STABILITY INDICATING SIMULTANEOUS ESTIMATION OF METFORMIN AND EMPAGLIFLOZIN IN PHARMACEUTICAL TABLET DOSAGE FORM BY RP-HPLC
HTML Full TextSTABILITY INDICATING SIMULTANEOUS ESTIMATION OF METFORMIN AND EMPAGLIFLOZIN IN PHARMACEUTICAL TABLET DOSAGE FORM BY RP-HPLC
Syed Irfan Ali and P. Bharath Rathna Kumar *
Department of Pharmaceutical Analysis, Anwarul Uloom College of Pharmacy, Hyderabad - 500001, Telangana, India.
ABSTRACT: A simple, rapid, accurate, precise, specific, sensitive and stability indicating RP-HPLC method has been developed and validated for the simultaneous estimation of Metformin and Empagliflozin in bulk drug and pharmaceutical dosage form. The chromatographic separation was performed on the Kromasil C18 column (250mm×4.6mm, 5μm particle size), using a mobile phase of buffer: Acetonitrile taken in the ratio 45:55 v/v, at a flow rate of 1.1 ml/min at an ambient temperature of 30˚C with the detection wavelength at 226nm. The retention times of Metformin and Empagliflozin were 2.182 min and 2.908 min, respectively. The linearity was performed in the concentration range of 125-750 ppm, 3.12-18.75 ppm each of Metformin and Empagliflozin with a correlation coefficient of 0.999 and 0.999 for Metformin and Empagliflozin respectively. The percentage purity of Metformin and Empagliflozin was found to be 100.59% and 100.07% respectively. The proposed method has been validated for specificity, linearity, range, accuracy, precision, and robustness were within the acceptance limit according to ICH Q2 (R1) guidelines, and the developed method can be employed for routine quality control analysis in bulk and combined pharmaceutical dosage form of Metformin and Empagliflozin.
Keywords: Metformin, Empagliflozin, RP-HPLC, Method development, Method validation
INTRODUCTION: Metformin hydrochloride (MET) Fig. 1 is an oral anti-diabetic drug, which reduces the elevated blood glucose concentration in patients with diabetes but does not increase insulin secretion. It does not lower the blood glucose in nondiabetic subjects 1. Metformin HCl decreases hyperglycemia by suppressing glucose production by the liver (hepatic gluconeogenesis).
It's Chemical Name is 1,1-Dimethyl biguanide. It is official in IP 2, USP 3 and other official books 4, 5. Literature survey revealed an estimation of metformin by HPLC 6, 7, 8.
FIG. 1: CHEMICAL STRUCTURE OF METFORMIN
Empagliflozin (EMP) Fig. 2 is an antidiabetic drug. Its chemical name is (2S, 3R, 4R, 5S, 6R)-2-[4-Chloro-3-[[4-[(3S)-oxolan-3-yl]oxyphenyl] methyl] phenyl]-6-(hydroxymethyl) oxane-3, 4, 5-triol. Empagliflozin is a drug of the gliflozin class, approved for the treatment of type 2 diabetes in adults in 2014. Empagliflozin is an inhibitor of the sodium glucoseco-transporter-2 (SGLT-2), which is found almost exclusively in the proximal tubules of nephrotic components in the kidneys 9.
Various analytical methods were reported in the literature for the determination of empagliflozin in pure drug, pharmaceutical dosage forms and biological samples using High-performance liquid chromatography 10, 11, Ultra performance liquid chromatography 12, either in single or in combined forms. Literature survey revealed estimation of Empagliflozin in combination with Metformin by HPLC was carried out by a few researchers 13, 14.
In the present work, simple, rapid, precise and sensitive stability indicating reverse phase HPLC method was developed and validated for the simultaneous estimation of Metformin and Empagliflozin in pharmaceutical tablet dosage form using Kromasil C18 column.
FIG. 2: CHEMICAL STRUCTURE OF EMPAGLIFLOZIN
MATERIALS AND METHODS:
Instrumental and Analytical Conditions:
Reagents and chemicals: The pharmaceutical drug samples Metformin and Empagliflozin were obtained from Spectrum Pharma Pvt. Ltd, Hyderabad. All the chemicals used in HPLC grade. The pharmaceutical dosage form SYNJARDY-XR was purchased from a local pharmacy. The solvents used in this work were of HPLC grade and obtained from Merck Specialties Private Limited, Mumbai. Milli Q Water was used in the buffer preparation.
Equipment: A Waters e2695 gradient system with Empower-2 software and 2996 module Photo Diode Array detector equipped with a quaternary solvent delivery pump, automatic sample injector, and column thermostat was used for the analysis.
Chromatographic Conditions: The column used was Kromasil C18 (250mm×4.6mm, 5μm particle size) for analytical separation. The mobile phase consists of an aqueous solution of 0.1% orthophosphoric acid and acetonitrile in the ratio of (45:55 v/v). The flow was adjusted to 1.1ml/min. The instrument was operated at ambient temperature. The injection volume was 10μL. The UV detection was achieved at 226 nm, which is the isosbestic point of both the drugs.
Preparation of Analytical Solutions:
Preparation of 0.1%OPA Buffer Solution: 1ml of Orthophosphoric acid was pipetted out and dissolved in a 500ml of Milli-Q water taken in a 1000ml Volumetric flask, and final volume was made up to the mark with Milli-Q water.
Preparation of Mobile Phase: Mixture of buffer and acetonitrile taken in the ratio 45:55 v/v was degassed in an ultrasonic water bath for 10min and filtered through 0.45µ filtered under vacuum filtration.
Diluent Preparation: Mixture of Water and acetonitrile were taken in the ratio 50:50 v/v was used as diluent.
Preparation of the Metformin and Empagliflozin standard preparation: Accurately weighed and transferred 50mg of Metformin and 1.25mg of Empagliflozin working Standards into a 10ml clean, dry volumetric flask, added a 3/4th volume of diluent, sonicated for 5 minutes and made up to the final volume with diluent. From the above stock solutions 1 ml of Metformin and 1 ml of Empagliflozin were pipetted into a 10ml volumetric flask and made up to 10 ml with diluent to get a mixed standard solution containing concentration of 500 ppm and 12.5 ppm of Metformin and Empagliflozin, so that the drugs Metformin and Empagliflozin were in the ratio equal to that of the marketed formulation.
Preparation of Sample Solution: Twenty tablets were weighed accurately and finely powdered. A quantity of tablet powder equivalent to 500 mg of Metformin and 12.5 mg of Empagliflozin was accurately weighed and transferred into a 100 ml volumetric flask. 50 ml of the mobile phase was added to the volumetric flask and ultrasonicated for 25 min; the volume was made up to the mark and mixed well. The solution was filtered through a 0.2 μm filter to ensure the absence of particulate matter (sample stock solution). From the filtered solution, 1ml was pipetted out into a 10 ml volumetric flask and made up to 10 ml with diluent get the concentration of 500 ppm and 12.5 ppm of Metformin and Empagliflozin.
Method Development and Validation of HPLC: The suggested analytical method was validated according to ICH guidelines (Q2 R1) 15 concerning certain parameters such as system suitability, specificity, linearity, accuracy, precision, robustness, and ruggedness.
Specificity: The specificity was carried out to determine whether there is any interference of any impurities (presence of components may be unexpected to present) in the retention time of analytical peak 16. The specificity of the method was determined by checking the interference of any of the possible degradation products generated during the forced degradation study of the drugs. The forced degradation of the drug was carried out with 2 N HCl, 2 N NaOH, 20% v/v hydrogen peroxide, heat (80°C) and photolysis (365 nm) for determining the stability nature of the drugs. The degradation samples were prepared by taking suitable aliquots of the drug and drug product solutions and then undertaking the respective stress testing procedures for each solution. After the fixed period the treated test solutions were diluted up to the mark with diluent.
Forced Degradation Studies: From the filtered sample stock solution 1 ml was pipetted out into a 10 ml volumetric flask and made up to 10 ml with diluent. For every stress condition, three sample solutions were prepared at a 500 ppm concentration of MET and 12.5 ppm of EMP. This sample solution is subjected to the following stress conditions.
Acidic Degradation Condition: Acidic degradation was carried out by adding 5 ml of 2N HCl, and after 45 min neutralizing the mixture by adding 5 ml of 2N NaOH.
Alkali Degradation Condition: Alkali degradation was carried out by adding 5 ml of 2N NaOH, and after 45 min neutralizing the mixture by adding5ml of 2N HCl.
Oxidative degradation condition: Oxidative degradation was performed by exposing the drug to 5 ml of 20% (v/v) H2O2 for 45 min.
Thermal degradation condition: Thermal degradation was performed by heating the drug content at 80 ºC on a thermostatically controlled water bath for 45 min.
Photolytic degradation condition: Photolytic degradation was carried out by exposing the drug content to UV light (365 nm) inside a UV chamber for 180 min.
Peak purity of the forced degraded samples was studied by the PDA detector. The peak purity of the main analytes was found to be within limits. The purity angles of main analytes were less than the purity threshold. The results are shown in Table 5.
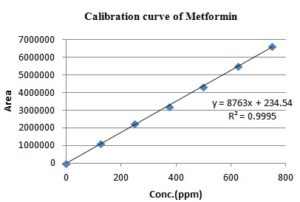
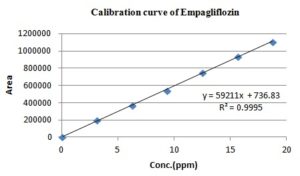
Linearity: Express ability to obtain test results where directly proportional to the concentration of an analyte in sample 17. The linearity of the proposed HPLC procedure was evaluated by analyzing a series of different concentrations for each of the two analytes. The linear regression equations were generated by least squares treatment of the calibration data. Under the optimized conditions described above, the measured peak areas were found to be proportional to concentrations of the analytes. Table 1 presents the performance data and statistical parameters, including linear regression equations, concentration ranges, correlation coefficients.
TABLE 1: LINEARITY RESULTS FOR MET AND EMP
| Parameter | Regression Equation Parameters | |
| Metformin | Empagliflozin | |
| Linearity range (ppm) | 125-750 | 3.12-18.75 |
| Correlation co-efficient | 0.999 | 0.999 |
| Slope | 8763 | 59211 |
| Y-intercept | 234.5 | 736.8 |
A stock solution of 1000 ppm of two analytes was prepared with diluent. From it, various working standard solutions were prepared in the range of 125 to 750 ppm and 3.12 to 18.75 ppm for MET and EMP respectively and injected into the HPLC system. It was shown that the selected drugs had linearity in the stated range. The calibration plot (peak area versus concentration) was generated by replicate analysis (n=3) at all concentration levels, and the linear relationship was evaluated using the least square method within the Microsoft Excel program. The retention time of standards was 2.182 min for MET and 2.908 min for EMP. A typical HPLC chromatogram of the standard mixtures is shown in Fig. 3.
FIG. 3: STANDARD CHROMATOGRAM OF METFORMIN AND EMPAGLIFLOZIN
Accuracy: Accuracy was determined by the standard addition method in terms of percentage recovery. The accuracy study was performed for 50%, 100% and 150 % of MET and EMP. Standard and sample solutions were injected into a HPLC system in triplicate, and percentage recoveries of MET and EMP were calculated. The area of each level was used for calculation of % recovery Table 2. In standard addition method 50%, 100% and 150 % of standard solutions were spiked with a known amount of optimized concentration of the drug. The amount recovered is calculated by subtracting the known optimized concentration of the drug.
Precision: Express the closeness of agreement between the series of measurement obtained from multiple sampling of the same homogeneous sample under the prescribed conditions. The precision of the method was ascertained from the peak area obtained by actual determination of six replicates of 500 ppm and 12.5 ppm of Metformin and Empagliflozin respectively.
The precision of the assay was also determined in terms of intra- and inter-day variation in the peak areas of a set of drug solutions on three different days. The intra and inter-day variation in the peak area of the drug solution was calculated in terms of relative standard deviation (RSD). The system precision values and method precision values are shown in Table 3 and Table 4.
Robustness: Robustness of the method was demonstrated by deliberately changing the chromatographic conditions 18. Robustness is defined as the influence of small, deliberate variations in procedure variables like flow rate (±5%), temperature (±5˚C), and change in wavelength (±5nm). The robustness was performed for the flow rate variations from 0.8ml/min to 1.2ml/min, and the method is robust only in less flow condition and even by a change in the mobile phase ±5%. The %RSD values for robustness study was found to be less than 2%, which indicates the method is robust.
System Suitability: System suitability tests are an integral part of the chromatographic method, which is used to verify the reproducibility of the chromatographic system. To ascertain its effectiveness, certain system suitability test parameters were checked by repetitively injecting the freshly prepared standard stock solutions at the concentration level 500 ppm and 12.5 ppm of Metformin, Empagliflozin respectively to check the reproducibility of the system and the results are shown in Table 6.
Stability of Solutions: The stability of standard working solutions, as well as sample solutions in distilled water, was examined, and no chromato-graphic changes were observed within 24 h at room temperature. Retention times and peak areas of the drugs remained unchanged, and no significant degradation was observed during these periods.
Detection and Quantification Limits: According to the pharmacopoeial recommendations (USP, 2011), the limit of detection is defined as the concentration that has a signal-to-noise ratio of 3:1, while for a limit of quantification, the ratio considered is 10:1.
The limit of detection values for MET and EMP were 0.01 and 0.01 ppm, respectively. The limit of quantification values for MET and EMP were 0.04 and 0.02 ppm, respectively.
FIG. 4: SHOWING LINEARITY FOR METFORMIN
FIG. 5: SHOWING LINEARITY FOR EMPAGLIFLOZIN
RESULTS AND DISCUSSION: The present investigation reported is a new RP-HPLC method development and validation of simultaneous estimation of MET and EMP. The method developed was proceeded with wavelength selection. The optimized wavelength was 226 nm. To get the optimized RP-HPLC method, various mobile phases and columns were used. From several trials final method is optimized with the following conditions: The mobile phase consists of 0.1% orthophosphoric acid buffer and acetonitrile in the ratio of 45:55%v/v and the column used was Kromacil C18 column (250mm×4.6mm, 5μm particle size). The flow rate was adjusted to 1.1 ml/min. The instrument was operated at ambient temperature. The UV detection was achieved at 226nm. The injection volume was 10 μL. The linearity was determined as linearity regression of the claimed analyte concentration of the range 125 to 750 ppm and 3.12 to 18.75ppm for MET and EMP respectively. The correlation coefficient was found to be 0.999, and 0.999 for MET and EMP, respectively.
TABLE 2: ACCURACY RESULTS OF MET AND EMP IN COMBINED TABLET FORM
| Drug | Standard Amount
Taken (ppm) |
Amount added* | Amount
recovered* |
%Amount
found |
%
Mean |
|
| % | (ppm) | |||||
| MET | 500 | 50 | 250 | 252.09 | 100.98 | 100.59 |
| 500 | 100 | 500 | 502.22 | 100.32 | ||
| 500 | 150 | 750 | 751.50 | 100.47 | ||
| EMP | 12.5 | 50 | 6.25 | 6.21 | 99.41 | 100.07 |
| 12.5 | 100 | 12.5 | 12.62 | 100.95 | ||
| 12.5 | 150 | 18.75 | 18.72 | 99.86 | ||
(*n=3)
TABLE 3: SYSTEM PRECISION VALUES FOR MET AND EMP STANDARD SOLUTIONS
| S.
no. |
Average area* | Rt(min)* | ||
| MET | EMP | MET | EMP | |
| 1 | 4295857 | 748275 | 2.185 | 2.913 |
| 2 | 4297872 | 749910 | 2.181 | 2.905 |
| 3 | 4333744 | 748261 | 2.185 | 2.905 |
| 4 | 4301964 | 749291 | 2.182 | 2.913 |
| 5 | 4321276 | 749145 | 2.185 | 2.903 |
| 6 | 4286875 | 743909 | 2.181 | 2.905 |
| Mean | 4306265 | 748132 | 2 | 3 |
| SD | 16092 | 1975 | 0 | 0 |
| % RSD | 0.4 | 0.3 | 0.1 | 0.1 |
(*n=6)
TABLE 4: METHOD PRECISION VALUES FOR MET AND EMP TABLET SAMPLE SOLUTIONS
| S.
no. |
Average area* | Rt(min)* | ||
| MET | EMP | MET | EMP | |
| 1 | 4289805 | 746601 | 2.182 | 2.906 |
| 2 | 4298769 | 748608 | 2.181 | 2.905 |
| 3 | 4318229 | 749676 | 2.18 | 2.901 |
| 4 | 4287690 | 758125 | 2.179 | 2.899 |
| 5 | 4299072 | 745505 | 2.181 | 2.908 |
| 6 | 4349813 | 745136 | 2.181 | 2.907 |
| Mean | 4307230 | 748942 | 2 | 3 |
| SD | 21439 | 4411 | 0 | 0 |
| % RSD | 0.5 | 0.6 | 0.0 | 0.1 |
(*n=6)
The precision of the method was ascertained from determinations of peak areas of six replicates of the sample solution. The %Relative Standard Deviation for method precision was found to be 0.5 and 0.6 for MET and EMP, respectively.
The accuracy study was performed at 50%, 100%, and 150%. The %RSD values of MET and EMP was 100.59 and 100.07% respectively. The system suitability parameters like theoretical plates (N), tailing factor (T) were calculated and were found to be more than 2000 and not more than 2 and ascertained that proposed RP-HPLC method was accurate and precise.
Forced degradation studies were performed to establish the stability indicating property and specificity of the proposed method. Degradation studies were carried out under conditions of acid hydrolysis, base hydrolysis, thermal, oxidation and photolysis, and the drug substances showed high degradation in acid and base comparative to remaining stress conditions. Thermal degradation conditions were performed by heating the drug sample at 80˚C. Acid hydrolysis showed 4 - 5.6% degradation. 4.3 - 4.9% of degradation observed in oxidation. 1-3% degradation observed in the thermal, base, and photolytic hydrolysis. There was no interference of degraded products in the analyte peaks; hence the method was proved to be stability indicating method. The results of forced degradation studies were given in Table 5.
TABLE 5: FORCED DEGRADATION STUDY OF MET AND EMP
| Stress
conditions |
% Assay of active ingredients | |||||||
| Metformin | Empagliflozin | |||||||
| %
Recovered |
% Degraded | Purity angle | Purity threshold | %
Recovered |
% Degraded | Purity
angle |
Purity threshold | |
| Acid, 2N HCl | 95.42 | 4.58 | 0.293 | 0.302 | 94.33 | 5.67 | 0.230 | 0.437 |
| Base, 2N NaOH | 97.66 | 2.34 | 0.142 | 0.296 | 97.89 | 2.11 | 0.167 | 0.371 |
| H2O2(20%,v/v) | 95.65 | 4.35 | 0.279 | 0.287 | 95.03 | 4.97 | 0.201 | 0.403 |
| Dry heat(80 ºC) | 98.83 | 1.17 | 0.135 | 0.287 | 99.4 | 0.6 | 0.149 | 0.353 |
| UV(365 nm) | 97.11 | 2.89 | 0.13 | 0.285 | 96.95 | 3.05 | 0.154 | 0.355 |
| Water | 99.57 | 0.43 | 0.152 | 0.287 | 99.16 | 0.84 | 0.149 | 0.352 |
TABLE 6: SYSTEM SUITABILITY PARAMETERS FOR MET AND EMP
| Parameter | Results | Limits | |
| Metformin | Empagliflozin | ||
| RSD of peak area | 0.4 | 0.3 | <2.0 |
| RSD of retention time | 0.1 | 0.1 | <1.0 |
| USP tailing factor (T) | 1.21 | 1.16 | T < 2 |
| USP plate count (N) | 2766 | 3543 | >2000 |
| USP resolution (R) | --- | 4.0 | > 2 |
CONCLUSION: The proposed method was found to be simple, precise, accurate, and rapid for determination of Metformin and Empagliflozin from the pharmaceutical dosage form. The method was validated for parameters like specificity, linearity, accuracy, precision, robustness, and system suitability values were found to be within limits. The method has significant advantages, in terms of shorter analysis time, selectivity, and accuracy than previously reported. The validation study indicates that the method can be considered suitable for carrying out quality control and routine determination of Metformin and Empagliflozin in bulk and pharmaceutical tablet dosage form.
ACKNOWLEDGEMENT: The authors are thankful to Spectrum Pharma Research Solutions, Hyderabad providing gift samples of Metformin and Empagliflozin. We are also grateful to Prof. Dr. A. Venkateshwar Reddy, Principal, Anwarul Uloom College of Pharmacy, Hyderabad, Telangana, India, for providing necessary facilities.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Hermann LS: Clinical Pharmacology of biguanides, Hand Book of Experimental Pharmacology. Heidelberg: Springer Veelog, 1995; 374 - 407.
- Government of India Ministry of health and family welfare, Indian Pharmacopoeia. The Controller of Publication, New Delhi, Vol- I & II, 2010: 1657-60.
- The United States pharmacopoeia: USP 34 NF 29, United States Pharmacopoeia Convention, lnc, Rockville, Vol. III, 2011; 3442.
- The Merck Index: Whitehouse Station, NJ: Merck & Co. Inc., Edition 14th, 2001.
- Martindale: The complete drug reference. London. Pharmaceutical Press, Edition 33th, 2002: 313-334.
- Chhetri HP, Thapa P and Schepdael AV: Simple HPLC-UV method for the quantification of metformin in human plasma with one step protein precipitation. Saudi Pharm J 2014; 22: 483-487.
- Kar M and Choudhury PK: Indian J Pharm Sci 2009; 71(3): 418-420.
- Madhukar A, Prince A, Vijay Kumar R and Sanjeeva: Simple and sensitive analytical method development and validation of metformin hydrochloride by RP-HPLC IJPPS 2011; 3(3): 117-120.
- Grempler R, Thomas L, Eckhardt M, Himmelsbach F, Sauer A, Sharp DE, Bakker RA, Mark M, Klein T and Eickelmann P: "Empagliflozin, a novel selective sodium glucose cotransporter-2 (SGLT-2) inhibitor: characterization and comparison with other SGLT-2 inhibitors". Diabetes Obes Metab 2012; 14(1): 83-90.
- Shyamala, Nirmala K, Mounika J and Nandini B: Der Pharmacia Lettre 2016; 8(2): 457-464.
- http://www.scholarsresearchlibrary.com/abstract/validated-stabilityindicating-rphplc-method-for-determination-of-empagliflozin-6371.html
- Padmaja N and Veerabhadram G: Der Pharmacia Lettre 2015; 7(12): 306-312.
- Ayoub and Bassam M: UPLC simultaneous determination of empagliflozin, linagliptin and metformin. RSC Advances 2015; 5: 116.
- Noorulla SM and Ali S: International Journal on Engineering Technology and Sciences 2015; 2(11): 66-71.
- http://www.ijets.in/Downloads/Published/E0140211011.pdf
- Suresh BK, Geetha SP, Lakshmana RK and Prasad KRS: Asian J Pharm Clin Res 2016; 9(1): 126-135.
- https://innovareacademics.in/journals/index.php/ajpcr/issue/view/211
- International Conference on Harmonization: Technical Requirements for Registration of Pharmaceuticals for Human Use, ICH Harmonized Tripartite Guideline Validation of Analytical Procedures: Text and Methodology Q2 (R1), Geneva, Switzerland 1996.
- Haritha A, Kumar PBR and Priya RV: Journal of Global Trends in Pharmaceutical Sciences 2015; 6(2): 2600- 06.
- Kandikattu K, Kumar PBR and Devanna N: Der Pharmacia Sinica 2014; 5(5): 74-81.
- Kumar PBR, Priya RV and Reddy AV: Asian Journal of Chemistry 2017; 9(5): 1061-64.
How to cite this article:
Ali SI and Kumar PBR: Stability indicating simultaneous estimation of Metformin and Empagliflozin in pharmaceutical tablet dosage form by RP-HPLC. Int J Life Sci & Rev 2017; 3(9): 92-99. doi: 10.13040/ IJPSR.0975-8232.IJLSR.3(9).92-99.
All © 2015 are reserved by International Journal of Life Sciences and Review. This Journal licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.
Article Information
1
92-99
422
1380
English
IJLSR
S. I. Ali and P. B. R. Kumar*
Department of Pharmaceutical Analysis, Anwarul Uloom College of Pharmacy, Hyderabad, Telangana, India.
bharathpharm@gmail.com
03 September 2017
22 September 2017
24 September 2017
10.13040/IJPSR.0975-8232.IJLSR.3(9).92-99
30 September 2017