STUDY OF RESPIRATORY CHAIN DYSFUNCTION IN HEART DISEASE
HTML Full TextSTUDY OF RESPIRATORY CHAIN DYSFUNCTION IN HEART DISEASE
Seyyed Hossein Hassanpour * 1 and Seyyedeh Zeinab Karami 2
Young Researchers and Elite Club 1, Yasooj Branch, Islamic Azad University, Yasooj, Iran.
Department of Biology 2, Faculty of Basic Sciences, Yasouj University, Yasouj, Iran.
ABSTRACT: The relentlessly beating heart has the greatest oxygen consumption of any organ in the body at rest, reflecting its huge metabolic turnover and energetic demands. The vast majority of its energy is produced and cycled in the form of ATP which stems mainly from oxidative phosphorylation occurring at the respiratory chain in the mitochondria. A part of energy production, the respiratory chain is also the main source of reactive oxygen species and plays a pivotal role in the regulation of oxidative stress. Dysfunction of the respiratory chain is therefore found in most common heart conditions. The pathophysiology of mitochondrial respiratory chain dysfunction in hereditary cardiac mitochondrial disease, the aging heart, in LV hypertrophy and heart failure, and ischemia-reperfusion injury is reviewed. We introduce the practicing clinician to the complex physiology of the respiratory chain, highlight its impact on common cardiac disorders, and review translational pharmacological and non-pharmacological treatment strategies.
Keywords: Beating heart, Metabolic, Respiratory chain, Mitochondrial, Cardiac disorders
INTRODUCTION: Approximately 25% of a human myocardial cell is made up of mitochondria. Mitochondria are cellular factories converting substrates from the diet into usable energy for many intracellular processes, including mechanical contraction of myofilaments. The ultimate substrate used by most enzymes to convert chemically stored energy into conformational changes, and finally, mechanical motion is adenosine-triphosphate (ATP). The heart has a voracious requirement for energy-indeed the human heart cycles approximately 6kg of ATP per day 1.
The majority of this ATP is generated in mitochondria at the respiratory chain by oxidative phosphorylation, and as a byproduct, the respiratory chain generates reactive oxygen species (ROS). Under physiological conditions, ROS plays an important role in intracellular signaling, but in pathological states increased ROS production can become detrimental to the cardiomyocyte.
Associated with energy balance are other mitochondrial key roles, namely regulation of calcium homeostasis and apoptotic signaling. It is beyond the scope of this review to discuss in detail the latter two important processes. It is not surprising that mitochondrial diseases preferentially affect tissues with high energy turnover, such as the heart. Impaired oxidative phosphorylation and defective electron transport chain (ETC) function are central to most cardiac conditions associated with mitochondrial dysfunction.
Their malfunction has been implicated in hereditary mitochondrial cardiomyopathies, in the aging heart, cardiac hypertrophy, heart failure, and in ischemia-reperfusion injury.
Review Method: In this study, we reviewed papers related to respiratory chain dysfunction in cardiac disease. For this purpose, we searched keywords such as beating heart, metabolic, respiratory chain, mitochondrial, cardiac disorder in databases include a web of science, PubMed, and Scopus since from 1992 to 2017.
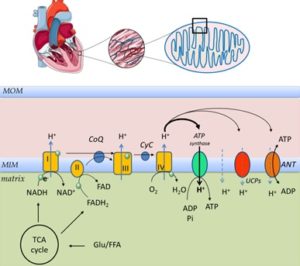
Physiology of Respiratory Chain: Mitochondria generate adenosine triphosphate (ATP), using the electron transport chain (ETC) and the oxidative phosphorylation system (OXPHOS). The proteins involved in this process are located in the mitochondrial inner membrane (MIM) and collectively referred to as the respiratory chain (RC), Fig. 1. Acetyl CoA generated from glycolysis, and fatty acid beta-oxidation (FAO) enters the Tricarboxylic acid cycle (TCA). The TCA cycle, glycolysis, and FAO all generate high energy electrons in the form of NADH (nicotinamide adenine dinucleotide).
These electrons are then passed along the ETC in a series of redox reactions. The ETC comprises 5 protein complexes and two shuttles, Fig. 1. NADH passes an electron to complex I (NADH dehydrogenase) and this, in turn, passes the electron through a shuttle Coenzyme Q (Ubiquinone) to complex III (cytochrome b-c1). Another source of high energy electrons for complex III stems from FADH2 which is generated in the TCA cycle by succinate dehydrogenase which is both a TCA cycle component and a component of the ETC complex II. The final common pathway through complex III transfers electrons to another electron carrier, cytochrome C, which in turn passes its electrons to complex IV (cytochrome oxidase).
Finally, the energy depleted electron (in the form of hydrogen) is accepted by molecular oxygen, completely reducing it to form water. This series of redox reactions release energy which drives the extrusion of protons outwards through complexes I, III, and IV to create an electrochemical gradient across the inner mitochondrial membrane. This gradient, in turn, drives the phosphorylation of ADP to ATP by ATP synthase (complex V), Fig. 1. This reaction is reversible, and during severe ischemia, large amounts of ATP may be ‘wasted’ in maintaining the electrochemical gradient via the dephosphorylation of ATP. However, OXPHOS is not fully efficient, and even under physiological conditions, some of the energy is dissipated as heat. This is due to proton leak from the inter mitochondrial membrane space into the matrix through uncoupling proteins (UCPs), adenine nucleotide translocase (ANT) or non-specific membrane proton slippage, Fig. 1.
FIG. 1: MITOCHONDRIAL RESPIRATORY CHAIN AND OXIDATIVE PHOSPHORYLATION. Under aerobic conditions, substrates from fatty acid oxidation and glycolysis are metabolized in the TCA cycle which delivers high energy loaded electrons (in the form of hydrogen via NADH and FADH2) to the electron transport chain (ETC) complexes located in the inner mitochondrial membrane. Amongst the ETC complexes electrons are passed over via shuffles coenzyme Q (ubiquinone) and cytochrome c. Electrons undergo a series of redox reactions at copper and iron centers of individual ETC complexes which releases energy. Associated conformational changes at complexes I, III and IV lead to extrusion of hydrogen ions from matrix into the inter-membranous space creating an electrochemical gradient, a driving force for phosphorylation of ADP at the ATP synthase (complex V). The electron flow is limited by the availability of oxygen, a terminal acceptor of the energy depleted electron at the complex IV (cytochrome oxidase). A part of the trans-membranous gradient is dissipated via uncoupling proteins (UCPs and ANT) resulting in a proton leak, playing an important role in the regulation of membrane potential with associated electron leak (ROS production) and oxidative phosphorylation efficiency.
Direct H+ slippage through the phospholipid bilayer is thought to play a negligible role only. MIM mitochondrial internal membrane, CoQ coenzyme Q (ubiquinone), CyC cytochrome C, I/II/III/IV electron chain transport complexes I, II, III and IV; ANT adenine nucleotide translocase, UCPs uncoupling proteins; with permission heart and tissue image provided by Servier Laboratories UK Medical Art Gallery.
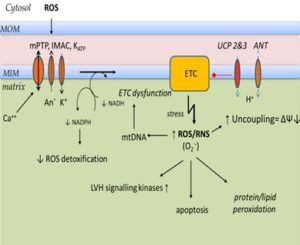
This non-ATPase related loss of trans-membrane potential makes ATP production by OXPHOS less efficient (less ATP produced per oxygen molecule consumed). ANT catalyzes the exchange of ADP and ATP between cytosol and mitochondria, but it also contributes to proton leak. UCP2 and UCP3 are found in human cardiac muscle, and their expression correlated positively with plasma free fatty acid concentrations 2. Their activities are increased by reactive oxygen species (ROS) 3, Fig. 2. Physiological uncoupling of OXPHOS may decrease excessive ROS production and reduce oxidative damage (‘Uncoupling to survive’) 4. Indeed rather than thermogenesis, this may be the major physiological role of uncoupling. Approximately 20-30% of resting cellular energy expenditure dissipates as heat due to proton leak. In a negative feedback cycle, ROS induced uncoupling leads in turn to suppression of ROS production by the ETC, Fig. 2. The decreased trans-membrane potential (ΔѰ) regulates the ETC redox state, which in turn suppresses superoxide anion production by complexes Ⅰ and III 5.
Superoxide production (O2.-) is related to electron leak, which is closely interlinked with proton leak regulation. Electron leak can occur when electrons exit the ETC early before their final reduction to water to form superoxide instead. Under experimental conditions a high ΔѰ (or ΔpH) can reverse the electron transport at the complex, reducing NAD+ to NADH and forming superoxide. An uncoupling induced decrease in the proton gradient reduces the reverse electron transport and the superoxide production at complex I 6. Mitochondria are thus both sources (complex I and III) and targets of reactive oxygen and nitrogen species (ROS and RNS). Electron slippage at complexes I and III lead to an incomplete reduction of molecular oxygen to form superoxide. At non-pathological levels, ROS play important functions in cellular signaling. However, when oxidative stress is increased, the associated mtDNA damage may further enhance ROS production, resulting in a vicious cycle 7.
Hereditary Cardiomyopathies: The RC system is made up of about 100 different proteins. Only 13 of these are encoded by mitochondrial DNA {(mtDNA) with a maternal pattern of inheritance 8, the remainder being encoded by nuclear DNA (nDNA), following a Mendelian inheritance pattern 9. All complexes of the ETC, except complex II which is encoded exclusively by mtDNA, have a double genetic origin (mtDNA and nDNA). Moreover, it is hypothesized that several hundred nuclear genes are also needed for various functions of the RC 10. Increasingly it has also been recognized that mutations of mtDNA encoding for tRNA genes can affect protein synthesis with impaired respiratory chain function and lead to cardiomyopathy 11. The great variability of clinical presentation of inherited disorders related to mutations of mitochondrial genes is largely attributed to peculiar features of mitochondrial genetics, heteroplasmy, and the threshold effect. A single mitochondrion can harbor both normal and mutant mtDNA- an effect known as heteroplasmy.
A critical amount of mutant mtDNA is necessary to cause RC dysfunction and clinical symptoms (known as a ‘threshold effect’) 12. Disease-based epidemiology studies estimate the population prevalence of mtDNA disease at ~1:5000, while heteroplasmic mtDNA mutations are found in 1:200 of newborns (Elliot 2008). Mitochondrial disease can present at any age and affect almost any organ, but most commonly it involves the heart, brain, skeletal muscle, eye, or endocrine system. Cardiologists need to consider the possibility of mtDNA disease if the cardiac disease in the form of unexplained LV non-compaction, LVH, HCM, DCM or conduction defects are associated with maternal inheritance, either in isolation (e.g., HCM) or with other clinical features suggesting mitochondrial diseases such as a combination of diabetes and deafness. Whereas syndromes like MELAS (myopathy, encephalopathy, lactic acidosis, optic atrophy, and stroke-like syndrome) are well defined, many patients do not fit these syndromic categories 13.
Barth syndrome, caused by impaired mitochondrial respiration was the first inherited disorder described as associated with left ventricular non-compaction, rare congenital cardiomyopathy characterized by extensive endomyocardial trabeculation. However, most LV non-compaction cardiomyopathies are caused by mutations of sarcomere genes overlapping with hypertrophic cardiomyopathy, rather than mitochondrial genes 14. Severe exercise limitation is typical of mitochondrial cardiomyopathies with associated skeletal myopathy, and further investigation frequently reveals premature lactate acidosis during exercise. The massive proliferation of abnormal mitochondria with ragged red fibers on skeletal muscle biopsy and positive genetic testing contribute to the diagnosis 12. Rarely in predominantly cardiac involvement may endomyocardial biopsy become necessary 13, 15.
Aging Heart: In 1956, Harman suggested mitochondria as the main source of ROS and its causative role in age-related changes 16. Short et al., have confirmed that in human’s mtDNA abundance and ATP production declines with advancing age, whereas the level of oxidative mtDNA lesions increases 17. mtDNA is not protected by histones, unlike nDNA and has less effective repair mechanisms 18. All of these factors contribute to a gradual increase in mtDNA mutation rates with age. This affects the expression and integrity of RC complexes, which can lead to further ROS production, perpetuating a vicious cycle of oxidative damage, Fig. 2.
A small age-related decline in heart mitochondria numbers has been described in rats and humans, but this occurs without the loss of volume taken by mitochondria within cardiomyocytes. Aging cardiac mitochondria loose cristae and the respiratory chain function becomes impaired with lower average trans-membrane potential (ΔѰ), decreased ATP synthesis efficiency, and augmented ROS production with sensitization to mPTP opening which promotes apoptosis 19. These processes were previously linked to age-related myocardial atrophy, stiffness, and diastolic dysfunction 19, 20. In animal models marked life span extension has been achieved by overexpression of enzymes which degrade ROS such as mitochondrial superoxide dismutases (MnSOD and Cu/ZnSOD) 21 or catalase 22. In skeletal muscle over-expression of UCP3 leads to blunting of the age-induced increase in ROS 23, and animal models have confirmed the association of increased uncoupling with increased life span 24 and improved mitochondrial biogenesis 25. However, the vicious cycle proposed by the mitochondrial theory of aging has been challenged by an experiment with mice expressing error-prone mtDNA polymerase 26. These mice accumulate substantial burdens of mtDNA mutations, associated with premature aging phenotypes and reduced life span. However, their ROS production was normal, and no increased sensitivity to oxidative stress-induced death was observed, despite severe RC dysfunction.
FIG. 2: PATHOPHYSIOLOGY OF RESPIRATORY CHAIN, REACTIVE OXYGEN SPECIES PRODUCTION/ SIGNALLING AND OXIDATIVE PHOSPHORYLATION, UNCOUPLING, ‘ROS INDUCES ROS’. Inherited or senescence acquired mtDNA abnormalities, LVH or heart failure all can lead to dysfunction of ETC with an increase of electron leak, mainly at complexes I and III and result in pathologic ROS production with detrimental consequences to the mitochondrion and the cell. In a vicious cycle, this oxidative stress can perpetuate ETC dysfunction further via damage to mtDNA and proteins involved in electron flow at the respiratory chain. To restore balance in a negative feedback matrix, ROS can induce increased proton leak across MIM via uncoupling proteins. A physiologic reduction of the trans-membrane electrochemical gradient (ΔѰ) can reduce the electron leak and hence counter fight the vicious cycle of oxidative stress. In failing hearts, cytosolic ROS is suspected of activating the mPTP, IMAC and the mito-KATP. Activated mPTP and IMAC can both release superoxide (ROS) from the negatively charged mitochondrial matrix into the cytosol (“ROS induces ROS”). H2O2 (ROS) requires for its detoxification NADPH, which stands in equilibrium with NADH, which is derived from the TCA cycle. Pathologic dissipation of ΔѰ via mPTP/IMAC/ mito-KATP leads to increased electron flow through the ETC and therefore reduction of NADH to NAD+. This results in a shortage of NADPH and therefore impaired H2O2 detoxification.
ROS/RNS reactive oxygen/nitrogen species, O2- superoxide anion, mtDNA mitochondrial DNA, ETC electron chain transport system, ΔѰ mitochondrial internal membrane potential, IMAC inner membrane anion channel, mito-KATP mitochondrial ATP-dependent K+ channel, mtPTP mitochondrial permeability transition pore , UCP2 & UCP3 uncoupling proteins. The authors concluded that the mtDNA mutation accumulation with severe RC dysfunction per se is the primary inducer of premature aging independent of elevated ROS production. The ROS production may be merely a consequence, rather than driving force of the aging process 26. Dysfunctional mitochondria can trigger the removal of damaged cells via apoptosis.
However, in non-proliferating tissues (such as the heart) apoptosis of whole cells would be detrimental, and therefore, a more efficient system of mitochondrial quality control is necessary 20. The quality control happens by the interplay of fusion, fission (splitting into two daughter mitochondria), autophagy (lysosomal break down of damaged proteins and organelles), and biogenesis of new mitochondria 20. Aging is associated with a decline in autophagy and accumulation of aberrant macromolecules in swollen giant mitochondria 27. Dysfunctional mitochondria with inhibited RC and depolarised membrane are unable to fuse with healthy mitochondria and later be targeted for removal by autophagy 28. Reduced autophagy in Atg5 (cardiac specific autophagy related 5 genes) deficient mice leads to age-related cardiomyopathy 29.
LV Hypertrophy and Heart Failure: Changes in the energetic mitochondrial profile are a hallmark of hypertrophied and failing hearts. Increased oxidative stress activates a variety of hypertrophy signaling kinases and transcription factors 30, 31. Initially, a pressure overload-induced LV hypertrophy leads to a shift of fatty acid oxidation towards more efficient glucose oxidation. However, it also leads to a reduction of maximal OXPHOS capacity with decreased activities of respiratory chain complexes and increase of electron leak 32. At the failing heart stage, the energy production decreases further and oxidative stress increases while facilitating cell dysfunction, and inducing apoptosis. ROS production also stimulates cardiac fibroblast proliferation, and expression and posttranslational activation of matrix metalloproteinases, which play a pivotal role in extracellular remodeling. Oxidative stress can additionally activate apoptosis and contribute to maladaptive myocardial remodeling 33.
The transition from compensated LV hypertrophy to failure is preceded by complex Ⅰ and II dysfunction, followed by an increase of pro-apoptotic markers (Bax/Bcl-2 ratio) 34. An increase in ROS production and other phenotypic similarities have been found in both the aging and the failing heart. Mitochondrial proteins, as well as lipids, may be targets of superoxide and its metabolites. This damage may lead to impaired mitochondrial respiration. Reduced maximal mitochondrial respiration was found in permeabilized cardiac muscle from dogs with ischemia-induced chronic heart failure 35 and in patients undergoing cardiac transplantation 36.
Some specific conditions, including cumulative iron-mediated damage to mtDNA in hemochromatosis or myocardial inflammation in Chagas cardiomyopathy, can alter structure, function, ETC activity leading to heart failure 37, 38, 39. Reduced activity of ETC subunits in patients with heart failure has been described previously, notably of complex I 40, complex III 41 and complex IV 42. These changes are found independently of the etiology of the cardiomyopathy (ischaemic or idiopathic DCM). Impaired ETC activity can lead to increased mitochondrial ROS production 43.
Additionally, the elimination of ROS may be impaired as a marked decrease of MnSOD activity has been described in human failing heart 44. Recently it was suggested that cytosolic ROS might lead to amplification of mitochondrial ROS production (“ROS induces ROS”) 45. The presence of mitochondrial nicotinamide adenine dinucleotide phosphate (NADPH) allows enzymatic detoxify-cation of H2O2 and is in equilibrium with NADH produced by the TCA cycle. In the failing heart, NADPH is more oxidized, leading to increased mitochondrial H2O2 formation 45. Increased cytosolic ROS production can activate the mitochondrial permeability transition pore (mPTP), the inner membrane anion channel (IMAC) and the mitochondrial ATP-dependent K+ channel (mito-KATP) found in the internal mitochondrial membrane. ANT is suspected as the core component of mPTP.
Triggered by increased calcium, cyclophylin D induces such ANT conformational change that the mPTP complex becomes freely permeable to any molecule of <1.5kDa 46. This leads to dissipation of transmembrane potential (ΔѰ) and subsequently to amplify electron flux through ETC (to maintain the ΔѰ) at the cost of increased NADH use. This may lead to increased NADPH oxidation and therefore impaired H2O2 detoxification. The opposite effect of ΔѰ dissipation on ROS accumulation by ‘physiologic uncoupling‘(ROS↓) versus cytosol ROS induced pathologic membrane depolarization (ROS↑) needs clarification. However it is possible that while physiologic uncoupling regulates proton gradient by close modulation of electron flow and inhibition of ROS production at the ETC complexes, the excessive pathologic uncoupling by cytosolic ROS (by non-UCP channels and non-specific leak) lead to dramatically increased ETC flux necessary to maintain proton gradient (ΔѰ) and impaired ROS detoxification in the matrix.
It is clear that ROS play an important role in the local pathogenesis of heart failure. This review focuses on the respiratory chain function and its major source of ROS production at the ETC of mitochondria; however, it is important to highlight that there are other intracellular enzymatic sources of ROS such as NADPH oxidase, xanthine oxidase and uncoupled nitric oxide synthases 33. Moreover circulating ROS metabolites could also be used clinically as a marker of heart failure severity and treatment efficiency. Biopyrrins, oxidative metabolites of bilirubin can be non-invasively measured in plasma or urine, and their levels correlated well to BNP levels and the severity of symptoms (NYHA class) in heart failure patients 47.
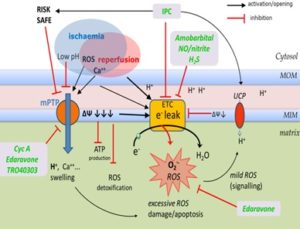
Ischemia-reperfusion injury – “To breathe or not to breathe?: Final infarct size is due to injury conferred during ischemia and also the injury incurred as a result of ischemia-reperfusion injury (IRI). The damage occurring on reperfusion is largely determined by a massive burst of ROS production originating from ischaemically damaged mitochondria. During ischemia intracellular ATP levels and pH drop due to impaired OXPHOS and a switch to anaerobic glycolysis with lactic acid production. The intracellular proton accumulation activates the Na/H antiporter, and sodium enters the intracellular space. The ATP dependent Na/K antiporter is now unable to remove the intracellular Na, and excess Na leads to a reversal of the Na/Ca antiporter with a resultant increase of intracellular calcium and mitochondrial swelling.
The Influx of calcium into the mitochondria and an increase in ROS production both favor the opening of the mitochondrial permeability transition pore (mPTP), but the associated low pH prevents its opening, Fig. 3. ROS production during ischemia is promoted by the accumulation of electrons within the ETC as hypoxia halts, or even reverses the electron flow. ETC complexes are in a reduced state which promotes acceptance of the electrons by the remaining oxygen to form superoxide 48. Following reperfusion pH recovers quickly towards normal and these results in the opening of the mPTP within a few minutes of reperfusion 49, 50. During early reperfusion and reoxygenation, the ischemia-damaged electron transport chain in the presence of an abrupt increase in the flow of accumulated electrons and an associated increase in electron leak (mainly at complex I and III) is responsible for a burst in superoxide production, Fig. 3. 51, 52, 53
Opening of the non-specific mPTP results in sudden dissipation of the electrochemical gradient across the inner mitochondrial membrane causing hydrolysis rather than synthesis of ATP and perpetuation of further ROS production which leads to irreversible oxidization of proteins, DNA, and lipids 54, the release of cytochrome c and activation of apoptotic pathways 49, 55. Reperfusion also results in local and systemic inflammatory reactions involving activation of neutrophils and platelets 56.
The inhibition of mPTP opening has become a common final target for cytoprotective strategies in ischemia-reperfusion injury 46, 57, 58. Cyclosporine A is a direct mPTP opening inhibitor and has been shown to decrease the infarct size following reperfusion in a pilot study of 58 patients presenting with acute STEMI 46, 59, Fig. 3. Another promising agent which is scavenging excess ROS and appears to inhibit mPTP opening is edaravone (MCI-186) 60, Fig. 3. This antioxidant, approved for the treatment of acute ischaemic stroke in Japan and China 61, was evaluated in a clinical trial in which it was administered 10 minutes before reperfusion in acute myocardial infarction and decreased the size and preserved cardiac function (n=80) 62. TRO40303 is another novel cyto-protective agent acting via inhibition of mPTP opening 63, which is currently being investigated for clinical use in acute myocardial infarction 64.
Other experimental strategies comprise induction of upstream endogenous protective mechanisms by ischaemic conditioning or pharmacological targeting of upstream conditioning cascade of cytosol located pro-survival enzymes which inhibit mPTP opening such as RISK (Reperfusion Injury Salvage Kinase) 65, SAFE (Survivor Activating Factor Enhancement) pathways 66 by adenosine, opioids, ANP, PDE5 inhibitors and others and are reviewed in detail elsewhere 57, 67, 68. Activation of AMPK (adenosine mono-phosphate activated protein kinase) which beneficially modulates substrate transport and substrate oxidation in the reperfusion phase is another recognized pathway to prevent IRI 69.
A novel strategy is modulation of the electron transport chain and related mitochondrial ROS production to confer cytoprotection against myocardial injury on reperfusion and is discussed below. Ischaemic conditioning is a strategy to limit myocardial infarction size by induction of ischemia either locally (by intermittent occlusion of the affected coronary vessel) or remotely in a distant organ (typically a limb) inducing myocardium cytoprotection. Ischaemic conditioning can be applied at different time points: before the beginning of ischemia (PRE- conditioning, IPC), during ongoing myocardial ischemia (PER-conditioning, IPerC) or at the onset of reperfusion (POST-conditioning, IPostC).
The ischemia or pharmacological IPerC and IPostC strategies are clinically relevant in the setting of acute myocardial infarction 70, 71; whereas IPC has been successfully used in elective cardiac surgery or elective PCI setting where it reduced infarct size and improved post-ischaemic function 72 - 75, Table 1. One of the cytoprotective mechanisms of IPC is an induction of a slight degree of MIM depolarization which protects against ROS induced damage 76. This mild proton leak induced by acute IPC is mediated mainly by UCPs (UCP 2 and 3) 77. Late IPC leads to an increase in UCP2 expression. On its own, the augmented uncoupling should impair energetic efficiency. However, IPC also increases expression of complex IV and ATP synthase supporting ATP production with a favorable energetic profile during repeated hypoxia 78. Inhibition of complex I (a major ROS source) by acute IPC mediated by reversible s-nitration is another mechanism protecting from damage on reperfusion, Fig. 3 79.
There has been a surge in interest in the potential use of nitrite (NO2-) in the treatment of IRI. Under hypoxic conditions, nitrite can be reduced to nitric oxide (NO) and is thought to act as the largest storage pool for the metabolically active NO 80, 81. Cardioprotection is conferred during ischemia by NO donors 82 inhibiting both complexes I (decreasing ROS production) and complex IV, Fig. 3 83, 84. Under hypoxic conditions, nitrite is reduced to NO and similarly to ischaemic preconditioning confers cytoprotection via blockade and S-nitrosation of complex I in mouse heart during ischemia and reperfusion 79, 82, 85, 86.
We and others are currently undertaking phase II trials investigating the cytoprotective effects of IV nitrite in the early reperfusion phase in patients with ST-elevation myocardial infarction 87, 88, Table 1. Amobarbital, a complex l inhibitor, preserves mitochondrial respiration and decreases myocardial injury both during ischemia 89 and during early reperfusion. It attenuates ROS generation with a consequent decrease in infarct size, Fig. 3 90, 91. Unfortunately despite strong animal experimental evidence of its cytoprotective properties, exerting its action even in aged hearts lacking upstream signaling pathways of post-conditioning 92, this barbiturate narcotic historically used as a “truth serum” has not found its way to human IRI studies yet. Multiple other therapies are currently developed for the treatment of ischaemic heart disease and IRI; however, they often do not target the respiratory chain directly and are reviewed elsewhere 93.
Other Potential Therapeutic Interventions Targeting The Respiratory Chain: Ischemia-reperfusion injury is a classic example where modulation of respiratory chain function has been extensively investigated in an experimental setting, and currently significant efforts are undertaken to translate these results into human applications. However, as described in the previous sections, respiratory chain dysfunction occurs in almost every pathology involving the working heart. Therefore it is not surprising that attempts to modify the electron transport chain to improve myocardial energetics and limit oxidative stress damage are increasingly being investigated in other conditions as well, Table 1.
TABLE 1: TRANSLATIONAL STRATEGIES TARGETING THE RESPIRATORY CHAIN IN CARDIAC DISEASE
| Clinical Usage | Intervention | Target | Clinical Trials | Reference |
| Ischemia-reperfusion injury | Ischaemic conditioning | Complex I, IV; increased uncoupling,
RISK, SAFE
|
AMI, remote preconditioning n=142, myocardial salvage index by perfusion imaging, p=0.03 |
71 |
| Elective CABG, remote pre-conditioning, n=57, Trop-TAUC, p=0.005 | 73 | |||
| Valve replacement surgery, remote preconditioning, n=81, Trop IAUC, p=0.05 | 72 | |||
| nitrite | Complex I and IV | Currently undergoing- NIAMI, multi-centre RCT (iv nitrite)NITRITE-AMI, single centre RCT (intracoronary nitrite) |
NCT01388504
NCT01584453 |
|
| melatonin | Stabilizes MIM preserving complex I and III function mPTP | currently undergoing-single center MARIA (iv in AMI) two center (intracoronary in AMI) | NCT00640094
NCT01172171 |
|
| cyclosporine A | AMI, n=58, CKAUC p=0.04, trop IAUC p= 0.15, MRI p=0.04 | 59 | ||
| TRO40303 | Mitochondrial translocator protein (TSPO), delays mPTP opening | Multi-centre RCT | NCT01374321 | |
| Edaravone (MCI-186) | ROS scavenger | AMI, n=80, CKAUC p=0.04, CK-MBAUC p=0.02 |
62 | |
| Other interventions showing benefit in animal models:
Amobarbital hydrogen sulfide caloric restriction resveratrol |
Complex I
Complex IV Complex I and III |
90, 91, 134
135. 128 |
||
| Heart failure | Coenzyme Q 10 | Corrects coenzyme Q deficit | Coenzyme Q10 + Selenium, 5 year follow up, n=443, CV mortality p=0.015, NT-proBNP 0.014, EF 0.03 | 103 |
| Symbio multi-center RCT | ISRCTN945062 | |||
| Other interventions showing benefit in animal models:
Trimetazidine105 SS-31109 |
Complex I and II
Mitochondria selective antioxidant |
|
117
122 |
|
| LVH | Animal model:
Low-intensity aerobic exercise |
Decreased sensitivity to Ca++ induced mPTP opening | 129 | |
| Aging | Animal models:
caloric restriction
Melatonin |
Complex I electron leak | 126 | |
| Preservation of complex I, III , IV activity | 109 | |||
| Hereditary mitochondrial cardiomyopathies | Pre-natal genetic diagnostics & Gene therapy Low-intensity exercise | Respiratory chain defects | (benefit documented in skeletal muscle) | 136 |
A) Nitrite and Nitrate: Inorganic nitrite (NO2-) or nitrate (NO3-) induced modulations of the respiratory chain could be potentially beneficial for the treatment of peripheral arterial disease 94, angina or heart failure 80. There is limited evidence that it may induce energetically favorable state and improve improved metabolic efficiency. In liver mitochondria NO induces depression of the maximal OXPHOS dependent ATP synthesis and this has been attributed mainly to inhibition of complex I and complex IV. The NO-induced kinetic constraint on complex IV is however more pronounced than the constraint on ATP synthesis leading to improved oxidative phosphorylation efficiency (amount of ATP produced per oxygen molecules consumed) 95.
Though this inhibition may overall result in restricted maximal ATP synthesis capacity which could be detrimental in the highest metabolic demand, it may well be beneficial in situations when hypoxia limits oxygen supply and promote cardiac hibernation, Fig. 3. There is emerging evidence that nitrate/nitrite improves metabolic efficiency of skeletal muscle of healthy volunteers by decreasing oxygen consumption at exercise 96, 97. It was proposed that nitrite may improve coupling of OXPHOX to ATP synthesis, and therefore the efficiency of ATP synthesis 96- 98. This increase in metabolic efficiency may be partly responsible for beneficial effects seen in peripheral artery disease patients with prolonged walking distances after beetroot juice nitrate supplementation 94. If similar action was present in cardiac muscle, this could open up strategies to develop an effective treatment for chronic heart failure patients and angina. However, some of the benefits are likely due to previously demonstrated nitrite-induced vasodilation of hypoxic tissue 99 and better local perfusion due to nitrite-induced angiogenesis 100, 101.
B) Coenzyme Q: Coenzyme Q10 is an important antioxidant and a part of the respiratory chain. Low Coenzyme Q10 levels have been documented in chronic heart failure 102. Its supplementation in smaller studies showed improvement in LVEF and cardiac output in HF patients. Long-term supplementation of a combination of selenium and coenzyme Q10 in an elderly Swedish population resulted in a significant decrease in cardiovascular mortality. The effect was also evident in multivariate analysis when adjusted for risk factors such as heart failure class or ejection fraction 103.
FIG. 3: ISCHAEMIA-REPERFUSION INJURY AND THERAPEUTIC TARGETS. Ischaemia led to impairment of mitochondrial OXPHOS related ATP production due to lack of O2 and decreased electrochemical gradient (ΔΨ) at the MIM. The backlog of substrates in the TCA cycle results in superfluous pyruvate, a substrate of glycolysis which is then diverted to lactate resulting in intracellular acidosis with low pH. H+ is being removed out of the cytosol by Na+/H+ antiporter resulting in intracellular Na+ load, which in turn is exported out of the cell via Na+/Ca++ antiporter leading to cytosolic Ca++ overload, the latter worsened by impaired Ca++ removal by ATP dependent Ca++ -pump. Ischemia-related halt of electron flow at the ETC leads to increased electron leak resulting in pathologic ROS rise. Both Ca++ and ROS favor opening of the mitochondrial permeability transition pore (mPTP), however, this is prevented by low pH during ischemia. On reperfusion, pH returns quickly to normal, which triggers mPTP opening. This results in sudden dissipation of the electrochemical gradient (ΔΨ) across the MIM, a loss of the driving force for ADP phosphorylation, and indeed even reversal inducing ATP dephosphorylation at the ATP synthase to maintain the ΔΨ. A decrease in production plus an increase in consumption of ATP further exacerbates the chemical dysbalance within the cell. On sudden reperfusion abundance of O2 combined with ischemia damaged ETC complexes perpetuates electron leak with massive ROS production, oxidative damage of protein, lipids, DNA and activation of apoptotic pathways. Whereas mild MIM gradient ΔΨ decrease induced by ischaemic preconditioning IPC via activation of uncoupling proteins cytoprotects by inhibition of the electron leak (ROS production) at the ETC, a sudden massive ΔΨ collapse due to mPTP opening at reperfusion promotes perpetuated electron leak (ROS production) at the ECT and simultaneously leads to impaired matrix ROS detoxification. Inhibition of mPTP opening either directly by Cyclosporine A or indirectly via RISK and SAFE pathways by IPC is a major therapeutic target resulting in reperfusion cytoprotection. Another novel strategy is direct inhibition of ETC complexes electron flow by nitrite, nitric oxide, amobarbital or H2S. The sudden massive increase in electron flow on reperfusion through ischemia damaged ETC complexes is slowed down, with a concomitant decrease in absolute electron leak, ROS production and therefore is cytoprotective.
Recently the preliminary results of a multi-center randomized control trial Q-Symbio were presented. Four hundred twenty patients with severe heart failure (NYHA III-IV) were randomized to receive either CoQ10 or placebo. CoQ10 decreased the risk of MACE (= hospitalization, CV death, mechanical circulatory support or cardiac transplantation) from 14% to 25% and halved the risk of dying from all causes compared to placebo 104, 105. The Q-Symbio data have to be regarded with caution as the full data are still to be published, however if it stands the post-publication peer-review then this could be a breakthrough for medication which act by augmentation of the energy production, rather than just inhibiting less effective pathways or preventing negative impact of pathologic remodelling in heart failure.
Beer (even alcohol-free) inhibits the enzymatic activity of complexes I and IV and decreases the oxidation of Coenzyme Q9 and Q10 in adriamycin-treated rats leading to decreased damage of mitochondrial components and preventing mitochondrial dysfunction 106. ROS reactive oxygen/nitrogen species, O2.- superoxide anion, ETC electron transport chain, mPTP mitochondrial permeability transition pore, UCP uncoupling proteins, MOM mitochondrial outer membrane, MIM mitochondrial inner membrane, RISK Reperfusion Injury Salvage Kinase pathway, SAFE Survivor Activating Factor Enhancement pathway, Cyc A cyclosporine A, NO nitric oxide, IPC ischaemic preconditioning
C) Melatonin: Melatonin is found in high concentration in mitochondria where it stabilizes the mitochondrial inner membrane and improves the activity of the ETC. It protects against ROS induced cardiolipin peroxidation which would otherwise promote cytochrome c detachment and mPTP opening 107. Melatonin protects myocardium from ischaemic reperfusion injury, lowering lipid peroxidation, preserving mitochondrial respiration, and preventing loss of function of complex I, and III and improves post-ischaemic hemodynamic function in isolated heart 107. The ongoing phase II trial MARIA is investigating if melatonin confers cardioprotection in patients presenting with myocardial infarction undergoing primary angioplasty 108. There may also be a role in protection against the consequences of aging as chronic melatonin administration reduces oxidative damage and mitochondrial function in hearts from senescence-accelerated mice 109.
D) Trimetazidine: Trimetazidine (TZD), an anti-anginal drug has been shown to improve myocardial function in both patients with ischaemic heart disease 110 or with idiopathic DCM 111 while preserving an advantageous energetic profile 112, 113. A favourable metabolic modulation 114 by a switch from fatty acid oxidation to glucose oxidation via inhibition of long-chain 3-ketoacyl CoA thiolase activity 115 may play only a part in the observed beneficial effects.
Further evidence, however, suggests that modulation of the ETC may be pivotal in the cytoprotection conferred by TZD. It protects cardiomyocytes in animal models of IRI 116 or HF 117 by inhibition of Ca2++ induced mPTP opening. In myocytes from failing hearts, an enhanced electron leak at complex II was suppressed by TZD, and hence the ROS generation was attenuated; the restoration of the redox balance by TZD was accompanied by an improvement of impaired activity of complex I 117.
E) Antioxidants: Multiple antioxidant agents have been investigated for their potential to reduce cardiovascular events, via oxidative stress reduction. These include Vitamin E 118, 119, or omega 3 120. Unfortunately, while many of these treatments show beneficial effects under experimental conditions, outcomes in human trials have been mixed and do not seem to translate into reduced mortality 121. One of the reasons for the lack of antioxidant effects may be the recognized concept of compartmentalized signaling. Mitochondrial ROS signaling may be dependent on localized proximity to target molecules, which may not reflect changes in their global concentration or effects on different isoforms of target proteins (with opposite effects) found in different compartments 42. This is supported by findings showing amelioration of experimental angiotensin II-induced cardiomyopathy by targeted mitochondrial ROS scavenging with SS-31 (a ROS scavenging peptide which accumulates > 1000 fold in mitochondria), but no effect of non-targeted ROS scavenger N-acetyl-cysteine (NAC) in the same experiment 122.
F) Caloric Restriction, Resveratrol, and Exercise: Caloric restriction (CR) is unique in that it has been shown to increase maximum life span in mammals 123, 124; possibly via the induction of autophagic pathways and mitochondrial biogenesis 125, and reduction of complex I related ROS production 126. It can ameliorate aging-associated changes in human cardiac diastolic function 127. CR preserves post-ischaemic mitochondrial respiration and attenuates post-ischaemic mitochondrial H2O2 production 128. Treatment with resveratrol (natural polyphenol) mimicked the effect of CR attenuating ROS production in ischemia and reoxygenation. Both CR and resveratrol appear to protect from oxidative stress by deacetylation of specific ETC proteins 128. Low-intensity exercise is known to attenuate pathological LV remodeling in human heart failure. In a swine model of pressure overload, low level, aerobic exercise prevented LV hypertrophy and systolic function. These beneficial changes were accompanied by attenuation of mitochondrial dysfunction 129.
Future Directions: A wealth of evidence is currently available to confirm the major role of mitochondrial respiratory dysfunction in metabolic disorders of the heart. An exciting novel approach to identify new cardioprotective agents is the use of high-throughput tests measuring cellular respiration following various stressors by screening blindly thousands of small molecules from commercially available chemical compound libraries 130, 131. Identified candidates are then subjected to more rigorous bench testing.
This approach can perpetuate the finding of new agents targeting mitochondrial function. Unfortunately, the reality is that the complex physiology of mitochondrial metabolism and artificial experimental methodology are among the main reasons why many previously hailed therapeutic strategies failed later in human experiments. Many experiments are performed on isolated mitochondria, which although easier to obtain and work with; they lack the cellular context 132. Experiments assessing mitochondrial function in the context of whole permeabilized fibers are more challenging, and also its physiology still leaves scope for error due to lack of organismal context 132. In respect of mitochondrial function and identification of the individual targets, proteomics and metabolomics approaches may prove crucial shortly. The picture becomes even more complex when various disease models are used. One attempt to overcome the variation in experimental ischemia-reperfusion models is CAESAR (Consortium for Preclinical Assessment of Cardioprotective Therapies) 133. Its mission is to introduce the same systematic randomization, standardized protocols, and statistical rigor to preclinical studies and bridge these to clinical trials. Similar structured approaches should be attempted for studies into other conditions such as heart failure models or the aging heart. Despite early days and multiple previous failures to translate promising in-vitro data into a clinical setting, we are currently witnessing the first few therapies succeeding in their translation.
CONCLUSION: Cardiac function is dependent on mitochondrial aerobic energy delivery by oxidative phosphorylation. However, the respiratory chain complex is important not only in aerobic energy delivery but also in the regulation of oxidative stress and cell signaling. There is a growing body of evidence suggesting a pivotal role of respiratory chain dysfunction in the pathogenesis of common cardiac conditions such as heart failure or ischemia-reperfusion injury. Understanding the molecular biology of these conditions is the premise for the successful development of therapeutic and preventative targets. Potential treatment strategies are currently being translated from the bench to the bedside.
FINANCIAL SUPPORT AND SPONSORSHIP: This study was supported by the authors named in this article.
CONTRIBUTION OF AUTHORS: This work was done by the authors named in this article and all liabilities about claims relating to the content of this article was borne by the authors named in this article.
ETHICAL APPROVAL: This review does not contain any studies with human participants or animals and was performed by the authors alone.
ACKNOWLEDGEMENT: Us acknowledgments and gratefulness at the beginning and at last is to God who gave us the gift of the mind. The authors thank Young Researchers and Elite Club, Yasooj Branch, Islamic Azad University due to cooperation in this study.
CONFLICT OF INTEREST: The authors declare that there is no conflict of interest regarding this study.
REFERENCES:
- Neubauer S: The failing heart - An engine out of fuel. The New England Journal of Medicine 2007; 356(11): 1140-1151.
- Murray AJ, Anderson RE, Watson GC, Radda GK and Clarke K: Uncoupling proteins in human heart. Lancet 2004; 364(9447): 1786-1788.
- Mailloux RJ and Harper M: Uncoupling proteins and the control of mitochondrial reactive oxygen species production. Free Radical Biology and Medicine 2011; 51(6): 1106-1115.
- Brand MD: Uncoupling to survive? The role of mitochondrial inefficiency in ageing. Experimental Gerontology 2000; 35(6-7): 811-820.
- Chan SL, Wei Z, Chigurupati S and Tu W: Compromised respiratory adaptation and thermoregulation in aging and age-related diseases. Ageing Research Reviews 2010; 9(1): 20-40.
- Jastroch M, Divakaruni AS, Mookerjee S, Treberg JR and Brand MD: Mitochondrial proton and electron leaks. Essays in Biochemistry 2010; 47: 53-67.
- Dranka BP, Benavides GA, Diers AR, Giordano S, Zelickson BR and Reily C: Assessing bioenergetic function in response to oxidative stress by metabolic profiling. Free Radical Biology and Medicine 2011; 51(9): 1621-1635.
- Wallace DC: A mitochondrial paradigm of metabolic and degenerative diseases, aging, and cancer: A dawn for evolutionary medicine. Annual Review of Genetics 2005; 39:359-407.
- Shoubridge EA: Nuclear genetic defects of oxidative phosphorylation. Human Molecular Genetics 2001; 10(20): 2277-2284.
- Rötig A: Genetic bases of mitochondrial respiratory chain disorders. Diabetes & Metabolism 2010; 36(2): 97-107.
- Giordano C, Perli E, Orlandi M, Pisano A, Tuppen HA and He L: Cardiomyopathies due to homoplasmic mitochondrial tRNA mutations: morphologic and molecular features. Human Pathology 2013; 44(7): 1262-1270.
- DiMauro S and Schon EA: Mitochondrial respiratory-chain diseases. The New England Journal of Medicine 2003; 348(26): 2656-2668.
- Bates MGD, Bourke JP, Giordano C, D' Amati G, Turnbull DM and Taylor RW: Cardiac involvement in mitochondrial DNA disease: Clinical spectrum, diagnosis, and management. European Heart Journal 2012; 33(24): 3023-3033.
- Bleyl SB, Mumford BR, Thompson V, Carey JC, Pysher TJ and Chin TK: Neonatal, lethal noncompaction of the left ventricular myocardium is allelic with Barth syndrome. The American Journal of Human Genetics 1997; 61(4): 868-872.
- Leone O, Veinot JP, Angelini A, Baandrup UT, Basso C and Berry G: 2011 Consensus statement on endomyocardial biopsy from the Association for European Cardiovascular Pathology and the Society for Cardiovascular Pathology. Cardiovascular Pathology 2012; 21(4): 245-274.
- Harman D: Free radical theory of aging. Mutation Research 1992; 275(3-6): 257-266.
- Short KR, Bigelow ML, Kahl J, Singh R, Coenen-Schimke J and Raghavakaimal S: Decline in skeletal muscle mitochondrial function with aging in humans. Proceedings of the National Academy of Sciences 2005; 102(15): 5618-5623.
- Richter C, Park JW and Ames BN: Normal oxidative damage to mitochondrial and nuclear DNA is extensive. Proceedings of the National Academy of Sciences 1988; 85(17): 6465-6467.
- Hagen TM, Moreau R, Suh JH and Visioli F: Mitochondrial decay in the aging rat heart: Evidence for improvement by dietary supplementation with acetyl-L-carnitine and/or lipoic acid. Annals of the New York Academy of Sciences 2002; 959: 491-507.
- Chaudhary KR, El-Sikhry H and Seubert JM: Mitochondria and the aging heart. Journal of Geriatric Cardiology 2011; 8(3): 159-167.
- Sun J, Folk D, Bradley TJ and Tower J: Induced over-expression of mitochondrial Mn-superoxide dismutase extends the life span of adult Drosophila melanogaster. Genetics 2002; 161(2): 661-672.
- Schriner SE and Linford NJ: Extension of mouse lifespan by overexpression of catalase. Age 2006; 28(2): 209-218.
- Nabben M, Hoeks J, Briedé JJ, Glatz JFC, Moonen-Kornips E and Hesselink MKC: The effect of UCP3 overexpression on mitochondrial ROS production in skeletal muscle of young versus aged mice. FEBS Letters 2008; 582(30): 4147-4152.
- Speakman JR, Talbot DA, Selman C, Snart S, McLaren JS and Redman P: Uncoupled and surviving: Individual mice with high metabolism have greater mitochondrial uncoupling and live longer. Aging Cell 2004; 3(3): 87-95.
- Cerqueira FM, Laurindo FRM and Kowaltowski AJ: Mild mitochondrial uncoupling and calorie restriction increase fasting eNOS, Akt and mitochondrial biogenesis. PLoS One 2011; 6(3): e18433
- Trifunovic A, Hansson A, Wredenberg A, Rovio AT, Dufour E and Khvorostov I: Somatic mtDNA mutations cause aging phenotypes without affecting reactive oxygen species production. Proceedings of the National Academy of Sciences 2005; 102(50): 17993-17998.
- Cuervo AM, Bergamini E, Brunk UT, Dröge W, Ffrench M and Terman A: Autophagy and aging: the importance of maintaining "clean" cells. Autophagy 2005; 1(3): 131-140.
- Liesa M, Palacín M and Zorzano A: Mitochondrial dynamics in mammalian health and disease. Physiological Reviews 2009; 89(3): 799-845.
- Taneike M, Yamaguchi O, Nakai A, Hikoso S, Takeda T and Mizote I: Inhibition of autophagy in the heart induces age-related cardiomyopathy. Autophag 2010; 6(5): 600 06.
- Giordano FJ: Oxygen, oxidative stress, hypoxia, and heart failure. The Journal of Clinical Investigation 2005; 115(3): 500-508.
- Sabri A, Hughie HH and Lucchesi PA: Regulation of hypertrophic and apoptotic signaling pathways by reactive oxygen species in cardiac myocytes. Antioxidants & Redox Signaling 2003; 5(6):731-740.
- Kindo M, Gerelli S, Bouitbir J, Charles AL, Zoll J and Minh TH: Pressure overload-induced mild cardiac hypertrophy reduces left ventricular transmural differences in mitochondrial respiratory chain activity and increases oxidative stress. Frontiers in Physiology 2012; 3: 332. Doi:10.3389/fphys.2012.00332
- Tsutsui H, Kinugawa S and Matsushima S: Oxidative stress and heart failure. American journal of physiology. Heart and Circulatory Physiology 2011; 301(6): 2181-2190.
- Griffiths ER, Friehs I, Scherr E, Poutias D, McGowan FX and del Nido PJ: Electron transport chain dysfunction in neonatal pressure-overload hypertrophy precedes cardiomyocyte apoptosis independent of oxidative stress. The Journal of Thoracic and Cardiovascular Surgery 2010; 139(6): 1609-1617.
- Sharov VG, Goussev A, Lesch M, Goldstein S and Sabbah HN: Abnormal mitochondrial function in the myocardium of dogs with chronic heart failure. Journal of Molecular and Cellular Cardiology 1998; 30(9): 1757-1762.
- Sharov VG, Todor AV, Silverman N, Goldstein S and Sabbah HN: Abnormal mitochondrial respiration in failed human myocardium. Journal of Molecular and Cellular Cardiology 2000; 32(12): 2361-2367.
- Gao X, Campian JL, Qian M, Sun XF and Eaton JW: Mitochondrial DNA damage in iron overload. The Journal of Biological Chemistry 2009; 284(8): 4767-4775.
- Gao X, Qian M, Campian JL, Marshall J, Zhou Z and Roberts AM: Mitochondrial dysfunction may explain the cardiomyopathy of chronic iron overload. Free Radical Biology & Medicine 2010; 49(3): 401-407.
- Báez A, Presti MSL, Rivarola HW, Mentesana GG, Pons P and Fretes R: Mitochondrial involvement in chronic chagasic cardiomyopathy. Transactions of the Royal Society of Tropical Medicine and Hygiene 2011; 105(5): 239-246.
- Scheubel RJ, Tostlebe M, Simm A, Rohrbach S, Prondzinsky R and Gellerich FN: Dysfunction of mitochondrial respiratory chain complex I in human failing myocardium is not due to disturbed mitochondrial gene expression. Journal of the American College of Cardiology 2002; 40(12): 2174-2181.
- Jarreta D, Orús J, Barrientos A, Miró O, Roig E and Heras M: Mitochondrial function in heart muscle from patients with idiopathic dilated cardiomyopathy. Cardiovascular Research 2000; 45(4): 860-865.
- Arbustini E, Diegoli M, Fasani R, Grasso M, Morbini P and Banchieri N: Mitochondrial DNA mutations and mitochondrial abnormalities in dilated cardiomyopathy. American Journal of Pathology 1998; 153(5): 1501-1510.
- Ide T, Tsutsui H, Kinugawa S, Utsumi H, Kang D and Hattori N: Mitochondrial electron transport complex I is a potential source of oxygen free radicals in the failing myocardium. Circulation Research 1999; 85(4): 357-363.
- Sam F, Kerstetter DL, Pimental DR, Mulukutla S, Tabaee A and Bristow MR: Increased reactive oxygen species production and functional alterations in antioxidant enzymes in human failing myocardium. Journal of Cardiac Failure 2005; 11(6): 473-480.
- Maack C and Bhm M: Targeting mitochondrial oxidative stress in heart failure: Throttling the afterburner. Journal of the American College of Cardiology 2011; 58(1):83-86.
- Halestrap AP, Clarke SJ and Javadov SA: Mitochondrial permeability transition pore opening during myocardial reperfusion - A target for cardioprotection. Cardiovascular Research 2004; 61(3): 372-385.
- Hokamaki J, Kawano H, Yoshimura M, Soejima H, Miyamoto S and Kajiwara I: Urinary biopyrrins levels are elevated about the severity of heart failure. Journal of the American College of Cardiology 2004; 43(10): 1880-1885.
- Murillo D, Kamga C, Mo L and Shiva S: Nitrite as a mediator of ischemic preconditioning and cytoprotection. Nitric Oxide 2011; 25(2):70-80.
- Halestrap AP: Mitochondria and reperfusion injury of the heart-A holey death but not beyond salvation. Journal of Bioenergetics and Biomembranes 2009; 41(2): 113-121.
- Griffiths EJ and Halestrap AP: Mitochondrial non-specific pores remain closed during cardiac ischemia, but open upon reperfusion. Biochemical Journal 1995; 307(1): 93-98.
- Chen YR, Chen CL, Pfeiffer DR and Zweier JL: Mitochondrial complex II in the post-ischemic heart: Oxidative injury and the role of protein S-glutathionylation. The Journal of Biological Chemistry 2007; 282(45): 32640-32654.
- Becker LB: New concepts in reactive oxygen species and cardiovascular reperfusion physiology. Cardiovascular Research 2004; 61(3): 461-470.
- O'Rourke B, Cortassa S and Aon MA: Mitochondrial ion channels: Gatekeepers of life and death. Physiology 2005; 20(5): 303-315.
- Ambrosio G, Zweier JL, Duilio C, Kuppusamy P, Santoro G and Elia PP: Evidence that mitochondrial respiration is a source of potentially toxic oxygen free radicals in intact rabbit hearts subjected to ischemia and reflow. The Journal of Biological Chemistry 1993; 268(25): 18532-18541.
- Murphy E and Steenbergen C: Ion transport and energetics during cell death and protection. Physiology 2008; 23(2): 115-123.
- Frangogiannis NG, Smith CW and Entman ML: The inflammatory response in myocardial infarction. Cardiovascular Research 2002; 53(1): 31-47.
- Kharbanda RK: Cardiac conditioning: A review of evolving strategies to reduce ischaemia - Reperfusion injury. Heart 2010; 96(15): 1179-1186.
- Javadov S, Karmazyn M and Escobales N: Mitochondrial permeability transition pore opening as a promising therapeutic target in cardiac diseases. Journal of Pharmacology and Experimental Therapeutics 2009; 330(3): 670-678.
- Piot C, Croisille P, Staat P, Thibault H, Rioufol G and Mewton N: Effect of cyclosporine on reperfusion injury in acute myocardial infarction. The New England Journal of Medicine 2008; 359(5): 473-481.
- Rajesh KG, Sasaguri S, Suzuki R and Maeda H: Antioxidant MCI-186 inhibits mitochondrial permeability transition pore and upregulates Bcl-2 expression. American Journal of Physiology. Heart and Circulatory Physiology 2003; 285(5): H2171-H2178.
- Feng S, Yang Q, Liu M, Li W, Yuan W and Zhang S: Edaravone for acute ischaemic stroke. The Cochrane Database of Systematic Reviews 2008; Doi: 10.1002/14651858.CD007230.pub2
- Tsujita K, Shimomura H, Kawano H, Hokamaki J, Fukuda M and Yamashita T: Effects of edaravone on reperfusion injury in patients with acute myocardial infarction. American Journal of Cardiology 2004; 94(4):481-484.
- Schaller S, Paradis S, Ngoh GA, Assaly R, Buisson B and Drouot C: TRO40303, a new cardioprotective compound, inhibits mitochondrial permeability transition. Journal of Pharmacology and Experimental Therapeutics 2010; 333(3): 696-706.
- Safety and Efficacy Study of TRO40303 for Reduction of Reperfusion Injury in Patients Undergoing Percutaneous Coronary Intervention for Acute Myocardial Infarction Available at http://clinicaltrials.gov/show/NCT01374321.
- Hausenloy DJ and Yellon DM: New directions for protecting the heart against ischemia-reperfusion injury: Targeting the Reperfusion Injury Salvage Kinase (RISK)-pathway. Cardiovascular Research 2004; 61(3): 448-460.
- Lacerda L, Somers S, Opie LH and Lecour S: Ischaemic postconditioning protects against reperfusion injury via the SAFE pathway. Cardiovascular Research 2009; 84(2): 201-208.
- Minamino T: Cardioprotection from ischemia/reperfusion injury: Basic and translational research. Circulation Journal 2012; 76(5): 1074-1082.
- Ebner B, Ebner A, Reetz A, Böhme S, Schauer A and Strasser RH: Pharmacological postconditioning by bolus injection of phosphodiesterase-5 inhibitors vardenafil and sildenafil. Molecular and Cellular Biochemistry 2013; 379(1-2): 43-49.
- Nagendran J, Waller TJ and Dyck JRB: AMPK signaling and the control of substrate use in the heart. Molecular and Cellular Endocrinology 2013; 366(2): 180-193.
- Schmidt MR, Smerup M, Konstantinov IE, Shimizu M, Li J and Cheung M: Intermittent peripheral tissue ischemia during coronary ischemia reduces myocardial infarction through a K ATP-dependent mechanism: First demonstration of remote ischemic preconditioning. American Journal of Physiology. Heart and Circulatory Physiology 2007; 292(4): H1883-H1890.
- Bøtker HE, Kharbanda R, Schmidt MR, Bøttcher M, Kaltoft AK and Terkelsen CJ: Remote ischaemic conditioning before hospital admission, as a complement to angioplasty, and effect on myocardial salvage in patients with acute myocardial infarction: a randomized trial. Lancet 2010; 375(9716): 727-734.
- Li L, Luo W, Huang L, Zhang W, Gao Y and Jiang H: Remote preconditioning reduces myocardial injury in adult valve replacement: A randomized controlled trial. Journal of Surgical Research 2010; 164(1): e21-e26.
- Hausenloy DJ, Mwamure PK, Venugopal V, Harris J, Barnard M and Grundy E: Effect of remote ischaemic preconditioning on myocardial injury in patients undergoing coronary artery bypass graft surgery: a randomised controlled trial. Lancet 2007; 370(9587): 575-579.
- Hoole SP, Heck PM, Sharples L, Khan SN, Duehmke R and Densem CG: Cardiac remote ischemic preconditioning in coronary stenting (CRISP stent) study. A prospective, randomized control trial. Circulation 2009; 119(6): 820-827.
- Thielmann M, Kottenberg E, Kleinbongard P, Wendt D, Gedik N and Pasa S: Cardioprotective and prognostic effects of remote ischaemic preconditioning in patients undergoing coronary artery bypass surgery: A single-centre randomised, double-blind, controlled trial. Lancet 2013; 382(9892): 597-604.
- Brookes PS: Mitochondrial H+ leak and ROS generation: An odd couple. Free Radical Biology & Medicine 2005; 38(1): 12-23.
- Nadtochiy SM, Tompkins AJ and Brookes PS: Different mechanisms of mitochondrial proton leak in ischaemia/ reperfusion injury and preconditioning: Implications for pathology and cardioprotection. Biochemical Journal 2006; 395(3): 611-618.
- Cabrera JA, Ziemba EA, Colbert R, Anderson LB, Sluiter W and Duncker DJ: Altered expression of mitochondrial electron transport chain proteins and improved myocardial energetic state during late ischemic preconditioning. American Journal of Physiology. Heart and Circulatory Physiology 2012; 302(10): H1974-H1982.
- Burwell LS, Nadtochiy SM, Tompkins AJ, Young S and Brookes PS: Direct evidence for S-nitrosation of mitochondrial complex I. Biochemistry Journal 2006; 394(3): 627-634.
- Lundberg JO, Weitzberg E and Gladwin MT: The nitrate-nitrite-nitric oxide pathway in physiology and therapeutics. Nature Reviews Drug Discovery 2008; 7(2): 156-167.
- Zweier JL, Li H, Samouilov A and Liu X: Mechanisms of nitrite reduction to nitric oxide in the heart and vessel wall. Nitric Oxide 2010; 22(2): 83-90.
- Nadtochiy SM, Burwell LS and Brookes PS: Cardioprotection and mitochondrial S-nitrosation: Effects of S-nitroso-2-mercaptopropionyl glycine (SNO-MPG) in cardiac ischemia-reperfusion injury. Journal of Molecular and Cellular Cardiology 2007; 42(4): 812-825.
- Cooper CE and Brown GC: The inhibition of mitochondrial cytochrome oxidase by the gases carbon monoxide, nitric oxide, hydrogen cyanide and hydrogen sulfide: Chemical mechanism and physiological significance. Journal of Bioenergetics and Biomembranes 2008; 40(5): 533-539.
- Murphy EJ, Metcalfe CL, Basran J, Moody PCE and Raven EL: Engineering the substrate specificity and reactivity of a heme protein: creation of an ascorbate binding site in cytochrome C peroxidase. Biochemistry (NY) 2008; 47(52): 13933-13941.
- Nadtochiy SM, Burwell LS, Ingraham CA, Spencer CM, Friedman AE and Pinkert CA: In-vivo cardioprotection by S-nitroso-2-mercaptopropionyl glycine. Journal of Molecular and Cellular Cardiology 2009; 46(6): 960-968.
- Shiva S, Sack MN, Greer JJ, Duranski M, Ringwood LA and Burwell L: Nitrite augments tolerance to ischemia/ reperfusion injury via the modulation of mitochondrial electron transfer. The Journal of Experimental Medicine 2007; 204(9): 2089-2102.
- http://clinicaltrials.gov/ct2/show/NCT01388504.
- Safety and Effectiveness of Intra-coronary Nitrite in Acute Myocardial Infarction (NITRITE-AMI)http://clinicaltrials. gov/ show/NCT01584453.
- Chen Q, Paillard M, Gomez L, Li H, Hu Y and Lesnefsky EJ: Postconditioning modulates ischemia-damaged mito-chondria during reperfusion. Journal of Cardiovascular Pharmacology 2012; 59(1): 101-108.
- Stewart S, Lesnefsky EJ and Chen Q: Reversible blockade of electron transport with amobarbital at the onset of reperfusion attenuates cardiac injury. Translational Research 2009; 153(5): 224-231.
- Chen Q, Camara AKS, Stowe DF, Hoppel CL and Lesnefsky EJ: Modulation of electron transport protects cardiac mitochondria and decreases myocardial injury during ischemia and reperfusion. American Journal of Physiology. Cell Physiology 2007; 292(1): C137-C147.
- Chen Q, Ross T, Hu Y and Lesnefsky EJ: Blockade of electron transport at the onset of reperfusion decreases cardiac injury in aged hearts by protecting the inner mitochondrial membrane. Journal of Aging Research 2012; Doi:10.1155/2012/753949
- Walters AM, Porter GA and Brookes PS: Mitochondria as a drug target in ischemic heart disease and cardio-myopathy. Circulation Research 2012; 111(9): 1222-1236.
- Kenjale AA, Ham KL, Stabler T, Robbins JL, Johnson JL and VanBruggen M: Dietary nitrate supplementation enhances exercise performance in peripheral arterial disease. J of Applied Physiology 2011; 110(6): 1582-91.
- Clerc P, Rigoulet M, Leverve X and Fontaine E: Nitric oxide increases oxidative phosphorylation efficiency. Journal of Bioenergetics and Biomembranes 2007; 39(2): 158-166.
- Bailey SJ, Winyard P, Vanhatalo A, Blackwell JR, DiMenna FJ, Wilkerson DP and: Dietary nitrate supplementation reduces the O2 cost of low-intensity exercise and enhances tolerance to high-intensity exercise in humans. Journal of Applied Physiology 2009; 107(4): 1144-1155.
- Larsen FJ, Weitzberg E, Lundberg JO and Ekblom B: Dietary nitrate reduces maximal oxygen consumption while maintaining work performance in maximal exercise. Free Radical Biology & Medicine 2010; 48(2): 342-347.
- Larsen FJ, Schiffer TA, Borniquel S, Sahlin K, Ekblom B and Lundberg JO: Dietary inorganic nitrate improves mitochondrial efficiency in humans. Cell Metabolism 2011; 13(2): 149-159.
- Maher AR, Milsom AB, Gunaruwan P, Abozguia K, Ahmed I and Weaver RA: Hypoxic modulation of exogenous nitrite-induced vasodilation in humans. Circulation 2008; 117(5): 670-677.
- Kumar D, Branch BG, Pattillo CB, Hood J, Thoma S and Simpson S: Chronic sodium nitrite therapy augments ischemia-induced angiogenesis and arteriogenesis. Proceedings of the National Academy of Sciences 2008; 105(21): 7540-7545.
- Hendgen-Cotta UB, Luedike P, Totzeck M, Kropp M, Schicho A and Stock P: Dietary nitrate supplementation improves revascularization in chronic ischemia. Circulation 2012; 126(16): 1983-1992.
- Molyneux SL, Florkowski CM, George PM, Pilbrow AP, Frampton CM and Lever M: Coenzyme Q 10: An independent predictor of mortality in chronic heart failure. Journal of the American College of Cardiology 2008; 52(18): 1435-1441.
- Alehagen U, Johansson P, Björnstedt M, Rosén A and Dahlström U: Cardiovascular mortality and N-terminal-proBNP reduced after combined selenium and coenzyme Q10 supplementation: A 5-year prospective randomized double-blind placebo-controlled trial among elderly Swedish citizens. International Journal of Cardiology 2013; 167(5): 1860-1866.
- Q-Symbio. http://isrctn.org/ISRCTN94506234.
- Mortensen SA, Rosenfeldt F, Kumar A, Dolliner P, Filipiak KJ and Pella D: The effect of coenzyme Q10 on morbidity and mortality in chronic heart failure: results from Q-SYMBIO: a randomized double-blind trial. JACC Heart Failure 2014; 2(6):641-649.
- Valls-Belles V, Torres C, Muñiz P and Codoñer-Franch P: Effect of beer consumption on levels of complex i and complex IV liver and heart mitochondrial enzymes and coenzymes Q9 and Q10 in adriamycin-treated rats. European Journal of Nutrition 2010; 49(3): 181-187.
- Paradies G, Petrosillo G, Paradies V, Reiter RJ and Ruggiero FM: Melatonin, cardiolipin and mitochondrial bioenergetics in health and disease. Journal of Pineal Research 2010; 48(4): 297-310.
- MARIA Trial. Available at:http://clinicaltrials.gov /ct2/show/NCT00640094.
- Rodríguez MI, Carretero M, Escames G, López LC, Maldonado MD and Tan DX: Chronic melatonin treatment prevents age-dependent cardiac mitochondrial dysfunction in senescence-accelerated mice. Free Radical Research 2007; 41(1): 15-24.
- Vitale C, Wajngaten M, Sposato B, Gebara O, Rossini P, Fini M and et al.: Trimetazidine improves left ventricular function and quality of life in elderly patients with coronary artery disease. European Heart Journal 2004; 25(20): 1814-1821.
- Tuunanen H, Engblom E, Naum A, Någren K, Scheinin M, Hesse B and: Trimetazidine, a metabolic modulator, has cardiac and extracardiac benefits in idiopathic dilated cardiomyopathy. Circulation 2008; 118(12): 1250-1258.
- Fragasso G, Perseghin G, De Cobelli F, Esposito A, Palloshi A and Lattuada G: Effects of metabolic modulation by trimetazidine on left ventricular function and phosphocreatine/adenosine triphosphate ratio in patients with heart failure. European Heart Journal 2006; 27(8): 942-948.
- Fragasso G, Salerno A, Lattuada G, Cuko A, Calori G and Scollo A: Effect of partial inhibition of fatty acid oxidation by trimetazidine on whole-body energy metabolism in patients with chronic heart failure. Heart 2011; 97(18): 1495-1500.
- Ashrafian H, Frenneaux MP and Opie LH: Metabolic mechanisms in heart failure. Circulation 2007; 116(4): 434-448.
- Kantor PF, Lucien A, Kozak R and Lopaschuk GD: The antianginal drug trimetazidine shifts cardiac energy metabolism from fatty acid oxidation to glucose oxidation by inhibiting mitochondrial long-chain 3-ketoacyl coenzyme A thiolase. Circulation Research 2000; 86(5): 580-588.
- Argaud L, Gomez L, Gateau-Roesch O, Couture-Lepetit E, Loufouat J and Robert D: Trimetazidine inhibits mitochondrial permeability transition pore opening and prevents lethal ischemia-reperfusion injury. Journal of Molecular and Cellular Cardiology 2005; 39(6): 893-899.
- Dedkova EN, Seidlmayer LK and Blatter LA: Mitochondria-mediated cardioprotection by trimetazidine in rabbit heart failure. Journal of Molecular and Cellular Cardiology 2013; 59: 41-54.
- Naidoo N, van Dam RM, Koh WP, Chen C, Lee YP and Yuan JM: Plasma vitamin E and coenzyme Q10 are not associated with a lower risk of acute myocardial infarction in Singapore Chinese adults. Journal of Nutrition 2012; 142(6): 1046-1052.
- Shekelle PG1, Morton SC, Jungvig LK, Udani J, Spar M and Tu W: Effect of supplemental vitamin E for the prevention and treatment of cardiovascular disease. Journal of General Internal Medicine 2004; 19(4): 380-89.
- Kwak SM, Myung SK, Lee YJ and Seo HG: Korean Meta-analysis Study Group. Efficacy of omega-3 fatty acid supplements (eicosapentaenoic acid and docosahexaenoic acid) in the secondary prevention of cardiovascular disease: A meta-analysis of randomized, double-blind, placebo-controlled trials. Archives of Internal Medicine 2012; 172(9): 686-694. doi: 10.1001/archinternmed. 2012.262
- Bayeva M, Gheorghiade M and Ardehali H: Mitochondria as a therapeutic target in heart failure. Journal of the American College of Cardiology 2013; 61(6): 599-610.
- Dai DF, Chen T, Szeto H, Nieves-Cintrón M, Kutyavin V and Santana LF: Mitochondrial targeted antioxidant peptide ameliorates hypertensive cardiomyopathy. J of the American College of Cardiology 2011; 58(1): 73-82.
- Barja G: Aging in vertebrates, and the effect of caloric restriction: A mitochondrial free radical production-DNA damage mechanism? Biological Reviews of the Cambridge Philosophical Society 2004; 79(2): 235-251.
- Redman LM and Ravussin E: Caloric restriction in humans: Impact on physiological, psychological, and behavioral outcomes. Antioxidants & Redox Signaling 2011; 14(2): 275-287.
- Gottlieb RA and Mentzer RM: Autophagy during cardiac stress: Joys and frustrations of autophagy. Annual Review of Physiology 2010; 72: 45-59.
- Gredilla R, Sanz A, Lopez-Torres M and Barja G: Caloric restriction decreases mitochondrial free radical generation at complex I and lowers oxidative damage to mitochondrial DNA in the rat heart. The FASEB Journal 2001; 15(9):1589-1591.
- Meyer TE, Kovács SJ, Ehsani AA, Klein S, Holloszy JO and Fontana L: Long-term caloric restriction ameliorates the decline in diastolic function in humans. Journal of the American College of Cardiology 2006; 47(2):398-402.
- Shinmura K, Tamaki K, Sano M, Nakashima-Kamimura N, Wolf AM and Amo T: Caloric restriction primes mitochondria for ischemic stress by deacetylating specific mitochondrial proteins of the electron transport chain. Circulation Research 2011; 109(4): 396-406.
- Emter CA and Baines CP: Low-intensity aerobic interval training attenuates pathological left ventricular remodeling and mitochondrial dysfunction in aortic-banded miniature swine. American Journal of Physiology. Heart and Circulatory Physiology 2010; 299(5): H1348-H1356.
- Guo S, Olm-Shipman A, Walters A, Urciuoli WR, Devito S and Nadtochiy SM: A cell-based phenotypic assay to identify cardioprotective agents. Circulation Research 2012; 110(7): 948-957.
- Gohil VM, Sheth SA, Nilsson R, Wojtovich AP, Lee JH and Perocchi F: Nutrient-sensitized screening for drugs that shift energy metabolism from mitochondrial respiration to glycolysis. Nature Biotechnology 2010; 28(3): 249-255.
- Brand MD and Nicholls DG: Assessing mitochondrial dysfunction in cells. Biochemical Journal 2011; 435(2): 297-312.
- Lefer DJ and Bolli R: Development of an NIH Consortium for Preclinical Assessment of Cardioprotective Therapies (CAESAR): A paradigm shift in studies of infarct size limitation. Journal of Cardiovascular Pharmacology and Therapeutics 2011; 16(3-4): 332-339.
- Chen Q and Lesnefsky EJ: Blockade of electron transport during ischemia preserves bcl-2 and inhibits opening of the mitochondrial permeability transition pore. FEBS Letters 2011; 585(6): 921-926.
- Elrod JW, Calvert JW, Morrison J, Doeller JE, Kraus DW and Tao L: Hydrogen sulfide attenuates myocardial ischemia-reperfusion injury by the preservation of mitochondrial function. Proceedings of the National Academy of Sciences 2007; 104(39): 15560-15565. Doi: 10.1073/pnas.0705891104
- Koene S and Smeitink J: Mitochondrial medicine: Entering the era of treatment. Journal of Internal Medicine 2009; 265(2): 193-209.
How to cite this article:
Hassanpour SH and Karami SZ: Study of respiratory chain dysfunction in heart disease. Int J Life Sci & Rev 2017; 3(10): 100-15. doi: 10.13040/IJPSR.0975-8232.IJLSR.3(10).100-15.
All © 2015 are reserved by International Journal of Life Sciences and Review. This Journal licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.
Article Information
1
100-115
842
1402
English
IJLSR
S. H. Hassanpour* and S. Z. Karami
Young Researchers and Elite Club, Yasooj Branch, Islamic Azad University, Yasooj, Iran.
Dr.hossein1366@yahoo.com
02 October 2017
22 October 2017
26 October 2017
10.13040/IJPSR.0975-8232.IJLSR.3(10).100-15
31 October 2017