STEVENS-JOHNSON SYNDROME (SJS): A CLINICAL CASE REPORT
HTML Full TextSTEVENS-JOHNSON SYNDROME (SJS): A CLINICAL CASE REPORT
Nahida Farheen Shaik, P. Ankitha and Balaiah Sandyapakula *
Department of Pharmacy Practice, Nirmala College of Pharmacy, Mangalagiri, Guntur - 522503, Andhra Pradesh, India.
ABSTRACT: We report a case of Stevens-Johnson syndrome (SJS) associated with the use of etodolac, sulfasalazine SR, ceftrioxime. The patient medical history shows that she is a known case of rheumatoid. She has no improvement of joint pain and developed a few fluid filled lesions in the oral cavity, which ruptured spontaneously. She had a history of pain and burning sensation while swallowing and her first biopsy report showed erythema multiforme on hospital admission and was finally diagnosed as toxic epidermal necrolysis. The patient was treated with inj. Hydrocortisone 100 mg BD, inj. Tagocid 400 mg OD, T. Dolo 600 mg TID, inj. Piptaz 45 g BD. SJS and toxic epidermal necrolysis (TEN) are very rare adverse cutaneous reactions that can be associated with the use of Sulfasalazine. The most severe cases can result in death, and for the others, permanent skin, mucosal or ocular sequelae, which can impair the quality of life in patients. Clinicians and patients need to be aware of the signs and symptoms that often precede the appearance of the diffuse exfoliations lesions in an SJS or TEN. Immediate withdrawal of the offending medication should be done when blisters or erosions appear in the course of a drug eruption, as this may improve the prognosis.
Keywords: Steven-Johnson syndrome, Toxic epidermal necrolysis, Severe cutaneous adverse drug reaction, Sulfasalazine
INTRODUCTION: Stevens-Johnson syndrome (SJS) is an immune-mediated life-threatening cutaneous hypersensitivity skin disorder 1. A review confirmed four highly suspected causative drugs: sulfonamide, phenobarbital, carbamazepine, and lamotrigine 2. SJS is a threatening mucocutaneous syndrome caused by drugs like antimicrobials, antiepileptic, and analgesics they’re in the main, however not invariably, caused by medication.
Erythema (EM) was antecedently thought of to be a milder style of SJS while not tissue layer involvement; but, the clinical classification outlined by Bastuji-Garin in 1993 separates EM as a clinically and aetiologically distinct disorder and has currently been accepted. The human leukocyte antigen HLA-B one of the investigation marker for SJS 4. A classification system, based largely on the extent of epidermal detachment and morphology of the skin lesions, aids in differentiating opposite spectrums of the same disease entity 15.
This system comprises the following: TEN along with spots, TEN without spots, Overlap Stevens-Johnson syndrome and TEN (SJS-TEN) Drugs with increased risk for SJS/TEN have been classified based on data from the Regis SCAR / Euro SCAR registry exceptionally, adverse skin reactions to sulfasalazine can manifest as Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN) 14. The incidence of SJS and TEN ranges from 0.4 to 1.2, and 1.2 to 6 per million person-years, respectively. We report a case of SJS associated with the use of Etodolac, Sulfasalazine SR, Cefpodoxime 200 mg OD. The case study was approved hospital ethical committee and also taken by patient consent.
Case Report: A 65 yr old female was hospitalized on 6th Dec 2016 for having Hives all over body and peripheries after intake of oral medication for joint pain -Etodolac, Sulfasalazine SR, Monocef. The patient used other medications like Tab. Levoflox 500 mg, Tab. Doxyphyllin, Tab. Dalacin. 300 mg. She developed diffuse exfoliations and lesions all over the body since 4 days with bold tongue and developed fever, and she was dehydrated.
The patient had the history of HTN, DM since 5years treated with CCB and HAI and Rheumatoid arthritis, and the patient was presented with oral ulcers, high spikes of fever, dyspnoea, exanthematous widespread maculopapular rash involving the trunk, upper and lower limbs. The patient had developed an ulcer over right leg 20 days back. The patient’s physical findings include P.R: 88/min, R.R: 24/min, BP: 110/70 mm of Hg, SPO2 at room temperature was 98%. Lab data includes Sr. sodium: 132L mmol/L, Sr. chloride: 94L mmol/L, total calcium 6.9L mg/dl, total Protein: 4.7L g/dl, uric acid: 7.1H mg/dl.
Patient admitted with above complaints and consulted Physician and was treated conservatively with inj. Hydrocortisone 100 mg BD, inj. Tagocid 400 mg OD, T. Dolo 600 mg TID, inj. Piptaz 45 g BD, inj. Calcium gluconate 10 cc TID slow over 10 min and other supportive measures like nasal inhalations, which include duolin and budecort TID. Dermatologist opinion took given SJS and treated by derma dew ointment BD, close ointment H/S, Tab. Levocetirizine OD, Tab. Ultracet SOS. Dietician adviced to take the liquid oral diabetic diet of 1000 k: calories and high protein diet of 50 gm in frequent intervals and to add extra salt. Patient is discharged on stable condition with prescription of Tab. Nofloxacin 400 mg OD, Tab. Lasilactone 2 tabs OD, Tab. Omnicortil 40 mg OD followed by 20 mg once a week, Tab. Montair FX OD, Tab. Dafron 100 mg BD, Tab. Glycomet 500 mg BD, Tab. Repace OD, syp. sucral 15 ml TID, syp. lacsihep 30 ml OD, syp. salvit-Z BD.
DISCUSSION: SJS is an acute, life-threatening skin inflammatory conditions 1, The clinical presentations of SJS and TEN are characterized by fever and influenza-like symptoms unexplained by infectious illness that often precedes the cutaneous and mucosal lesions (present in about 90% of patients) 3, caused by drugs commonly used in general practice for chronic diseases, it includes sulfonamides, antiepileptics, NSAIDs, etc. some of the studies state that the diffuse exfoliation lesions are produced majorly by NSAIDs and sulfonamides 2, 3, 4. The primary interventions when SJS or TEN is suspected are early recognition and immediate cessation of any potential causative agents and to provide supportive treatment in an appropriate clinical setting. Prompt withdrawal of the drug suspected to have caused the SJS or TEN may reduce mortality and should be done at the first sign of mucosal involvement 11.
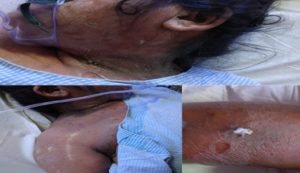
FIG. 1: SHOWS SKIN TEN REACTIONS
In this case, the patient was treated by antibiotics, corticosteroids for symptoms of SJS, and prevention of disease progression, antihistamines, and analgesics are used for symptomatic relief. Additionally, the patient received some skin ointments like derma dew ointment and close ointment 5, 6. Steroids have been accepted as a treatment option as they suppress the necrolytic process in the skin and internal organs. A study shows that early treatment with corticosteroids reduced morbidity and improved survival in patients with SJS 10. The patient was presented with oral ulcers, high spikes of fever, dyspnoea, xanthomatous widespread maculopapular rash involving the trunk, upper and lower limbs 12, 13.
In this case of sulfasalazine-induced SJS, despite the presence of a macular-papular rash and vulvar involvement, resulting in a more severe reaction. The withdrawal of sulfasalazine was followed by the gradual disappearance of lesions and complete recovery without sequelae in the patient. Clinicians and patients need to be aware of the signs and symptoms that often precede the appearance of the mucocutaneous lesions in an SJS or TEN. These include fever, influenza-like symptoms, sore throat, or burning eyes. Prompt intervention in the early course of the disease may improve the prognosis. Patient education is of the utmost importance after an episode of SJS and TEN. It is imperative that the patient be aware of the causative drug, and of other chemically related drugs that have the potential to cross-react with sulfonamide and potentially with sulfasalazine 9, 10.
Identification of predisposing HLA subtypes could help to predict rare allergic severe adverse cutaneous reactions, such as SJS or TEN, to sulfonamides or other drugs. Studies done in Asian patients have demonstrated an association between specific HLA subtypes, and SJS/TEN induced by allopurinol and carbamazepine 16. Thus, it is uncertain if pharmacogenomics will help to predict those rare hypersensitivity reactions 17. For now, immediate cessation of drugs suspected to cause SJS or TEN remains the most important measure in clinical practice.
Treatment of SJS and TEN: SJS treatment was supportive; there are no established treatments for SJS. Discontinue causative medications. Treat like a burns patient with aggressive fluids, pain control, aseptic handling, nutritional support, and antibacterial treatment. Standardized treatment protocols were associated with lower mortality rate and children were thought to have a better prognosis and faster re-epithelization 7, 16.
CONCLUSION: Steven-Johnson syndrome and toxic epidermal necrolysis (diffuse exfoliations lesions) is severe cutaneous adverse drug reactions (SCAR) associated with high mortality in patients prescribed with antibiotics and sulfonamides like sulfasalazine, educating them regarding the appropriate use of medications are of utmost importance. It is also advisable to provide personalized “alert card”, with the description of adverse drug reaction, to the patient who suffered from such serious reactions. The patient we reported recovered from the reaction after the specific and symptomatic treatment of the presenting illness
ACKNOWLEDGEMENT: We are sincerely acknowledged to Dr. Srinivasa Rao T., HOD of MICU of Manipal Hospital, Vijayawada, to conduct this research and there support in every potential manner.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Stevens-Johnson Syndrome; Derm IS (Dermatology Information System)
- Mockenhaupt M; The current understanding of Stevens-Johnson syndrome and toxic epidermal necrolysis. Expert Rev Clin Immunol 2011; 7(6): 803-13; 814-5.
- Maggini V, Lombardi N, Lenti MC, Masi S and Trapani S: A case of pediatric Steven–Johnson syndrome associated with albuterol consumption. Inter Immunopharmacology 2015; 27: 154-155.
- Tremblay L: Stevens-Johnson syndrome with sulfasalazine treatment: Report of two cases. Journal of Crohn's and Colitis 2011; 5: 457-460.
- Patel: Systematic review of Stevens-Johnson syndrome and toxic epidermal necrolysis. Ind J of Dermatology, Venereology, and Leprology 2013; 79(3): 389-398.
- Mockenhaupt M, Viboud C, Dunant A, Naldi L, Halevy S, Bouwes Bavinck JN, Sidoroff A, Schneck J, Roujeau JC and Flahault A: Stevens-johnson syndrome and toxic epidermal necrolysis: assessment of medication risks with emphasis on recently marketed drugs. The Euro SCAR-study. J Invest Dermatol 2008; 128: 35-44.
- Sassolas B, Haddad C, Mockenhaupt M, Dunant A, Liss Y, Bork K, Haustein UF, Vieluf D, Roujeau JC and Le Louet H: ALDEN, an algorithm for assessment of drug causality in Stevens-johnson syndrome and toxic epidermal necrolysis: comparison with case-control analysis. Clin Pharmacol Ther 2010; 88: 60-8.
- Revuz J, Penso D, Roujeau JC, Guillaume JC and Payne CR: Toxic epidermal necrolysis. Clinical findings and prognosis factors in 87 patients. Arch Dermatol 1987; 123(9): 1160-5.
- Harr and French: Toxic epidermal necrolysis and Stevens-johnson syndrome. Orphanet J of Rare Dis 2010; 5: 39.
- Levi N, Bastuji-Garin S and Mockenhaupt M: Medications as risk factors of Stevens-johnson syndrome and toxic epidermal necrolysis in children: a pooled analysis. Pediatrics 2009; 123(2): 1.
- Halevy S, Ghislain PD and Mockenhaupt M: Allopurinol is the most common cause of Stevens-johnson syndrome and toxic epidermal necrolysis in Europe and Israel. J Am Acad Dermatol 2008; 58(1): 25-32.
- Patel PP: An analysis of drug-induced Stevens-johnson syndrome. Indian J Med Res 2012; 136(6): 1051-1053.
- https://www.drugs.com/sfx/sulfasalazine-side-effects.html
- Roujeau JC: Medication use and the risk of Stevens-Johnson syndrome or toxic epidermal necrolysis. N Engl J Med 1995; 333(24): 1600-7.
- Bastuji-Garin S, Rzany B, Stern RS, Shear NH, Naldi L and Roujeau JC: Clinical classification of cases of toxic epidermal necrolysis, Stevens-johnson syndrome, and erythema multiforme. Arch Dermatol 1993; 129(1):92-6.
- Das S, Roy AK and Biswas I: A six-month prospective study to find out the treatment outcome, prognosis and offending drugs in toxic epidermal necrolysis from an urban institution in Kolkata. Indian J Dermatol 2013; 58(3): 191-3.
- Garcia-Doval I, LeCleach L, Bocquet H, Otero XL and Roujeau JC: Toxic epidermal necrolysis and Stevens-Johnson syndrome. Does early withdrawal of causative drugs decrease the risk of death? Arch Dermatol 2000; 136: 323-7.
How to cite this article:
Shaik NF, Ankitha P and Sandyapakula B: Stevens-Johnson syndrome (SJS): A clinical case report. Int J Life Sci & Rev 2018; 4(6): 94-97. doi: 10.13040/IJPSR.0975-8232.IJLSR.4(6).94-97.
All © 2015 are reserved by International Journal of Life Sciences and Review. This Journal licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.
Article Information
1
94-97
565
1288
English
IJLSR
N. F. Shaik, P. Ankitha and B. Sandyapakula *
Department of Pharmacy Practice, Nirmala College of Pharmacy, Mangalagiri, Guntur, Andhra Pradesh, India.
balu.slns@gmail.com
08 December 2017
24 March 2018
21 June 2018
10.13040/IJPSR.0975-8232.IJLSR.4(6).94-97
30 June 2018