SIMULTANEOUS ESTIMATION OF IBUPROFEN AND CARISOPRODOL IN SYNTHETIC MIXTURE BY HPTLC METHOD
HTML Full TextSIMULTANEOUS ESTIMATION OF IBUPROFEN AND CARISOPRODOL IN SYNTHETIC MIXTURE BY HPTLC METHOD
Chandani Patel, Laxman Prajapati *, Amit Joshi and Mohammadali Kharodiya
Shri B. M. Shah College of Pharmaceutical Education and Research, Modasa - 383315, Gujarat, India.
ABSTRACT: A simple, accurate, and precise high-performance thin layer chromatography method for the simultaneous estimation of ibuprofen and carisoprodol in the marketed formulation was developed. The determination was carried on silica gel 60 GF254 HPTLC plates using a mobile phase n-butanol: glacial acetic acid: Acetone (5:2:3 v/v/v). The absorbance of the spots was measured by densitometry at 254 nm. The Retention Factor (Rf) was found to be 0.87 for ibuprofen and 0.77 for carisoprodol. Ibuprofen and carisoprodol showed a linear response in the concentration range 60-180 µg/band and 35-78.75µg/band, respectively. The correlation coefficient (r2) for ibuprofen and carisoprodol was found to be 0.9992 and 0.9997, respectively. The result of the analysis has been validated statistically and by recovery studies. The percentage recoveries obtained for ibuprofen and carisoprodol ranges from 101.92 and 100.46 respectively. The proposed HPTLC method can be applied for the quantitative determination of carisoprodol and ibuprofen in bulk and drug formulation.
Keywords: HPTLC, Ibuprofen, Carisoprodol, 1, 2-Naphthoquinone-4-sulfonic acid sodium salt, Simultaneous
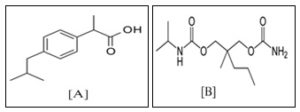
INTRODUCTION: Ibuprofen is an NSAIDs drug which has non-narcotic analgesic with anti-inflammatory and antipyretic activity Fig. 1. The Ibuprofen inhibits the production of prostaglandins by non-selective inhibition of COX (cyclo-oxygenase). It is effective for pain of mild to moderate intensity, including musculoskeletal and postoperative pain, and osteo and inflammatory arthritis. Unlike opioids, they have the advantage of not causing dependence 1, 2. Ibuprofen is a widely used drug alone and with its combination.
Ibuprofen is official in Indian Pharmacopoeia, British Pharmacopoeia, United State Pharmacopoeia 3, 4, 5, 6, 7. Carisoprodol is a dicarbamate, centrally acting, oral skeletal muscle relaxant Fig. 1. It may be used in various acute, painful musculoskeletal conditions, such as muscle strains and back pain. The mechanism of action of carisoprodol in relieving acute muscle spasm of local origin has not been identified, but may be related to its sedative properties 1, 2.
FIG. 1: CHEMICAL STRUCTURE OF (A) IBUPROFEN AND (B) CARISOPRODOL
Carisoprodol is official in Indian Pharmacopeia, British Pharmacopoeia, and United state Pharmacopoeia 3-7. The literature survey reports many analytical methods like spectrophotometry, chromatography of ibuprofen alone or in combination with other drugs 8-14. Literature survey revealed many analytical methods for the determination of carisopropdol alone or in combination with some other drugs 15-22.
Only two UV-spectrophotometric methods have been reported for ibuprofen and carisoprodol in combination 23-24. In the present work, the HPTLC method for simultaneous estimation of ibuprofen (IBU) and carisoprodol (CAR) in the synthetic mixture was developed. The method was validated according to the ICH Q2 (R1) guidelines 23. As carisoprodol does not have any chromophrore, the present work involves derivatization of carisoprodol with 1,2 naphthoquinone 4- sulphonic acid sodium salt.
MATERIALS AND METHODS:
Apparatus: The weighing of all chemical was carried out on the Electronic balance (Shimadzu analytical balance Model AX 20). Chromatographic separation of drugs was performed on TLC plates precoated with silica gel 60/UV254 (10×10 cm and 20 × 10 cm with 250 mm layer thickness). The samples were applied onto the plates as a band with a width of 5 mm using Camag 100 µl sample syringe (Hamilton) with an applicator (Linomat-V, Camag). Linear ascending development was carried out in a twin trough glass chamber (10 × 10 cm, 20 × 10 cm). Densitometry scanning was performed using the TLC scanner (Camag) and operated by software (Wincats).
Reagent and Solution: Carisopodrol and ibuprofen were kindly gifted from Centurian Laboratory, Vadodara (India). All the chemical and reagent were of A.R grade and purchased from Oxford Laboratory. The alkaline borate buffer solutions (pH 9) were made by dissolving 3.2 gm boric acid in 500 mL water and adjusted to the desired pH with 2 M NaOH solution. A stock solution of 1,2 naphthoquinone 4- sulphonic acid sodium salt was freshly prepared by dissolving 5 mg in 100 mL distilled water and stored in the dark (a flask coated with aluminium foil) at room temperature.
Optimization of Reaction Condition: A series of experiments were conducted to established optimum analytical conditions for the reaction of carisoprodol with 1,2 naphthoquinone 4-sulphonic acid sodium salt. The parameter optimized was on all the studies carisoprodol by altering each variable in turn while keeping the other constant.
A) Effect of NQS Concentration and Volume: The effect of NQS concentration was investigated using 2 ml of different concentration of the reagent in the range 30-60 µg/ml. by applying different volume (0.5 ml to 3 ml) of the same concentration.
B) Effect of Heating Temperature and Time: Influence of different heating temperature and incubation time were studied using a thermostat water bath. The effect of different heating temperature on the sensitivity of the method was studied for 5-25 min heating temperature.
C) Effect of Alkaline Media (pH): The effect of pH was investigated using borate buffer of pH 8, 8.5, 9, 9.5, and 10.
Chromatographic Condition: The analysis was carried out by HPTLC using n-butanol: Glacial acetic acid: Acetone (5:2: 3 v/v/v) as a mobile phase and silica gel 60GF 254 HPTLC plates (10×10cm) as a stationary phase.
TABLE 1: CHROMATOGRAPHIC CONDITION
| Condition parameter | Optimized |
| Stationary phase: | Pre-coated Silica gel G60 F aluminum Sheets 10×10 cm,
layer Thickness 0.2 mm |
| Activation: | TLC plates prewashed with methanol and activated in Oven
at 50 °C for 5min |
| Mobile phase: | n-butanol: glacial acetic acid: acetone (5:2:3 v/v) |
| Chamber saturation time | 20 mins. |
| Temperature | Room temperature |
| Band width: | 6 mm |
| Distance between two tracks: | 14 mm |
| Spraying rate | 10 sec/μL |
| Slit dimension: | 6 × 0.45 mm |
| Wavelength of detection | 254 nm |
| Lamp: | Deuterium |
| Measurement mode: | Absorption |
Sample was applied on HPTLC plates as 6 mm bands, by means of camag lino mat v automatic sample applicator fitted with 100 µl Hamilton syringe with the nitrogen flow the plate was developed in Camag twin-trough glass chamber previously saturated for 15 min after development to a distance of 14 cm dried in hot air oven and scanned at 254 nm by means of Camag TLC scanner Table 1.
Preparation of Standard Stock Solution:
A) Carisoprodol Standard Stock Solution (100000 µg/ml): Accurately weighed 1000 mg CAR reference standard was transferred to 10 ml volumetric flask individually, and was dissolved in a minimum quantity of Methanol. The volume was made up to the mark with methanol.
B) Ibuprofen Standard Stock Solution (100000 µg/ml): Accurately weighed 1000 mg IBU reference standard was transferred to 10 ml volumetric flask individually, and was dissolved in a minimum quantity of methanol. The volume was made up to the mark with methanol.
C) Preparation of Working Standard Solution: Stock solution of carisoprodol (1.75 ml) and stock solution of IBU (4 ml) transferred to 10 ml volumetric flask and add 2.5 ml of reagent, 1 ml borate buffer heated at 70 ºC temperature for 20 minutes, cool at room temperature. Further, dilute with borate buffer (17500 µg/ml and 40000 µg/ml).
Preparation of Calibration Curve: To obtain a calibration curve, working standard solution of different range from were applied by Hamilton syringe with the help of Linomet V applicator on TLC plate.
Preparation of Sample Solution: An accurately weighed synthetic mixture 175 mg carisoprodol and 400 mg of ibuprofen was transferred to a 10 ml volumetric flask, dissolved in 4 ml methanol and diluted with methanol. The solution was filtered through Whatman filter paper. 1 ml of filtrate (1750 µg ml-1 Carisoprodol and 4000 µg ml-1 ibuprofen) from stock solution was transferred to 10 ml volumetric flask.
To this solution, 2.5 ml of reagent and 1ml borate buffer were added. The solution heated at 70 ºC temperature for 20 minutes, cooled at room temperature and diluted with borate buffer. The resultant solution was found to contain carisoprodol (175 µg/ml) and ibuprofen (400 µg/ml). 20 μL of this solution was applied on a TLC plate followed by development and scanning at 254 nm. The analysis was repeated for six times.
Method Validation: The repeatability of the method was confirmed by the mixture analysis, repeated for six times with the same concentration. The amount of each drug present in the mixture was calculated. The percentage of RSD was calculated. The intermediate precision of the method was confirmed by intra-day and inter-day analysis, i.e. the analysis of mixture was repeated three times in the same day and on three successive days, respectively. The amount of drugs was determined, and % RSD was calculated. Accuracy of the method was studied by recovery experiments. The recovery experiments were performed by adding known amounts of the standard drug to mixture samples. The recovery was performed at three different concentrations levels (i.e., 80%, 100%, and 120%). This procedure was repeated for three times for each concentration. The results of recovery studies were calculated for %RSD.
Specificity is the ability of the method to measure the analyte in the presence of other relevant components. The evaluation of the specificity of the method was determined against placebo. The limit of detection (LOD) and the limit of quantitation (LOQ) of all selected combination of drugs were derived by calculating the signal-to-noise ratio using the following equations as per the ICH guidelines.
LOD = SD/Slope × 3.3 and LOD = SD/Slop × 10
Where, S.D - standard deviation of the response.
The solution stability study of standard and sample solution is determined by taking the absorbance for the 0 h, 12 h, and 24 h. The % RSD is calculated. Estimation of carisoprodol and ibuprofen were carried out in synthetic mixture (35 µgml-1 Carisoprodol and 80 µg ml-1 Ibuprofen).
RESULTS AND DISCUSSION: As carisoprodol does not contain any chromophoric group, NQS reagent was used to develop chromophore. All optimized parameters and conditions are mentioned in Table 2.
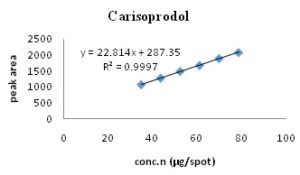
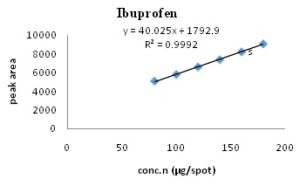
The linearity range for carisoprodol and ibuprofen were found to be 35-78.75 μg/spot and 60-180 μg/spot, respectively. Calibration spectra and curves are shown in Fig. 2- 5 and Table 3.
TABLE 2: OPTIMIZATION OF REACTION PARAMETER
| Parameter of reaction | Optimized values |
| Concentration of NQS reagent | 50 µg/ml |
| The volume of NQS reagent | 2.5 ml |
| Temperature | 70º C |
| Time | 20 min |
| pH | 9 |
TABLE 3: DETAILS OF CALIBRATION CURVE
| Std.
ID |
The volume used for spotting (µL) | The conc. of CAR µg/spot | Conc. of IBU µg/spot |
| S1 | 2 | 35 | 80 |
| S2 | 2.5 | 43.7 | 100 |
| S3 | 3 | 52.5 | 120 |
| S4 | 3.5 | 61.2 | 140 |
| S5 | 4 | 70 | 160 |
| S6 | 4.5 | 78.7 | 180 |
FIG. 2: CHROMATOGRAM OF STANDARD AND SAMPLE
FIG. 3: CHROMATOGRAM OF CARISOPRODOL (35 µg/spot TO 78.75 µg/spot) AND IBUPROFEN (80 µg/spot TO 180 µg/spot)
FIG. 4: CALIBRATION CURVE OF CARISOPRODOL (35-78.75 µg/spot) AT 254 nm
FIG. 5: CALIBRATION CURVE OF IBUPROFEN (60-180 µg/ spot) AT 254 nm
The recovery experiments were performed by the standard addition method. The mean recoveries were found 101.92 and 100.46 for ibuprofen and carisoprodol respectively. The low value of the standard deviation indicates that the proposed method is accurate. Results of recovery studies are shown in Table 4.
The stability of the solution was performed and was found to be stable up to 24 h Table 5. The robustness was studied by evaluating the effect of small changes in the mobile phase composition; the effect on the results was examined. The method was found to be robust Table 6.
Repeatability of CAR (35 µg/ spot) and IBU (80 µg/ spot) was carried out by taking absorbance five times and then find their % R.S.D. RSD was less than 2 %, which indicates that the proposed method is repeatable. LOD values for carisoprodol and ibuprofen were found to be 0.04505 and 0.0500, LOQ value for carisoprodol and ibuprofen were found to be 0.1365 and 0.15150 Table 7. These data show that the method is sensitive for the determination of ibuprofen and carisoprodol.
TABLE 4: ACCURACY OF CARISOPRODOL AND IBUPROFEN (n=3)
| Drug | % Amount added | Amount taken (sample) | Amount added
(standard) |
Total
amount |
Amount
found |
%
Recovery |
%
R.S.D |
| Carisoprodol | 80% | 35 | 28 | 63 | 62.37 | 99.00 | 0.09652 |
| 100% | 35 | 40 | 70 | 69.44 | 99.20 | 0.024086 | |
| 120% | 35 | 42 | 77 | 76.66 | 99.55 | 0.1202 | |
| Ibuprofen | 80% | 80 | 64 | 144 | 142.84 | 99.19 | 0.007951 |
| 100% | 80 | 80 | 160 | 160.07 | 100.04 | 0.009809 | |
| 120% | 80 | 96 | 176 | 178.71 | 178.71 | 0.01602 |
TABLE 5: STABILITY DETERMINATION OF SOLUTION
| Time for stability | Std. the solution of CAR (35µg/spot)measured peak area | %
R.S.D |
Std. the solution of IBU (80µg/spot) measured peak area | %
R.S.D |
| 0 h | 1082.96 | 0.1626 | 5056.83 | 0.1539 |
| 12 h | 1082 | 0.1848 | 5052.9 | 0.03433 |
| 24 h | 1081.13 | 0.02973 | 5050.63 | 0.01016 |
TABLE 6: ROBUSTNESS DATA FOR CAR AND IBU
| Name of
drug |
Condition 1- n-butanol: Glacial acetic acid: Acetone (5:2:3v/v) | Condition 2- n-butanol: Glacial acetic acid: Acetone (5:2.5:2.5 v/v) | ||
| Mean of peak Area | % R.S.D | Mean Area of peak | % R.S.D | |
| CAR(35µg/spot) | 1082.96 | 0.1626 | 1082 | 0.1848 |
| IBU(80µg/spot) | 5056.83 | 0.1539 | 5052.9 | 0.03433 |
TABLE 7: SUMMARY OF RESULT
| Parameters | Carisoprodol | Ibuprofen | |
| Linearity Range(µg/ml) | 35-78.75 | 60-180 | |
| Regression equation | y = 22.814x + 287.35 | y = 40.025x + 1792.9 | |
| Correlation co-efficient (r2) | 0.9997 | 0.9992 | |
| Accuracy
(%) |
80% | 99.00 | 99.19 |
| 100% | 99.20 | 100.04 | |
| 120% | 99.55 | 178.71 | |
| Precision
(% RSD) |
Intra day | 0.1626 | 0.1539 |
| Inter day | 0.1848 | 0.03433 | |
| Repeatability | 0.02879 | 0.01201 | |
| LOD(µg/ml) | 0.045050 | 0.0500 | |
| LOQ(µg/ml) | 0.1365 | 0.1515 | |
TABLE 8: ASSAY OF CARISOPRODOL AND IBUPROFEN IN SYNTHETIC MIXTURE
| Mixture | Amount added (mg) | Amount found (mg) | % Recovery | |||
| CAR | IBU | CAR | IBU | CAR | IBU | |
| IBU +CAR | 175 | 400 | 174.05 | 407.15 | 99.45 | 101.78 |
Summary of method parameters is shown in Table 7. Satisfactory results were obtained for both drugs in the synthetic mixture, which are in good Table 8.
CONCLUSION: This is a novel method and can be employed for routine analysis in quality control. The described method is giving accurate sensitive and precise results for the determination of carisoprodol and ibuprofen in the mixture.
ACKNOWLEDGEMENT: The authors are thankful to Centurian Laboratory, Vadodara (India) for supplying gift samples of drugs.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Tripathi KD: Essentials of Medical Pharmacology. Jaypee Brother’s Medical Publishers (P) Ltd., Edition 6th, 2003.
- Rang HP, Dale MM, Ritter JN and Moore P: Pharmacology. Churchill Livingstone, Edition 5th, 2005.
- Indian Pharmacopoeia: Government of Indian Ministry of Health and Family Welfare: The Indian Pharmacopoeial Commission, Ghaziabad, Vol. I, 1996.
- Indian Pharmacopoeia: Government of Indian Ministry of Health and Family Welfare: The Indian Pharmacopoeial Commission, Ghaziabad, Vol. I, 2010.
- United States Pharmacopoeia 31/NF27: The Official Compendia of Standards. Electronic edition. United States Pharmacopoeial Convention Inc. Rockville, MD, 2008.
- British Pharmacopoeia: The Department of Health, London, Vol. I,
- British Pharmacopoeia: The Department of Health, London, Vol. I, 2005.
- Pattanic S, Mukhi S, Pattanic G and Panda J: Assay method development and validation of ibuprofen tablets by HPLC. Der Pharmacia Sinica 2013; 4(4): 91-96.
- Hapse SA, Kadaskr PT and Shirsath AS: Difference spectrophotometric estimation and validation of ibuprofen from bulk and tablet dosage form. Der Pharmacia Lettre 2011; 3(6): 18-23.
- Wani Y and Patil D: Development and validation of spectrophotometric methods for the estimation of ibuprofen and famotidine. International Journal of Pharmacy and Pharmaceutical Science 2013; 5(3): 358-363.
- Khalid M and Younis MH: Development of UV spectrophotometric method for the simultaneous estimation of ibuprofen and famotidine in tablet dosage form by absorbance correction method and absorbance ratio method. International Journal of Research in Pharmacy and Science 2012; 2(3): 69-80.
- Gracy S, Kumar A, Debnath M and Rao S: RP HPLC analytical method development and validation for simultaneous estimation of ibuprofen and famitodine in bulk as well in pharmaceutical dosage form by using PDA detector. International Research Journal of Pharmacy 2014; 5(4): 300-305.
- Patel R, Tanna R and Thumar K: HPTLC Method for simultaneous estimation of ibuprofen and famotidine from the tablet dosage form. Inventi Rapid: Pharm Ana & Qual Assur 2012; 2012(3): 1-6.
- Solaman WD and Kumar RA: Derivatized HPTLC method for simultaneous estimation of glucosamine and ibuprofen in tablets. Journal of Pharmaceutical Research and Health Care 2010; 2(2): 156-162.
- Patwekar M and Salunkhe VR: Diazotization of carisoprodol as a skeletal muscle relaxant by derivatization spectroscopy. International Journal of Universal Pharmacy and BioScience 2013: 2(4): 474-486.
- Rohith T, Ananda S, Netkal M and Gowda M: Method development and validation of carisoprodol and its impurities by Ultra Violet-High Performance Liquid Chromatography. Advances in Analytical Chemistry 2013; 3(2): 15-19.
- Skinner W, Mackmie D and Stanley S: Quantitative determination of carisoprodol and its metabolites in equine urine and serum by Liquid Chromatography-Tandem Mass Spectrometry. Chromatographia 2004; 59(1): S61-S67.
- Mohrhaus S and Amuel R: Identification and determination of carisoprodol in tablets by Liquid Chromatography/Mass Spectrometry. Microgram Journal 2004: 2(1-4): 36-41.
- Hadad GM, Abdel-Salam RA and Emara S: Determination of glucosamine and carisoprodol in pharmaceutical formulations by LC with pre-column derivatization and UV detection. Journal of Chromatographic Science 2012: 50(4): 307-315.
- Patel H, Patel V and Patel M: Development and validation of analytical methods for simultaneous estimation of carisoprodol and aspirin in bulk and synthetic mixture by absorption ratio method using 1, 2 Naphthoquinone 4 Sulphonic acid sodium salt. International Journal of Universal Pharmacy and Bio Sciences 2013: 2(3): 1-10.
- Sreenivasulu V, Babu R and Rao N: Simultaneous determination of carisoprodol and aspirin in human plasma using liquid chromatography-tandem mass spectrometry in polarity switch mode: application to a human pharmacokinetic study: Biomedical Chromatography 2013; 27(2): 179-185.
- Patel V and Patel H: Development and validation of hptlc method for the simultaneous estimation of aspirin and carisoprodol in combined dosage form. International Journal of Universal Pharmacy and Bio Sciences 2013; 2(2): 218-227.
- Patel C, Prajapati L, Joshi A and Kharodiya A: simultaneous estimation of ibuprofen and carisoprodol in synthetic mixture. Inventi Rapid: Pharm Ana & Qual Assur 2015; 2015(2): 1-5.
- Patel C, Prajapati L, Joshi A and Kharodiya A: Simultaneous estimation of ibuprofen and carisoprodol in synthetic mixture by absorption ratio method. Inventi Rapid: Pharm Ana & Qual Assur 2015; 2015(3): 1-5.
- ICH validation of analytical procedures: text and, methodology Q2(R1). 2005.
How to cite this article:
Patel C, Prajapati L, Joshi A and Kharodiya M: Simultaneous estimation of ibuprofen and carisoprodol in synthetic mixture by HPTLC method. Int J Life Sci & Rev 2015; 1(11): 316-21. doi: 10.13040/IJPSR.0975-8232.IJLSR.1(11).316-21.
All © 2015 are reserved by International Journal of Life Sciences and Review. This Journal licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.
Article Information
2
316-321
648
1950
English
IJLSR
C. Patel, L. Prajapati*, A. Joshi and M. Kharodiya
Shri B. M. Shah College of Pharmaceutical Education and Research, Modasa, Gujarat, India.
laxchem@rediffmail.com
09 October 2015
20 November 2015
28 November 2015
10.13040/IJPSR.0975-8232.IJLSR.1(11).316-21
30 November 2015