N, Sahu MK, Kumar R, Kushram T, and Kumar VBS: Growth of rice blast fungus Pyricularia oryzae on different solid and liquid media, Int J Curr Microbiol App Sci 2017; 6(6): 1154-60.
- Anonymous 2016. Statistical database. www.fao.org.
- Kulkarni S: Studies on the blast diseases of the Eleusine coracana (L.) Gaertn. In Mysore state. M.Sc. (Agri) Thesis, U.A.S., Bangalore, India 1973: 104.
- Hossain M: Studies on blast disease of rice caused by Pyricularia grisea (cooke) Sacc. In upland area. M.Sc. Thesis, Univ Agric Sci Dharwad 2000: 52-53.
- Netam RS, Bahadur AN, Tiwari RKS and Tiwari U: Effect of different culture Media, carbon source, nitrogen Source, temperature and pH, level on the growth and sporulation of Pyriculariagrisea isolate from finger millet. Res J Agric Sci 2013; 4(1): 83-86.
- Nishikado Y: Studies on rice blast disease. Japanese J Botany 1926; 3: 239-44.
- Onofeghara FA, Kaporia, RG and Ademokuba DO: Studies onPyriculariaoryzae Lav. in Sierralaone morphological and physiological variability of some isolates. Annals of Bot 1973; 37: 193-02.
- Ou SH: Rice Diseases (2nd edn). CABI Publishing, Wallingford, UK 1985: 380.
- Srivastava RK, Bhatt RP, Bandyopadhyay BB and Kumar J: Effect of media on growth, sporulation and production of perithecia of blast pathogen Pyricularia grisea. Res Environ Life Sci 2009; 2(1): 3740.
- Vanaraj P, Saveetha K. Sankaralingam A, Rabindram R. and Robin S: Variability in Pyricularia oryzae from different rice growing regions of Tamil Nadu, India. Afr J Microbiol 2013; 7(26): 3379-88.
How to cite this article:
Manjunatha B and Krishnappa M: Morphological characterization of Pyricularia oryzae causing blast disease in rice (Oryza sativa L.) from different zones of Karnataka. Int J Life Sci & Rev 2019; 5(7): 112-16. doi: 10.13040/IJPSR.0975-8232.IJLSR.5(7).112-16.
All © 2015 are reserved by International Journal of Life Sciences and Review. This Journal licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.