NUTRACEUTICAL FORMULATION OF GROUNDNUT OIL CAKE AND STUDY OF DRUG RELEASE AT VARIOUS pH
HTML Full TextNUTRACEUTICAL FORMULATION OF GROUNDNUT OIL CAKE AND STUDY OF DRUG RELEASE AT VARIOUS pH
Deepika S. Agrawal, Devanand K. Patil, Rahul R. Mahire and Dhananjay H. More *
School of Chemical Science, North Maharashtra University, Jalgaon - 425001, Maharashtra, India.
ABSTRACT: The drug discovery and computational advances have helped chemists to develop drugs having utility for specific functional applications along with nutritional aspects. This enables researchers to design formulations and synthesized proper medicines as nutrients. Preparation of nutraceutical formulation that to a veterinary formulation is one of the challenging tasks. Various types of formulations have been devised for sustainable release, easier dissolution with no side effects, and target specific delivery of the chosen drug. The oral administration of veterinary drugs is one of the difficult task, particularly the formulation with the disagreeable smell, taste as well as low nutritional value. Considering these, it was planned to develop easy and likable preparation of nutraceutical formulation of the veterinary drug for easy oral delivery, comprising sustain release of the drug. In the present report, discussion of the formulation of ketoprofen in which veterinary drug is incorporated along with the groundnut or cottonseed oil cake through adsorption method of formulation preparation. Such a formulation can be assimilated effectively by ruminants as the oil cakes are liked by animals, as well contains a very good amount of fat-producing nutrients. The prepared formulation was characterized by FTIR, FESEM, TGA techniques, and the studies related to the release rate of the drug were made using the spectrophotometric technique. The detail about the formulation and other related studies are presented and discussed.
Keywords: Veterinary drug (Ketoprofen), Groundnut oil cake, Drug delivery system, Drug release study, Kinetics
INTRODUCTION: The fields of drug discovery and simulations have emerged as sciences of applications. In the 20th century, a large part of the research had been focused on drug development and its formulation designing using nutrients, as far as human consumption is concerned 1. It is felt that in developing countries, similar awareness about the animal nutritional sources along with active pharmaceutical ingredient (API) that is nutraceuticals for a live veterinary stock is lacking.
Similarly, there is a need for the development of such formulation which has not been attended seriously for the drugs used for veterinary and preventions of diseases for veterinary animals.
Development of nutraceuticals, along with drug delivery property is essential. Wide variety of drug delivery formulations have been reported2 using different methods like as adsorptions to solid carriers, spray drying 3, spray cooling, melt extrusion, nanoparticles technology, supercritical fluid-based methods, etc. 4 solid dispersion 5, 6 encapsulation 7-10, etc. Amongst these methods, the adsorption technique is easy and involves the simple addition of liquid formulation onto carriers. Bermudez et al. reviewed many different drug delivery systems for veterinary used 11. Formulation of different drugs such as celecoxib 12, tarazepide 13, aspirin 14, paracetamol 15, ibuprofen16, etc. are reported, some of which are veterinary drugs.
Moreover, there are some nutraceutical been reported for humans 17-19, but veterinary nutraceutical formulation for ruminant has not been attended till the date. Therefore, for every living species in this world, formulation with a good nutritive value should be available to overcome the nutrition in the formulation. Further formulation of veterinary drugs becomes necessary as farmers face lot many difficulties, particularly in oral drug administration for their pet animals. Also, animals hesitate to swallow the drug, especially with big size and bitter taste. The literature survey revealed that such a subject is not attended in details or has not been given due attention. Therefore, focusing on such kind of formulations of drugs particularly for animals, having usability for milk, fertilizer and nutritional assets as food (e.g., chickens, cows, pigs, etc.) are necessary to overcome above-said difficulties.
In the present work, an attempt is made to provide nutritional value to the veterinary drug using groundnut oil cake or cottonseed oil cake, which is the loving animal feed especially of ruminants like cow, buffaloes, Bullock, etc. Particularly in India groundnut oil cake is most widely preferred by cattle and constitutes as animal feed due to its exceptional quality and nutritional values. The natural groundnut oil cake offers wholesome nutrition to cattle’s and other animals as a source of proteins, fats, and oils, moisture, fibers, etc. Therefore formulation preparation using such a cake can serve two purposes, nutritive value and easy assimilation by animals. Moreover, it will be preferred for oral drug administration by farmers and doctors for the animals as the formulation is prepared by likable animal food, i.e. incorporation of the drug in oil cake using adsorption method.
Antibacterials, nonsteroidal anti-inflammatory drugs, β-blockers, lipid regulators, hormones, antiepileptics, and antiseptics are the most widely used medical and veterinary drugs in present scenario 20, 21. Oral drug ketoprofen a nonsteroidal anti-inflammatory drug 22, 23 is used in the present formulation as it is commonly used the veterinary drug.
Ketoprofen is (RS)2-(3-benzoylphenyl)-propionic acid with chemical formula C16H14O3 and is one of the propionic acid class of nonsteroidal anti-inflammatory drug (NSAID) with analgesic and antipyretic effects 24. It acts by inhibiting prostaglandin production in the body. It is used as a mild painkiller in smaller animals, generally following surgical procedures. It is most commonly used for musculoskeletal pain, joint problems, soft tissue injury, in laminitis, control fevers and prevents endotoxemia. The protein binding efficiency of ketoprofen is 99%, its half-life period is 2-2.5 h while its log P value is 3.12 and log S value is -3.7 (log P and log S indicates partition coefficient and aqueous solubility respectively).
Ketoprofen consists of such important characteristic features, thus it was used for the preparation of formulation and related studies. Also, the health benefits of foods to consumers (farmer), as nutraceuticals are one of the important applications of present work which supplies nutrition as well as a drug to ruminant animals.
After successful preparation of veterinary formulation for ruminants using oil cake, it was characterized by FTIR, FESEM, TGA, etc. The drug release study and its kinetics have been made using UV-Vis Spectrophotometry.
EXPERIMENTAL:
MATERIAL AND METHOD:
Material: Ketoprofen (API) was obtained as a gift sample from Enaltec Labs Private Ltd., (purity >99%). Fresh groundnut oil cake was purchased from the market. Absolute ethanol was purchased from S D Fine-Chem Limited, Mumbai, India.
Methods:
Preparation of Formulation: Groundnut oil cake was crushed into a fine powder using Mortar and Pestle. 5g of powder of oil cake was taken in a Petri plate. To crushed powder, solution of the drug (1g in 10ml ethanol) was mixed. The drug was found to get adsorbed on an oil cake. The obtained precipitate in a Petri plate was dried at room temperature (~30 ºC) up to till the solvent gets completely evaporated from oil cake. Subsequently, oil cake powder containing drug was pressed using a pressing/molding machine (30-35 ºC) to obtain oil cake again. This pressed oil cake was used for further studies.
Preparation of Sample for Drug Release Study: Drug release in-vitro study was carried out as per available literature 27 (Saravanan et al., 2011) at different pH using aqueous buffer solutions at ambient temperature. The acidic buffer solutions containing ~0.1N and 0.01N HCl having pH=1.2 and 2 which resembles the physiological environment of empty stomach fluid and buffer solutions are having pH=6.8 similar to the pH of the intestine and filled stomach fluid. These solutions were used to carry out drug release study. Prepared formulation (5g oil cake + 1g drug) was poured into 500 ml of each pH solutions. Nearly 5-10 ml of solution was collected with suitable time intervals, and the same volume was replaced by the buffer solutions. Collected samples were suitably diluted using respective buffer solutions. After successive sample collections and dilutions, UV Spectra were recorded for each collected samples and using the maximum absorbance values at a different time interval from the recorded spectra, the kinetics of release study was studied and analyzed.
Characterization:
Fourier Transform Infrared (FTIR) spectroscopy: The FTIR spectral measurements were made at ambient temperature on Perkin Elmer (spectrum one) spectrophotometer using nujol mull technique.
Field Emission Scanning Electron Microscope (FESEM): The prepared formulation was observed for their morphological study under scanning electron microscopy using a thin layer of samples coated with gold on a FESEM (FESEM, HITACHI High Technologies Corporation, Japan, Model No. S-4800-II) with an applied voltage of 15 kV.
Thermogravimetric Analysis (TGA): Thermal respond for the prepared formulation was studied by the thermogravimetric analysis (Model - TGA 4000, PerkinElmer, USA) in the range of 90–the 690 ºC temperature at a heating rate of 20 ºC/min in the presence of N2 as an inert atmosphere.
UV-VIS Spectrophotometer: Drug release study was carried out using double beam UV-VIS Spectrophotometer (Chemito, Spectrascan UV 2700).
Chemical Analysis: Iodine Value of the prepared formulation was calculated by Wij’s method (ASTM D5768-02).
RESULTS AND DISCUSSION:
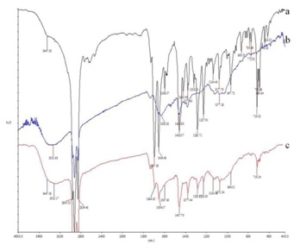
FTIR Spectroscopy: The spectral trace of FTIR analysis of the prepared formulation incorporated with drug and oil cake are depicted in Fig. 1. FTIR spectra of drug and formulation both show absorption bands at 3447 cm−1 and 1694 cm-1 corresponding to OH and C=O stretching frequencies of the acidic group as well as 1654 cm-1 (stretching) of the ketonic group present in the drug molecule. The presence of these specific functional groups of ketoprofen in the formulation exhibits the formation of the targeted formulation. Further the bands at 1285 cm−1 and 1377 cm−1 assign to C-O bond of the acid and CH3 bending vibrations respectively.
FIG. 1: FTIR SPECTRAL ANALYSIS OF DRUG AND OIL CAKE. a- drug (ketoprofen), b- groundnut oil cake, c- formulation
ESEM Images of Formulation: The FESEM study revealed that the surface of the cake is somewhat smooth. The cake with drug shows surface on which rod-shaped particles are present Fig. 2b. The rod-shaped particles are of ketoprofen. The shape of drug particles has been confirmed by a literature SEM image 25. Thus, SEM images confirm adsorption of the drug on groundnut oil cake, the i.e. drug gets settled on oil cake surface due to adsorption involving van der-Waals forces of attraction.
FIG. 2: FESEM IMAGES OF UNLOADED, DRUG, AND LOADED SAMPLES
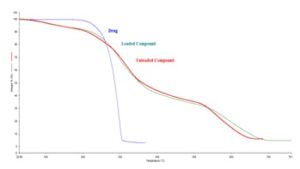
Thermogravimetric Analysis (TGA): Thermogram of the masked drug sample is shown in Fig. 3. TGA curves of the sample showed the involvement of three degradation steps. The first step of the degradation occurs in the temperature range of 94 - 210 °C and degradation resulted into 7.246 % weight loss, which may be attributed to the loss of moistures and volatile impurities present in the formulation. The second step of degradation occurred between 210 and 530 °C temperature where degradation is found up to 60% weight loss. At this time, degradations would have taken place due to the decomposition of other materials present in the composition of oil cake. The last, third step of degradation is seen in the temperature range of 540 - 720 °C with a decomposition of 26 weight %. The third step of degradation may be the result of decomposition of the hydrocarbon chains of the oil and fats present in oil cake. Thus thermal stability of a present formulation is found to be very good, below about 100 °C.
FIG. 3: TGA THERMOGRAPHS
Determination of Iodine Value: Iodine value was determined by Wij’s Method, to evaluate whether there is a reaction taking place between adsorbed drug and groundnut oil cake. The calculated iodine values are collected in Table 1. It was observed that the values of unloaded and loaded drug samples of oil cake were nearly the same, indicating that there is no chemical reaction between groundnut oil cake and drug. This concluded the assimilation of drug molecules with groundnut oil cake takes place with the help of adsorption phenomena at the surface while this adsorption may be due to Vander-Waals forces.
TABLE 1: IODINE VALUES
| Unloaded Drug Sample (groundnut oil cake) | Loaded Drug Sample
(groundnut oil cake + ketoprofen) |
| 7.1 | 7.5 |
Drug Release Study: Drug release study was carried out using solvents of (~0.1N HCl) pH=1.2, (0.01N HCl) pH=2, and buffer solution of pH=6.8, as these are similar to the pH of different parts of the digestive tract of the ruminants. After solution preparation and sample collection, UV spectra were recorded for each sample and are presented in Fig. 4a, 4b, and 4c, respectively. The wavelength of maximum absorption for ketoprofen is found to be at ~258nm which is in agreement with the literature data proposed by Chawla, et al. 26 It was observed that absorbance increases as a function of time Fig. 4a, 4b, and 4c. The trend of curves suggests that the concentration of drug slowly increases with increase in time duration at a given pH. These observations signify that sustained release of drug has been achieved by cake formulation.
Also from the release pattern, it was observed that in lower pH solution (pH=1.2) release was up to 6 hours but as pH gets changed from 1.2 to 2, release time interval increases up till 8 h. Further, as pH is increased to 6.8, the release time also gets increased till 9 h. All these mean that as pH value increases, i.e. it changes from acidic pH to neutral pH. The release time interval of the drug from the oil cake formulation gets prolonged.
Kinetics of Drug Release Study: By studying the plot of the recorded UV-visible absorption profile for release of drug molecules/ions at different pH (varying the acidity/basicity ratio) solutions, one can analyze the kinetics of the drug release. Its related calculations are depicted in Table 2. It was assumed that the drug release follows the first-order kinetics and this was confirmed by making a graph of log ( ) verses time Fig. 5. Where refers to absorption values (optical density / extinction coefficient) at infinite time, at tia me ‘t’ and initially respectively. The Fig. 5a, 5b and 5c incorporate these and reveals, a good least square analysis fit having a correlation factor of 0.94 ±0.02 for all the variations made using different pH solutions. Thus it was satisfying from the Figures to confirm the first order drug release kinetic phenomenon. The equation used for the calculation of first-order velocity constant K is given below.
Where “t” is the time in hours.
It is to be noted from Fig. (5a, 5b, and 5c) that velocity constant for the release of drug is highest at pH=2
TABLE 2: MATHEMATICAL VALUES FOR KINETIC STUDY
| pH=1.2 | pH=2 | pH=6.8 | ||||||||
| Time
(hrs) |
Abs. | Time(hrs) | Abs. | Time(hrs) | Abs. | |||||
| 0 | 0.256=Ao | -- | 0 | 0.094=Ao | -- | 0 | 0.11=Ao | -- | ||
| 1 | 0.37 | 0.0573 | 1 | 0.475 | 0.2191 | 1 | 0.166 | 0.0217 | ||
| 2 | 0.443 | 0.0983 | 2 | 0.684 | 0.4103 | 2 | 0.249 | 0.0561 | ||
| 3 | 0.522 | 0.148 | 3 | 0.686 | 0.415 | 3 | 0.533 | 0.2001 | ||
| 4 | 0.588 | 0.194 | 4 | 0.856 | 0.682 | 4 | 0.637 | 0.2675 | ||
| 5 | 0.726 | 0.3096 | 5 | 0.905 | 0.804 | 5 | 0.714 | 0.3252 | ||
| 6 | 1.178=A∞ | -- | 6 | 0.95 | 0.9578 | 6 | 0.725 | 0.3341 | ||
| 7 | 1.006 | 1.2842 | 7 | 0.839 | 0.439 | |||||
| 8 | 1.053=A∞ | -- | 8 | 0.841 | 0.4411 | |||||
| 9 | 1.256=A∞ | -- | ||||||||
| Velocity constant
K (0.0549) |
Velocity constant K
(0.0549) |
Velocity constant K
(0.0549) |
CONCLUSION: In the present report, nutraceuticals formulation is prepared using animal liking food for ruminants by the adsorption method. The formulation has pharmaceutical value because it can accomplish many different requirements and satisfy various clinical needs, to deliver the drug at the right moment, at the right place, and an adequate concentration in terms of therapeutic efficacy and compliance.
The study is promising for animal nutrition as well as for easy assimilation of the drug. Moreover, drug release study revealed that formulation could be used for sustained drug delivery as it shows first order kinetics.
ACKNOWLEDGEMENT: The Authors are grateful to Prof. K. J. Patil for his valuable suggestions and Enaltec Laboratories Mumbai for the gift of a sample of ketoprofen. The Author (DSA) is also grateful to UGC, New Delhi for BSR fellowship.
CONFLICT OF INTEREST: Nil
REFERENCES:
- McClements DJ and Xiao H: Excipient foods: designing food matrices that improve the oral bioavailability of pharmaceuticals and nutraceuticals. Food and function 2014; 5: 1320-33.
- Díaz A, Saxena V, González J, David A, Casañas B, Carpenter C, Batteas JD, Colon JL, Abraham C and Hussain MD: Zirconium phosphate nano-platelets: a novel A platform for drug delivery in cancer therapy. Chemical Communications 2012; 48: 1754-56.
- Thiele J, Windbergs M, Abate AR, Trebbin M, Shum HC, Förster S and Weitz DA: Early development drug formulation on a chip: Fabrication of nanoparticles using a microfluidic spray dryer. Lab on a Chip 2011; 11: 2362-68.
- Bhagwat and D’Souza JI: Formulation and evaluation of solid self-micro emulsifying drug delivery system using aerosil 200 as a solid carrier. International Current Pharmaceutical Journal 2012; 1: 414-19.
- Larson AB and Banker GS: Attainment of highly uniform solid drug dispersions employing molecular scale drug entrapment in polymeric latices. Journal of Pharmaceutical Sciences 1976; 6: 838-43.
- Leuner C and Dressman J: Improving drug solubility for oral delivery using solid dispersions. European journal of Pharmaceutics and Biopharmaceutics 2000; 50: 47-60.
- Dördelmann G, Kozlova D, Karczewski S, Lizio R, Knauer S and Epple M: Calcium phosphate increases the encapsulation efficiency of hydrophilic drugs (proteins, nucleic acids) into poly (d, l-lactide-co-glycolide acid) nanoparticles for intracellular delivery. Journal of Materials Chemistry B 2014; 2: 7250-59.
- Anton N, Bally F, Serra CA, Ali A, Arntz Y, Mely Y, Zhao M, Marchioni E, Jakhmola A and Vandamme TF: A new microfluidic setup for precise control of the polymer nanoprecipitation process and lipophilic drug encapsulation. Soft Matter 2012; 8: 10628-635.
- Tan MX and Danquah MK: Drug and Protein Encapsulation by Emulsification: Technology Enhancement Using Foam Formulations. Chemical Engineering and Technology2012; 35: 618-26.
- Misic Z, Muffler K, Sydow G and Kuentz M: Novel starch‐based PVA thermoplastic capsules for hydrophilic lipid‐based formulations. Journal of Pharmaceutical Sciences 2012; 1: 4516-28.
- Bermudez JM, Cid AG, Ramírez‐Rigo MV, Quinteros D, Simonazzi A, Sánchez Bruni S and Palma S: Challenges and opportunities in polymer technology applied to veterinary medicine. Journal of Veterinary Pharmacology and Therapeutics 2014; 37: 105-24.
- Nguyen TH, Tan A, Santos L, Ngo D, Edwards GA, Porter CJ, Prestidge CA and Boyd BJ: Silica–lipid hybrid (SLH) formulations enhance the oral bioavailability and efficacy of celecoxib: An in-vivo Journal of Controlled Release 2013; 167: 85-91.
- Jacobs C, Kayser O and Müller RH: Nanosuspensions as a new approach for the formulation for the poorly soluble drug tarazepide. International Journal of Pharmaceutics 2000; 196: 161-64.
- Muir N. Nichols JD, Stillings MR and Sykes J: Comparative bioavailability of aspirin and paracetamol following single dose administration of soluble and plain tablets. Current medical Research and Opinion 1997; 13: 491-500.
- Kubo W, Miyazaki S and Attwoodv D: Oral sustained delivery of paracetamol from in situ-gelling gellan and sodium alginate formulations. International Journal of Pharmaceutics 2003; 258: 55-64.
- Chen H, Chang X, Du D, Li J, Xu H and Yang X: Microemulsion-based hydrogel formulation of ibuprofen for topical delivery. International journal of Pharmaceutics 2006; 315: 52-58.
- Chen L, Remondetto GE and Subirade M: Food protein-based materials as nutraceutical delivery systems. Trends in Food Science and Technology 2006; 17: 272-83.
- Duranti M: Grain legume proteins and nutraceutical properties. Fitoterapia 2006; 77: 67-82.
- Farrar JL, Hartle DK, Hargrove JL and Greenspan P: A novel nutraceutical property of select sorghum (Sorghum bicolor) brans: inhibition of protein glycation. Phytotherapy Research 2008; 22: 1052-56.
- Azzouz A, Jurado-Sánchez B, Souhail B and Ballesteros E: Simultaneous determination of 20 pharmacologically active substances in cow's milk, goat's milk, and human breast milk by gas chromatography-mass spectrometry. Journal of Agricultural and Food Chemistry 2011; 59: 5125-32.
- Baynes R, Riviere J, Franz T, Monteiro‐Riviere N, Lehman P, Peyrou M, and Toutain PL: Challenges are obtaining a biowaiver for topical veterinary dosage forms. Journal of Veterinary Pharmacology and Therapeutics 2012; 35: 103-14.
- Mahire RR, Agrawal DS, Patil DK and More DH: Fabrication of organogels achieved by prodrug-based organogelators of ketoprofen. RSC Advances 2014; 4: 33286-291.
- Shohin, IE, Kulinich JI, Ramenskaya GV, Abrahamsson B, Kopp S, Langguth PPolli JE, Shah VP, Groot DW, Barends DM and Dressman JB: Biowaiver monographs for immediate‐release solid oral dosage forms: Ketoprofen. Journal of Pharmaceutical Sciences 2012; 101: 3593-03.
- Kantor TG: Ketoprofen: a review of its pharmacologic and clinical properties. Pharmacotherapy: The Journal of Human Pharmacology and Drug Therapy1986; 6: 93-02.
- Dixit M, Kulkarni PK, Shivakumar A and Kini AG: Preparation and characterization of spherical agglomerates of ketoprofen by neutralization method. Int J Pharm Bio Sci 2010; 1: 395-06.
- Chawla S, Ghosh S, Sihorkar V, Nellore R, Kumar TR, and Srinivas NR: High‐performance liquid chromatography method development and validation for simultaneous determination of five model compounds, antipyrine, metoprolol, ketoprofen, furosemide and phenol red, as a tool for the standardization of rat in situ intestinal permeability studies using timed wavelength detection. Biomedical Chromatography 2006; 20: 349-57.
- Saravanan M, Bhaskar K, Maharajan G and Pillai KS: Development of gelatin microspheres loaded with diclofenac sodium for intra-articular administration. Journal of Drug Targeting 2011; 19: 96-03.
How to cite this article:
Agrawal DS, Patil DK, Mahire RR and More DH: nutraceutical formulation of groundnut oil cake and study of drug release at various pH. Int J Life Sci & Rev 2016; 2(2): 29-36. doi: 10.13040/IJPSR.0975-8232.IJLSR.2(2).29-36.
All © 2015 are reserved by International Journal of Life Sciences and Review. This Journal licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.
Article Information
3
29-36
753
1764
English
IJLSR
D. S. Agrawal, D. K. Patil, R. R. Mahire and D. H. More *
School of Chemical Science, North Maharashtra University, Jalgaon, Maharashtra, India.
dhmore@rediffmail.com
10 December 2015
05 February 2016
24 February 2016
10.13040/IJPSR.0975-8232.IJLSR.2(2).29-36
29 February 2016