NANOSUSPENSIONS: A PROMISING DRUG DELIVERY STRATEGY
HTML Full TextNANOSUSPENSIONS: A PROMISING DRUG DELIVERY STRATEGY
Afrin * 1, Sunil Kumar Prajapati 1, Dilip Kumar Chanchal 1, Neha Agnihotri 1, Sakshi Tiwari 1, Deepti Singh 2 and Chandni Ali 2
Institute of Pharmacy 1, Bundelkhand University, Jhansi - 284128, Uttar Pradesh, India.
Department of Pharmaceutics 2, Agra Public Pharmacy College, Artoni, Agra - 282001, Uttar Pradesh, India.
ABSTRACT: Nanosuspensions have emerged as a promising strategy for the efficient delivery of hydrophobic drugs because of their versatile features and unique advantages. Recently, nanoscale systems have received much interest as a way to resolve solubility issues because of their cost-effectiveness and technical simplicity compared to liposomes and other colloidal drug carriers. Nanosuspensions have proven to be a better alternative over other approaches currently available for improving the bioavailability of several drugs with low solubility. A formulation as nanosuspension is an attractive and promising alternative to solve these problems. Nanosuspension consists of the pure poorly water-soluble drug without any matrix material suspended in dispersion. Preparation of nanosuspension is simple and applicable to all drugs, which are water insoluble. A nanosuspension not only solves the problems of poor solubility and bioavailability but also alters the pharmacokinetics of the drug and thus improves drug safety and efficacy. Nanosuspensions have been extensively developed for a wide range of drugs and have been evaluated for in-vitro and in-vivo applications by various routes: parenteral, oral, pulmonary, and topical. They have also been used for drug targeting. Different preparation methods for Nanosuspensions and their application are being reported and patented. The number of products based on nanosuspension in the market and under clinical study is higher than that of other nanotechnology-based applications.
Keywords: Nanosuspensions, Hydrophobic drugs, Solubility issues, and applications
INTRODUCTION: The formulation of poorly water-soluble drugs has always been a challenging problem faced by pharmaceutical scientists, and it is expected to increase because approximately 40% or more of the new chemical entities being generated through drug discovery programs are poorly water-soluble.
The problem is even more intense for drugs such as itraconazole and carbamazepine (belonging to class III as classified), as they are poorly soluble in both aqueous and organic media, and for drugs having a log P value of 2. Such drugs often have an erratic absorption profile and highly variable bioavailability because their performance is dissolution-rate limited and is affected by the fed/fasted state of the patient.
Traditional strategies, such as micronization, solubilization using co-solvents, the use of permeation enhancers, oily solutions, and surfactant dispersions, which evolved earlier to tackle the formulation challenges, have limited use.
Although reasonable success has been achieved in formulating water-insoluble drugs using liposomes, emulsions, microemulsions, solid dispersion technology, and inclusion complexes employing cyclodextrins, there is no universal approach applicable to all drugs. Hence, there is a growing need for a unique strategy that can tackle the formulation-related problems associated with the delivery of hydrophobic drugs to improve their clinical efficacy and optimize their therapy concerning pharmaco-economics 1.
Nanotechnology can be used to solve the problems associated with various approaches described earlier. Nanotechnology is defined as the science and engineering carried out in the nanoscale that is 10–9 m. The micro drug particles/micronized drug powder is transferred to drug nanoparticles by techniques like Bottom-Up Technology and Top-Down Technology 2. A pharmaceutical nano-suspension is defined as very finely dispersed solid drug particles in an aqueous vehicle for either oral and topical use or parenteral and pulmonary administration. The particle size distribution of the solid particles in Nanosuspensions is usually less than one micron with an average particle size ranging between 200 and 600 nm nanosuspension is a sub-micron colloidal dispersion of drug particles which are stabilized by surfactants, polymers or a mixture of both.
This formulation has a high drug loading, low incidence of side effects by the excipients, and low cost Owing to the increased surface-to-volume ratio of the nanocrystals, an increase in saturated solubility and very fast dissolution rate can be seen, especially below particle sizes of 1 µm. Nano-suspension technology can also be used for drugs, which are insoluble in both water and organic solvents 3. A pharmaceutical nanosuspension is defined as very finely dispersed solid drug particles in an aqueous vehicle for either oral or topical use or parenteral and pulmonary administration. The particle size distribution of the solid particles in nanosuspensions is usually less than one micron with an average particle size ranging between 200 to 600 nm 4-5.
In nanosuspension technology, the drug is maintained in the required crystalline state with reduced particle size, leading to an increased dissolution rate and therefore improved bioavailability. An increase in the dissolution rate of micronized particles (particle size < 10 μm) is related to an increase in the surface area and consequently, the dissolution velocity. Nanosized particles can increase solution velocity and saturation solubility because of the vapor pressure effect 6.
Nanosuspensions are colloidal dispersions of nanosized drug particles stabilized by surfactants. They can also be defined as a biphasic system consisting of pure drug particles dispersed in an aqueous vehicle in which the diameter of the suspended particle is less than 1μm in size. The Nanosuspensions can also be lyophilized or spray dried, and the nanoparticles of a nanosuspension can also be incorporated in a solid matrix 7-12. Nanosuspensions can be used to enhance the solubility of drugs that are poorly soluble in aqueous as well as lipid media. As a result, the rate of flooding of the active compound increases and the maximum plasma level is reached faster (e.g., oral or intravenous [IV] administration of the nanosuspension). This is one of the unique advantages that it has over other approaches for enhancing solubility. It is useful for molecules with poor solubility, poor permeability, or both, which poses a significant challenge for the formulators. The reduced particle size renders the possibility of intravenous administration of poorly soluble drugs without blockade of the blood capillaries.
2. Advantages:
- Suspension can improve the chemical stability of a certain drug.
- The drug in suspension exhibits a higher rate of bioavailability than Other dosage forms bioavailability is in the following the order,
- Solution > Suspension > Capsule > Compressed tablet.
- Duration and onset of action can be controlled.
- Suspension can mask the unpleasant/ bitter taste of drug 13.
3. Disadvantages:
- Physical stability, sedimentation, and compaction can causes problems.
- It is bulky sufficient care must be taken during handling and transport.
- Uniform and accurate dose cannot be achieved unless the suspension is in a proper dose, 13.
4. Application of Nanosuspension:
Bioavailability Enhancement: The poor oral bioavailability of the drug may be due to poor solubility, poor permeability, or poor stability in the Gastrointestinal Tract (GIT). Nanosuspensions resolve the problem of poor bioavailability by solving the twin problems of poor solubility and poor permeability across the membrane. Bioavailability of poorly soluble oleanolic acid, a hepatoprotective agent, was improved using a nanosuspension formulation. The therapeutic effect was significantly enhanced, which indicated higher bioavailability. This was due to the faster dissolution (90% in 20 min) of the lyophilized nanosuspension powder when compared with the dissolution from a coarse powder (15% in 20 min) 17.
Intravenous Administration: The parenteral route of administration provides a quick onset of action, rapid targeting, and reduced dosage of the drug. It is the preferred route for drugs undergoing first-pass metabolism and those that are not absorbed in the GIT or degraded in the GIT. One of the important applications of nanosuspension technology is the formulation of intravenously administered products 13.
Pulmonary Administration: Aqueous Nanosuspensions can be nebulized using mechanical or ultrasonic nebulizers for lung delivery. Because of their small size, it is likely that in each aerosol droplet at least one drug particle is contained, leading to a more uniform distribution of the drug in the lungs. They also increase adhesiveness and thus cause a prolonged residence time 18.
Drug Targeting: Nanosuspensions can also be used for targeting as their surface properties and changing of the stabilizer can easily alter the in vivo behavior. The drug will be uptaken by the mononuclear phagocytic system to allow regional-specific delivery 16.
Mucoadhesion of the Nanoparticles: Nano-particles orally administered in the form of a suspension diffuse into the liquid media and rapidly encounter the mucosal surface. The particles are immobilized at the intestinal surface by an adhesion mechanism referred to as “bio-adhesion.” From this moment on, the concentrated suspension acts as a reservoir of particles, and an adsorption process takes place very rapidly. The direct contact of the particles with the intestinal cells through a bioadhesive phase is the first step before particle absorption 15.
Ophthalmic Drug Delivery: Nanosuspensions could prove to be vital for drugs that exhibit poor solubility in lachrymal fluids. Suspensions offer advantages such as prolonged residence time in a cul-de-sac, which is desirable for most ocular diseases for effective treatment and avoidance of high tonicity created by water-soluble drugs. One example of a nanosuspension intended for ophthalmic controlled delivery was developed as a polymeric nanosuspension of Ibuprofen. This nano-suspension is successfully prepared using Eudragit RS100 by a quasi-emulsion and solvent diffusion method. Nanosuspensions of glucocorticoid drugs; hydrocortisone, prednisolone, and dexamethasone enhance rate, drug absorption, and increase the duration of drug action 18.
TABLE 1: ADVANTAGE OF NANOSUSPENSIONS OVER CONVENTIONAL FORMULATIONS 14
| Route of administration | Disadvantages of conventional formulations | Benefit of nanosuspensions |
| Oral | Slow onset of action/poor absorption | Rapid onset of action/ improved solubility so improved bioavailability |
| Ocular | Lacrimal wash off/low bioavailability | Higher bioavailability/dose consistency |
| Intravenous | Poor dissolution/ non specific Action | Rapid dissolution/tissue targeting |
| Intramuscular Inhalations | Low patient compliance due to pain
Low bioavailability due to low solubility |
Reduced tissue irritation rapid dissolution/ high bioavailability/dose regulation |
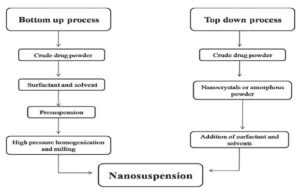
5. Preparation of Nanosuspension: For the preparation of nanosuspension, mostly two methods namely “Bottom-up technology” and “Top-down technology” are used as Fig. 1.19 Bottom-up technology is an assembling method to form nanoparticles from precipitation, micro-emulsion, melt emulsification method and top-down technology involves the disintegration of larger particles into nanoparticles, examples of which are high-pressure homogenization and milling methods 20-21.
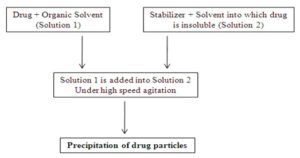
6. Precipitation Method: Precipitation method is a general method used to prepare submicron particles of poorly soluble drugs 22-23. In this method, the drug is dissolved in the solvent, and the solution is mixed with a solvent to which drug is insoluble in the presence of surfactant. Rapid addition of solution to such solvent (generally water) leads to rapid supersaturation of drug in the solution, and formation of ultrafine amorphous or crystalline drug. This method involves nuclei formation and crystal growth, which are mainly dependent on temperature. High nucleation rate and low crystal growth rate are primary requirements for preparing a stable suspension with minimum particle size 24.
7. High-Pressure Homogenization: This technique involve the following three steps: First, drug powders are dispersed in a stabilizer solution to form presuspension; after that, presuspension is homogenized by high-pressure homogenizer at a low pressure sometimes for premilling; and finally homogenized at a high pressure for 10 to 25 cycles until the nanosuspensions are formed with desired size 25.
FIG. 1: APPROACHES FOR PREPARATION OF NANOSUSPENSION
There are two approaches for the guidance of nanosuspension. They're 26
1. Bottom-Up Technology: The period “Bottom-up technology” way that one begins from the molecular degree, and goes by way of molecular organization to the arrangement of a strong molecule. That we are talking about established precipitation approaches by lowering the solvent excellent, for illustration, using pouring the solvent right into a non-solvent or altering the temperature or a combo of each. Precipitation is a classical process in pharmaceutical chemistry and technology 27-28.
Advantages:
- Use of easy and low-cost apparatus and
- Bigger saturation solubility is the advantage for precipitation manner in comparison to different methods of nanosuspension guidance risks 28-30.
Disadvantages: 28-32
- Precipitation system is no more material to medications which are inadequately solvent in fluid and non-watery media. In this system, the medication goals to be dissolvable in at least one dissolvable, which is miscible with non-solvent.
- Prevent crystal progress due to Ostwald ripening being brought about via unique saturation solubilities in the vicinity of otherwise sized particles.
2. Top-Down Technology: the top down technologies include 31-35
- Media milling
- High-pressure homogenization
- Emulsion diffusion method
- Melt emulsification method
FIG. 2: APPROACHES FOR PREPARATION OF NANOSUSPENSION36
TABLE 2: PREPARATIVE TECHNIQUES FOR NANOSUSPENSION WITH MERITS AND DEMERITS 37-38
| Technique | Merits | Demerits |
| Precipitation | Simple process, stable product, low cost of equipment, ease of scale-up | Growing of drug crystals need to be limit by surfactant addition, the drug must be soluble at least in one solvent, narrowly applying space, wide size distribution and potential toxicity of the non-aqueous solvent |
| High-pressure homogenization | Simple technique, general applicability to most drug useful for the formulation of very dilute as well as highly concentrate nanosuspension, aseptic production possible, low risk of the product, contamination ease of scale-up | The high number of homogenization cycle, pre-treatment of micronized drug particles and pre suspending material before subjecting it to homogenization, possible contamination of product could occur from metal ions coming through the wall of the homogenizer |
| Media milling | High flexibility in handling, very few batches to batch variation in particle size, high flexibility in handling large quantities of the drug, ease of scale-up | Possible erosion of material from the milling pearls require milling process for hours to days; prolonged milling may induce the formation of amorphous lead to instability |
| Dry co grinding | Easy process, required short grinding time, no organic solvent | Generation of the residue of milling media |
| Liquid emulsion/microemulsion template | Simple process, small size particles, stable product, high drug solubilization, uniform particle distribution, ease of manufacture | Use of high amount of surfactant and stabilizer, use of hazardous solvent |
| Melt emulsification | Avoidance of organic solvent compared to the solvent diffusion | Formation of a large particle of solvent diffusion |
III. Lipid Emulsion/Microemulsion Template: Another way to produce Nanosuspensions is to use an emulsion which is formed by the conventional method using a partially water-miscible solvent as the dispersed phase. Nanosuspensions are obtained by just diluting the emulsion. Moreover, microemulsions as templates can produce Nano-suspensions. Microemulsions are thermo-dynamically stable and isotropically clear dispersions of two immiscible liquids such as oil and water stabilized by an interfacial film of surfactant and co-surfactant. The drug can be either loaded into the internal phase, or the pre-formed microemulsion can be saturated with the drug by intimate mixing. Suitable dilution of the microemulsion yields the drug nanosuspension. An example of this technique is the griseofulvin nano-suspension, which is prepared by the microemulsion technique using water, butyl lactate, lecithin, and the sodium salt of taurodeoxycholate. The advantages of lipid emulsions as templates for nanosuspension formation are that they are easy to produce by controlling the emulsion droplet and easy for scale-up. However, the use of organic solvents affects the environment, and large amounts of surfactant or stabilizer are required 39.
Melt Emulsification Method: In this method, the drug is dispersed in the aqueous solution of stabilizer and heated above the melting point of the drug and homogenized to give an emulsion. During this process, the sample holder was enwrapped with a heating tape fitted with temperature controller, and the temperature of the emulsion was maintained above the melting point of the drug. The emulsion was then cooled down either slowly to room temperature or on a nice‐bath. The main advantage of melt emulsification technique relative to the solvent diffusion method is total avoidance of organic solvents during the production process 40.
1. Milling Techniques:
Media Milling: Nanosuspensions are produced by using high-shear media mills or pearl mills. The mill consists of a milling chamber, milling shaft, and a recirculation chamber. An aqueous suspension of the drug is then fed into the mill containing small grinding balls/pearls. As these balls rotate at a very high shear rate under controlled temperature, they fly through the grinding jar interior and impact against the sample on the opposite grinding jar wall. The combined forces of friction and impact produce a high degree of particle size reduction. The milling media or balls are made of ceramic-sintered aluminum oxide or zirconium oxide or highly cross-linked polystyrene resin with high abrasion resistance. Planetary ball mills (PM100 and PM200) is one example of equipment that can be used to achieve a grind size below 0.1 μm.
A nanosuspension of Zn-Insulin with a mean particle size of 150 nm was prepared using the wet milling technique. The major drawbacks of this technology include the erosion of balls/pearls that can leave residues as contaminants in the final product, degradation of the thermolabile drugs due to the heat generated during the process and presence of relatively high proportions of particles ≥5 μm.
Advantages:
- Simple technology,
- The low-cost process regarding the milling itself,
- Large-scale production is possible to some extent (batch process).
Disadvantages:
- Potential erosion from the milling material leading to product contamination.
- Duration of the process not being very production friendly.
- Potential growth of germs in the water phase when milling for a long time.
- Time and costs associated with the separation procedure of the milling material from the drug nanoparticle suspension, especially when producing parenteral sterile products 41-44.
FIG. 3: MEDIA MILLING PROCESS
Dry Co-Grinding: Recently, nanosuspensions can be obtained by dry milling techniques. Dry co-grinding can be carried out easily and economically and can be conducted without organic solvents. The co-grinding technique can reduce particles to the submicron level, and a stable amorphous solid can be obtained 45.
2. Supercritical Fluid Method: Supercritical fluid technology can be used to produce nanoparticles from drug solutions. The various methods attempted are a rapid expansion of supercritical solution process (RESS), supercritical anti-solvent process, and precipitation with the compressed anti-solvent process (PCA). The RESS involves expansion of the drug solution in supercritical fluid through a nozzle, which leads to loss of solvent power of the supercritical fluid resulting in precipitation of the drug as fine particles. Cyclosporine nanoparticles prepared in size range of 400-700 nm using this process.
In the PCA method, the drug solution is atomized into a chamber containing compressed CO2. As the solvent is removed, the solution gets supersaturated and thus precipitates as fine crystals. The supercritical antisolvent process uses a supercritical fluid in which a drug is poorly soluble and a solvent for the drug that is also miscible with the supercritical fluid. The drug solution is injected into the supercritical fluid, and the solvent gets extracted by the supercritical fluid, and the drug solution gets supersaturated. The drug is then precipitated as fine crystals. Nanoparticles of griseofulvin, a drug with poor solubility, using this method 46-51.
3. Micro Precipitation – High-Pressure Homogenization (Nanoedge): Nanoedge is a combination of micro precipitation and high-pressure homogenization techniques 52-53. The method includes precipitation of friable materials followed by fragmentation under high shear and thermal energy. The preparation method of nano edge is showing Fig. 4.
FIG. 4: METHOD FOR PREPARATION OF NANOEDGE 54
VII. Nano Jet Technology: This technique, called opposite stream or nanojet technology, uses a chamber where a stream of suspension is divided into two or more parts, which colloid with each other at high pressure. The high shear force produced during the process results in particle size reduction. Equipment using this principle includes the M110L and M110S microfluidizers (Microfluidics).
Nanosuspensions of atovaquone using the microfluidization process. The major disadvantage of this technique is the high number of passes through the microfluidizer and that the product obtained contains a relatively larger fraction of microparticles 55.
VIII. Emulsification-Solvent:
Evaporation Technique: This technique involves preparing a solution of drug followed by its emulsification in another liquid that is a non-solvent for the drug. Evaporation of the solvent leads to precipitation of the drug. Crystal growth and particle aggregation can be controlled by creating high shear forces using a high-speed stirrer 45.
6. Advantages of Nanosuspension:
-
- Enhance the solubility and bioavailability of drugs.
- Suitable for hydrophilic drugs.
- Higher drug loading can be achieved.
- Dose reduction is possible.
- Enhance the physical and chemical stability of drugs.
- Provides a passive drug targeting 56-57
- Most cost effective.
- Useful for poorly soluble drugs.
- Physically more stable than liposomes.
- Provide ease of manufacture and scale up for large scale production.
- Rapid dissolution and tissue targeting.
- Reduction in tissue irritation.
- Higher bioavailability in ocular and inhalational drug delivery 58.
Advantages and Disadvantages of Various Preparation Techniques of Nanosuspension: The following Table 3 shows the advantages and disadvantages of various preparation techniques used in nanosuspension.
TABLE 3: ADVANTAGES AND DISADVANTAGES OF VARIOUS PREPARATION TECHNIQUES OF NANOSUSPENSION
| Method | Advantages | Disadvantages |
| Precipitation 59 | Low need for energy, stable products, and simple process | Narrowly applying space, wide size distribution and potential toxicity of the non-aqueous solvent |
| High-Pressure Homogenization 60 | Widely applying regions, ease of scale-up and little batch to batch variation, narrow size distribution in the final product, allowing aseptic production of nanosuspensions for parenteral administration and flexibility in handling the drug quantity | Pre-treatment of micronized drug particles and pre-suspending materials before subjecting it to homogenization |
| Lipid emulsion/microemulsion template 61 | Low need for energy, stable products, simple process, the small size of particles and uniform particle distribution | high concentration undesired surfactants and residual solvents |
| Milling 61-62 | widely applying regions, ease of scale-up and little batch to batch variation, narrow size distribution in the final product | potential erosion of material from the milling pearls |
| Microprecipitation-high pressure homogenization | much smaller, more uniform and more stable compared to that by the microprecipitation; less mechanical force and energy compared with the high-pressure homogenization | The manufacturing process is complicated |
7. Evaluation of Nanosuspension:
- A) In-vitro Evaluations:
- Particle size and size distribution
- Particle charge (Zeta Potential)
- Crystalline state and morphology
- Saturation solubility and dissolution velocity
B) In-vivo Evaluation:
C) Evaluation for Surface-Modified Nanosuspensions
- Surface hydrophilicity
- Adhesion properties
- Interaction with body proteins
1. Mean Particle Size and Size Distribution: The mean particle size and the width of the particle size distribution (called Polydispersity Index) are determined by Photon Correlation Spectroscopy 63 (PCS). Particle size and polydispersity index (PI) governs the saturation solubility; dissolution velocity and biological performance. It is proved that change in particle size changes saturation solubility and dissolution velocity. PCS measures the particle size in the range of 3 nm - 3 μm only.
PI governs the physical stability of nanosuspension and should be as low as possible for long-term stability (should be close to zero). PCS is a versatile technique but has a low measuring range. In addition to PCS, analysis nanosuspensions are analyzed by Laser Diffractometry (LD). LD measures volume size distribution and measures particles ranging from 0.05-80 μm up to 2000μm. Atomic Force Microscopy 64 is used for visualization of particle shape.
TABLE 4: NANOSUSPENSION TECHNOLOGY OVER OTHER CONVENTIONAL FORMULATIONS TECHNOLOGIES FOR POORLY SOLUBLE DRUGS
| Route of administration | Potential benefits |
| Oral | Rapid onset reduced fed/fasted ratio improved bioavailability |
| Intravenous | Rapid dissolution tissue targeting |
| Ocular | Higher bioavailability more consistent dosing |
| Inhalation | Higher bioavailability more consistent dosing |
| Subcutaneous/ intramuscular | Higher bioavailability rapid onset of reduced tissue irritation |
2. Particle Charge (Zeta Potential): Particle charge determines the stability of nanosuspension. For electrostatically stabilized nanosuspension a minimum zeta potential of ± 30mV and for combined steric and electrostatic stabilization it should be a minimum of ±20mV.
3. Crystalline State and Particle Morphology: Differential Scanning Calorimetry (DSC) determines the crystalline structure. When nanosuspensions are prepared, drug particles get converted to amorphous form; hence, it is essential to measure the extent of amorphous drug generated during the production of nanosuspensions. The X-Ray Diffraction (XRD) is also used for determining the change in physical state and extent of an amorphous drug.
4. Saturation Solubility and Dissolution Velocity: The nanosuspension increase the saturation solubility as well as dissolution velocity. Saturation solubility is compound specific constant depending upon temperature and the properties of dissolution medium. Kelvin equation and the Ostwald-Freundlich equations can explain the increase in saturation solubility 65.
5. In-vivo Evaluation: The establishment of an in-vitro / in-vivo correlation and the monitoring of the in-vivo performance of the drug is an essential part of the study, irrespective of the route, and the delivery system employed. It is of the utmost importance in the case of intravenously injected nanosuspensions since the in-vivo behavior of the drug depends on the organ distribution, which in turn depends on its surface properties, such as surface hydrophobicity and interactions with plasma proteins.
In fact, the qualitative and quantitative composition of the protein absorption pattern observed after the intravenous injection of nanoparticles is recognized as the essential factor for organ distribution. Hence, suitable techniques have to be used to evaluate the surface properties and protein interactions to get an idea of in-vivo behavior. Techniques such as hydrophobic interaction chromatography can be used to determine surface hydrophobicity, whereas 2-D PAGE can be employed for the quantitative and qualitative measurement of protein adsorption after intravenous injection of drug nano-suspensions in animals 66.
8. Formulation of Nanosuspension: 67, 68
TABLE 5: FORMULATION CONSIDERATION FOR NANOSUSPENSION
| Excipient | Function | Example |
| Stabilizers | Wet the drug particles thoroughly, prevent Ostwald’s ripening and agglomeration of nanosuspensions, providing a steric or ionic barrier | Lecithins, poloxamers, polysorbate, Cellulosics, povidones |
| Co-surfactants | Influence phase behavior when microemulsions are used to formulate nanosuspensions | Bile salts, dipotassium glycyrrhizinate, transcutol, glycofurol, Ethanol, isopropanol, |
| Organic Solvent | Pharmaceutically acceptable less hazardous solvent for preparation of the formulation | Methanol, ethanol, chloroform, isopropanol, ethyl acetate, ethyl Formate, butyl lactate, triacetin, propylene carbonate, benzyl alcohol |
| Other Additives | According to the requirement of the route of administration or the properties of the drug moiety | Buffers, salts, polyols, osmogens, cryoprotectant, etc. |
CONCLUSION: Nanosuspensions appears to be a unique and yet commercially viable approach to combating problems such as poor bioavailability that are associated with the delivery of hydrophobic drugs, including those that are poorly soluble in aqueous as well as organic media. The nanosuspension can be proved as a gift as the poorly water-soluble drugs can be easily formulated into nanosuspension. One of the critical problems associated with poorly soluble drugs is too low bioavailability. There is several formulation approaches to resolving the problems of low solubility and low bioavailability. Nanosuspension not only solves the problems of poor solubility and bioavailability but also alters the pharmacokinetics of the drug and thus improves drug safety and efficacy.
Nanosuspensions are chiefly seen as vehicles for administering poorly water-soluble drugs have been largely solved the dissolution problems to improve drug absorption and bioavailability. It has many formulations and therapeutic advantages, such as the simple method of preparation, less requirement of excipients, increased dissolution velocity and saturation solubility, improved adhesion, increases the bioavailability leading to a decrease in the dose and fast-fed variability and ease of large-scale manufacturing. Nanosuspension technology can be combined with traditional dosage forms: tablets, capsules, pellets, and can be used for parenteral products. Many drug delivery and pharmaceutical companies are exploiting this technology to re-examine active ingredients that were abandoned from formulation programs because of their poor solubility. To take advantage of nanosuspension drug delivery, simple formation technologies, and variety applications, nanosuspensions will continue to be of interest as oral formulations and non-oral administration develop in the future.
ACKNOWLEDGEMENT: The author’s thankful to Dr. S. K. Prajapati and Mr. Dilip Kumar Chanchal for his valuable guidance.
CONFLICT OF INTEREST: NIl
REFERENCES:
- Patravale VB: Nanosuspensions: a promising drug delivery strategy. JPP 2004; 56: 827-40.
- Nagaraju P: Nanosuspensions: A promising drug delivery systems. Int J Pharm Sci Nano 2010; 2: 679-84.
- Muller RH: Nanosuspensions for the formulation of poorly soluble drugs. In F Nielloud, Pharma emulsion and suspension. New York 2000; 383-07.
- Nash RA: Suspensions, In: J Swarbrick, JC Boylan (ed), Encyclopedia of Pharmatechnol 2002; 2(3): 2045-32.
- Müller RH and Peters K: Nanosuspensions for the formulation of the poorly soluble drug. Int J Pharm 1998; 160: 229-37.
- Banavath H: Nanosuspension: an attempt to enhance the bioavailability of poorly soluble drugs. Int J of Pharma Sci and Res 2010; 1(9): 1-11.
- Shegokar R and Müller RH: Nanocrystals: Industrially feasible multifunctional formulation technology for poorly soluble actives. Int J of Pharma 2010; 129-39.
- Chingunpituk J: Nanosuspension technology for drug delivery. Walailak J Sci & Tech 2007; 4(2): 139-53.
- Patravale B: Nanosuspensions: a promising drug delivery strategy. J of Pharmacy and Pharmacol 2004; 59: 827-40.
- Prasanna L: Nanosuspension technology: A review. Int J of Pharmacy and Pharma Sci 2010; 2(4): 35-40.
- Xiaohui P: Formulation of nanosuspensions as a new approach for the delivery of poorly soluble drugs. Current Nanoscience 2009; 5: 417-27.
- Bhowmik D: Nanosuspension -A novel approaches in drug delivery system 2013; 1(2): 50-63.
- Liversidge GG and Cundy KC: Particle size reduction for improvement of oral bioavailability of hydrophobic drugs: Absolute oral bioavailability of nanocrystalline danazol in beagle dogs. Int J Pharm 1995; 125: 91-7.
- Bond L: Differential scanning calorimetry and scanning thermal microscopy analysis of pharmaceutical materials. Int J Pharm 2002; 243: 71-82.
- Kayser O: The impact of nanobiotechnology on the development of new drug delivery systems. Curr Pharm Biotech 2005; 6: 3-5.
- Chaudhari SP: Nanosuspension -A novel approaches in drug delivery system. IJPRR 2013; 2(12): 30-39.
- Tripathi JK: Nanotechnology article/nanosuspensions types of nanosuspension methods and various applications 893.html.2011; 12:36: 25.
- Grau MJ: Nanosuspensions of poorly soluble drugs reproducibility of small-scale production. Int J Pharm 2000; 196: 155-7.
- Chingunpituk J: Nanosuspension technology for drug delivery. Walailak J Sci-Tech 2007; 4: 139-53.
- Pu X: Formulation of nanosuspensions as a new approach for the delivery of poorly soluble drugs. Curr Nanosci 2009; 5: 417-27.
- Matteucci ME: Design of potent amorphous drug nanoparticles for rapid generation of highly supersaturated media. Mol Pharm 2007; 4: 782-93.
- Gassmann P: Hydrosols-alternatives for the parenteral application of poorly water-soluble drugs. Eur J Pharm Biopharm 1994; 40: 64-72.
- Myerson AS: HB of Industrial Crystallization 1992: 45-6.
- Bodmeier R and McGinty JM: Solvent selection in the preparation of poly (DL-lactide) microspheres prepared by a solvent evaporation method. Int J Phar 1998; 43: 179-86.
- Liversidge GG and Cundy K: Particle size reduction for improvement of oral bioavailability of hydrophobic drugs: Absolute oral bioavailability of nanocrystalline danazol in beagle dogs. Int J Pharm 1995; 125: 91-7.
- Kondo M: NPC-11 Phase III Trial concerning apnea of prematurity in Japanese neonates: a study of safety, efficacy and pharmacokinetics. Phar Anal Act 2016; 7: 458.
- Garcia AJ and Smith JM: Bile Duct Brushings in a Jaundiced Woman 2015.
- Alvarez AM: Non-communicating mucinous biliary cyst adenoma as a rare cause of jaundice. J Cytol Histol 2015; 6: 369.
- Morin C: Late-onset infections after surgical treatment of spinal deformities in children. J Spine 2015; 4: 262.
- Bhat IH: Clinical profile and outcome in distal gastrointestinal tract obstruction in neonates with special emphasis on the role of colostomy and its complications. Anat Physiol 2016; 6: 222.
- Abbas A: Screening and prevention of transmission of HIV-1 in neonates born to mothers with HIV. Int J Pub Health Safe 2016; 1: 103.
- Susanne B: In-vitro and in-vivo evaluation of a new sublingual tablet system for rapid Oro mucosal absorption using fentanyl citrate as the active substance. European J Pharm Sc 2003; 20: 327-34.
- Nibha KP and Pancholi SS: An overview on a sublingual route for systemic drug delivery. Int J Res Pharm Biomed Sc 2012; 3: 913-23.
- Saito M: High dose octreotide for the treatment of chylo-thorax in three neonates. J Neonatal Biol 2016; 5: 218.
- Strehlow B: A novel micro particulate formulation with allicin in-situ J Phar Drug Deliv Res 2016; 5: 1.
- Grau MJ: Nanosuspensions of poorly soluble drugs reproducibility of small-scale production. Int J Pharm 2000; 196: 155-7.
- Chingunpituk J: Nanosuspension technology for drug delivery. Walailak J Sci-Tech 2007; 4: 139-53.
- Pu X: Formulation of nanosuspensions as a new approach for the delivery of poorly soluble drugs. Curr Nanosci 2009; 5: 417-27.
- Patravale VB: Nanosuspension: a promising drug delivery strategy.J Pharm Pharmacology 2004; 56: 827-40.
- Chaudhari S: IJPRR 2013; 2(12): 30-39.
- Venkatesh T: Nanosuspensions: Ideal approach for the drug delivery of poorly water-soluble drugs. Der Pharmacia Lettre 2011; 3(2): 203-13.
- Yadav GV: Nanosuspension: A promising drug delivery system. Pharmacophore 2012; 3(5): 217-43.
- Pandey S: Nanosuspension: Formulation, characterization and evaluation. Int J of Phar and Bio Sci 2010; 1(2): 1-10.
- Toshi C: A review on nanosuspensions promising drug delivery strategy. Current Pharma R 2012; 3(1): 764-76.
- Aher SS: Int J Curr Pharm Res 2017; 9(3): 19-23.
- Venkatesh T: Nanosuspensions: Ideal approach for the drug delivery of poorly water-soluble drugs. Der Pharmacia Lettre 2011; 3(2): 203-13.
- Paun JS: Nanosuspension: An emerging trend for bioavailability enhancement of poorly soluble drugs. Asian J Pharm Tech 2012; 2(4): 157-68.
- Vaghela A: Nanosuspension technology. Int J of Uni Pharmacy and Life Sci 2012; 2(2): 306-17.
- Bhargavi A: Technical review of nanosuspensions. Int J of Pharmacy & Technol 2011; 3(3): 1503-11.
- Verma KAK: Nanosuspensions: advantages and disadvantages. Ind J of Novel Dru Del 2012; 4(3): 179-88.
- Srinivasa RK: An overview of statins as hypolipidemic drugs. Int J of Pharma Sci and Drug R 2011; 3(3): 178-83.
- Kipp JE: Method for preparing submicron particle suspensions. US Patent2003; 0031719 A1.
- Noyes AA and Whitney WR: The rate of solution of solid substances in their own solutions. J Am Chem Soc 1897; 19: 930-4.
- Hintz RJ and Johnson KC: The effect of particle size distribution on dissolution rate and oral absorption. Int J Pharm 1989; 51: 9-17.
- Dearns R: Atovaquone pharmaceutical compositions. 2000 US Patent US 6018080.
- Ogbalu OK: A new trend of omphalitis complicated with myiasis in neonates of the Niger delta, Nigeria. Epidemiology Sunnyvale 2016; 6: 231.
- Kurt A: Exposure to environmental tobacco smoke during pregnancy restrain the antioxidant response of their neonates. J Neonatal Biol 2016; 5: 210.
- Kavitha VB: Nanosuspension formulation: An improved drug delivery system. An Int J 2014; 4(1): 1-5.
- Bodmeier R and McGinity JM: Solvent selection in the preparation of poly (DL-lactide) microspheres prepared by solvent evaporation method. Int J Pharm 1998; 43: 179-86.
- Liversidge GG and Cundy KC: Particle size reduction for improvement of oral bioavailability of hydrophobic drugs: Absolute oral bioavailability of nanocrystalline danazol in beagle dogs. Int J Pharm 1995; 125: 91-7.
- Patravale VB: Nanosuspension: a promising drug delivery strategy. J Pharm Pharmacology 2004; 56: 827-40.
- Itoh K: Chem Pharm Bull 2003; 51: 171-4.
- Muller RH: Nanosuspensions as particulate drug formulations in therapy Rationale for development and what we can expect for the future. Ad Drug Del Rev 2001; 47: 3‐19.
- Muller BW and Muller RH: Particle size analysis of latex suspensions and microemulsions by Photon Correlation Spectroscopy. J Pharm Sci 1984; 73: 915‐18.
- Bhowmik D: Nanosuspension -A novel approaches in drug delivery system. ICJ 2013; 1(12): 50-63.
- Patel HM:Nanosuspension: A novel approach to enhance the solubility of poorly water-soluble drugs - A review. J Home Page 2016; 5(2): 22-39.
- Kavitha VB: Nanosuspension formulation: An improved drug delivery system. An Int J 2014; 4(1): 1-5.
- Mukesh D: Nanosuspension technology for solubilizing poorly soluble drugs. Int J Drug Dev & Res 2012; 4(4): 40-49.
How to cite this article:
Afrin, Prajapati SK, Chanchal DK, Agnihotri N, Tiwari S, Singh D and Ali C: Nanosuspensions: a promising drug delivery strategy. Int J Life Sci & Rev 2019; 5(6): 82-91. doi: 10.13040/IJPSR.0975-8232.IJLSR.5(6).82-91.
All © 2015 are reserved by International Journal of Life Sciences and Review. This Journal licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.
Article Information
1
82-91
772
1172
English
IJLSR
Afrin *, S. K. Prajapati, D. K. Chanchal, N. Agnihotri, S. Tiwari, D. Singh and C. Ali
Department of Pharmacy, Institute of Pharmacy, Bundelkhand University Jhansi, Uttar Pradesh, India.
afreen.khan01995@gmail.com
30 April 2019
23 May 2019
21 June 2019
10.13040/IJPSR.0975-8232.IJLSR.5(6).82-91
30 June 2019