SYNTHESIS OF SOME NOVEL 2-SUBSTITUTED IMINO-7-SUBSTITUTED PHENYL-3, 7-DIHYDRO-2H-[1, 3] THIAZOLO [5, 4-E] – THIAZIN-5-AMINE AS ANTIFUNGAL ACTIVITY
HTML Full TextSYNTHESIS OF SOME NOVEL 2-SUBSTITUTED IMINO-7-SUBSTITUTED PHENYL-3, 7-DIHYDRO-2H-[1, 3] THIAZOLO [5, 4-E] - THIAZIN-5-AMINE AS ANTIFUNGAL ACTIVITY
B. Chaubey 1, Vineet K. Singh * 2, D. Singh 1, Rajeev K. Verma 2 and Vimal K. Yadav 2
Synthetical Organic Research Lab, Department of Chemistry, T.D.P.G. College 1, Jaunpur - 222002, Uttar Pradesh, India.
Prasad Institute of Technology 2, Jaunpur - 222002, Uttar Pradesh, India.
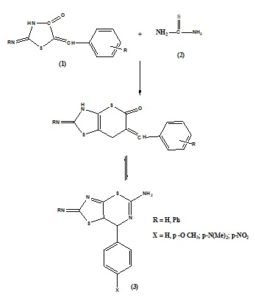
ABSTRACT: The condensation of 2-imino-5 substituted benzylidene-4-thiazolidinone (1) with thiourea (2) in the presence of glacial acetic acid to gives 2-substituted imino-7-substituted phenyl-3,7-dihydro-2H-[1,3] thiazolo [5,4-e]- thiazin-5-amine (3). The antifungal activities of the compounds (3a-h) were tested against A.niger and A. flavus. The growth of both fungi A. niger and A. flavus are inhibited to some extent by all the synthesized compounds. Hence all are antifungal agents.
Keywords: Thiazole, Thiazole derivatives, Substituted Thiazine derivatives, Antifungal activity
INTRODUCTION: Heterocyclic compounds comprise the major family of organic compounds. These are enormously essential with a wide range of synthetic, pharmaceutical, and industrial applications and are famous for their biological activities. There is an extensive spectrum of biological activities shown by many compounds containing five-membered heterocyclic rings in their structure. The high therapeutic properties of these heterocycles have encouraged the medicinal chemists to synthesize a large number of novel chemotherapeutic agents. These heterocyclic compounds have broadened the scope in remedying various dispositions in clinical medicines. Thiazoles have been reported to show pharmacological activities 1.
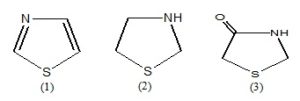
Thiazole (1) is a well known heterocyclic compound with two heteroatoms, sulfur, and nitrogen at position 1 and 3. The tetrahydro derivatives of thiazole are known as thiazolidine (2), and oxo-derivative of thiazolidine is called thiazolidinone (3). Thiazolidinone is an important compound of this group.
Thiazoles represent an important class of heterocyclic compounds. The thiazole derivatives are widely used in the field of medicine, industry and agriculture. The derivatives of thiazole have been reported as fungicides 2-3, Pesticides 4, insecticides 5, antibacterial 6-7, anti-inflammatory 8, anticonvulsant 9, antitubercular 10, antiviral 11, and anticancer 12. An attempt is to synthesize some novel 2-substituted imino-7-substituted phenyl - 3, 7-dihydro-2H-[1, 3] thiazolo [5, 4-e] - thiazin-5-amine. The synthesized compounds (3) have been screened for antifungal activity against A. niger and A. flavus.
MATERIALS AND METHODS: Melting points were taken in open capillary tubes and are uncorrected. The IR spectra were recorded in KBr on Perkin-Elmer-720 spectrophotometer. The 1H NMR spectra were recorded in CDCl3 on Varian A-60 D. spectrophotometer. The chemical shifts are recorded in ppm downfield from TMS, which are used as an internal standard.
2-substituted imino-7-substituted phenyl-3, 7-dihydro-2H-[1, 3] thiazolo [5, 4-e] - thiazin-5-amine (3a-h) (General Method).
A 0.05 mol of 2-substituted imino-5-benzylidene-4-thiazolidinone and 0.05 mol thiourea was dissolved in 100 ml of glacial acetic acid by refluxing for 5 hours, it was allowed to cool and the solid obtained was filtered. The crude products were pressed firmly and recrystallized from ethanol: water. The physical data of all the synthesized compounds are given in Table 1.
TABLE 1: PHYSICAL DATA OF ALL THE SYNTHESIZED COMPOUNDS
| Comp.
no |
R | X | M.P.(0C) | Yield
(%) |
Molecular
formula |
Elemental Analysis | IR
υcm-1 |
1H NMR
(CDCl3) |
||
| C | H | N | ||||||||
| 3a | H | H | 189 | 65 | C11H10N4S2 | 50.38 | 3.81 | 21.37 | 3390(NH), 760 (C-S), 1680 (C=N), 3080 (Ar-CH) | 7.19-8.25 (m,5H,Ar-H), 5.71 (s,4H,NH),2.81 (s,1H,CH) |
| 3b | H | p-OMe | 177 | 62 | C12H12N4S2O | 49.31 | 4.10 | 19.18 | 3380 (NH), 780 (C-S), 1673 (C=N), 3075 (Ar-CH), 1250 (C-OCH3) | 7.20-8.24 (m,4H,Ar-H), 5.461 (s,4H,NH), 2.69 (s,1H,CH), 3.53 (s,3H,OCH3) |
| 3c | H | p-N(Me)2 | 162 | 64 | C13H15N5S2 | 51.14 | 4.91 | 22.95 | 3383 (NH), 778 (C-S), 3079 (Ar-CH) | 7.22-8.26 (m,4H,Ar-H), 5.61 (s,4H,NH), 2.7 (s,1H,CH), 3.79 (s.6H,NMe2) |
| 3d | H | p-NO2 | 159 | 73 | C11H9N5S2O2 | 42.99 | 2.93 | 22.80 | 3370 (NH), 775 (C-S), 3101 (Ar-CH), 1470 (C-NO2) | 7.26-8.35 (m,4H,Ar-H),5.48 (s,4H,NH),2.75(s,1H,CH) |
| 3e | Ph | H | 197 | 72 | C17H14N4S2 | 60.35 | 4.14 | 16.56 | 3365 (NH), 770 (C-S), 3098 (Ar-CH) | 7.23-8.36 (m,16H,Ar-H),5.52 (s,3H,NH),2.68(s,1H,CH) |
| 3f | Ph | p-OMe | 186 | 66 | C18H16N4S2O | 58.69 | 4.34 | 15.21 | 3349 (NH), 772 (C-S), 3083 (Ar-CH), 1265 (C-OCH3) | 7.25-8.37 (m,9H,Ar-H),5.56 (s,3H,NH),2.73(s,1H,CH), 3.53 (s,3H.OCH3) |
| 3g | Ph | p-N(Me)2 | 178 | 64 | C19H19N5S2 | 59.84 | 4.98 | 18.37 | 3384 (NH), 762 (C-S), 3075 (Ar-CH) | 7.24-8.31 (m,9H,Ar-H),5.47 (s,3H,NH),2.67(s,1H,CH), 3.7 (s,6H,N-Me2) |
| 3h | Ph | p-NO2 | 164 | 71 | C17H13N5S2O2 | 53.26 | 3.39 | 18.27 | 3382 (NH), 765 (C-S), 3062 (Ar-CH), 1435 (C-NO2) | 7.27-8.35 (m,9H,Ar-H),5.54 (s,3H,NH),2.72 (s,1H,CH) |
Antifungal Activity: The compounds (3a-h) were screened for their antifungal activity against A. niger and A. flavus by known method 13 at the three concentrations viz. 1000, 100 and 10 ppm. The screening data of compounds are listed in Table 2. Results were compared with commercial fungicide miconazole tested under similar conditions.
The percentage inhibition has been calculated by the formula
% inhibition = (C-T) × 100/C
Where C and T are a diameter (in mm) of fungus colony in control and treated plates respectively.
TABLE 2: SCREENING DATA OF COMPOUNDS
| Comp.
no. |
R | X | A. niger Concentration (ppm) used | A. flavus Concentration (ppm) used | ||||
| 1000 | 100 | 10 | 1000 | 100 | 10 | |||
| 3a | H | H | 69.7 | 53.2 | 35.7 | 67.2 | 56.5 | 32.1 |
| 3b | H | p-OMe | 67.9 | 51.8 | 36.3 | 68.3 | 56.8 | 33.2 |
| 3c | H | p-N(Me)2 | 66.5 | 53.8 | 35.4 | 67.1 | 56.3 | 32.0 |
| 3d | H | p-NO2 | 73.2 | 57.6 | 39.0 | 70.0 | 57.1 | 34.8 |
| 3e | Ph | H | 67.1 | 56.5 | 32.0 | 64.3 | 50.1 | 32.8 |
| 3f | Ph | p-OMe | 67.2 | 53.5 | 34.0 | 65.9 | 52.3 | 34.2 |
| 3g | Ph | p-N(Me)2 | 65.8 | 51.4 | 32.1 | 64.7 | 52.1 | 33.8 |
| 3h | Ph | p-NO2 | 70.8 | 57.3 | 36.7 | 72.3 | 53.4 | 34.6 |
SCHEME 1
RESULT AND DISCUSSIONS: It is observed from the antifungal screening data that most of the compounds have significant toxicity at 1000 ppm, but their toxicity decreases upon dilution Table 2. The antifungal activity of the compounds is due to the presence of biolabile groups N=C-S, and N-N=C present in the nuclei of the compound and slight variation in their antifungal activity is due to OCH3, N-(Me)2 and NO2 group attached with benzene nucleus. All the synthesized compounds show antifungal activity against A. niger and A. flavus.
CONCLUSION: The growth of both fungi A. niger and A. flavus are inhibited to some extent by all the synthesized compounds. The compounds having a p-nitro group in each set of derivatives are more active. The compounds having p-nitrophenyl and p-methoxy phenyl groups in thiazinopyrazole enhanced the fungicidal activity against both the test fungi A. niger and A. flavus.
ACKNOWLEDGEMENT: The authors are very grateful to Dr. U. P. Singh (Principal), Dr. Daroga Singh (Ex-Head), Department of Chemistry T.D.P.G. College, Jaunpur for providing necessary facilities.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Gupta V and Kant V: A review on biological activity of imidazole and thiazole moieties and their derivatives. Science International 2013; 1: 253-60.
- Narayana B, Raj KKV, Ashalatha BV, Kumari NS and Sarojini BK: Synthesis of some new 5-(2-substituted-1, 3-thiazol-5-yl)-2-hydroxy benzamides and their 2-alkoxy derivatives as possible antifungal agents. European Journal of Medicinal Chemistry 2004; 39: 867-72.
- Mahapatra GN: Non metallic fungicides. Natue 1956; 177: 938.
- Kennedy AD and Summers AL: Chemical constitution and activity of herbicides. Journal of Heterocyclic Chemistry 1981; 18: 409.
- Holla BS, Prasanna CS, Poojary B, Rao KS, Shridhara K and Bhat UG: Synthesis and insecticidal activity of some 1, 3, 4-oxadiazoles derived from 2-chloropyridine-5-acetic acid. Indian Journal of Chemistry 2004; 43(4): 864-8.
- Tiwari N and Nizamuddin: Synthesis of some 1-aroyl-3-methyl-4-substituted-phenyl-6-imino-4, 7-dihydro-1, 3-thiazino-5, 4-pyrazoles as pesticides. Journal of Indian Chemical Society 1991; 68: 144-46.
- Desai NC, Parekh BR and Thaker AJ: Preparation of some important medicinal compounds. Thiosemicarbazones, thiadiazolines, 4-thiazolidinones and 5-arylidine derivatives as antibacterial and tuberculostatic agents. Indian Chemical Society 1987; 64(8): 491.
- Geronikaki A, Litina DH, Zablotskaya A and Segal I: Organosilicon-containing thiazole derivatives as potential lipoxygenase inhibitors and anti-inflammatory agents. Bioinorganic Chemistry and Applications 2007: 1-7.
- Kshirsagar A, Turaskar MP, Kulkarni VM, Dhanashire S and Kadam V: Microwave Assisted Synthesis of Potential Anti Infective and Anticonvulsant Thiosemicarbazones. International Journal of Chemical Technology and Research 2009; 1: 696.
- Karali N, Giirsoy A, Kandemirli F, Shvets N, Kaynak , Ozbey S, Kovalishym V and Dimoglo A: Synthesis and structure-antituberculosis activity relationship of 1H-indole-2,3-dione derivatives. Bioorganic Medicinal Chemistry 2007; 15: 5888.
- Rawat R, Tripathi R, Katti SB, Pannecouque C and Declereq E: Design, synthesis, and evaluation of 2-aryl-3-heteroaryl-1, 3-thiazolidin-4-ones as anti-HIV agents. Bioorganic Medicinal Chemistry 2007; 15: 1725.
- Gududura V, Hurth E, Dalton JT and Miller DD: Discovery of 2-arylthiazolidine-4-carboxylic acid amides as a new class of cytotoxic agents for prostate cancer. Journal of Medicinal Chemistry 2005; 48: 2584.
- Horsfall JG: Quantitative bioassay of fungicides in the laboratory. Botanical Review 1945; 11(7): 357-97.
How to cite this article:
Chaubey B, Singh VK, Singh D, Verma RK and Yadav VK: Synthesis of some novel 2-substituted imino-7-substituted phenyl-3,7-dihydro-2h-[1, 3]thiazolo[5, 4-e]-thiazin-5-amine as antifungal activity. Int J Life Sci & Rev 2015; 1(1): 20-22. doi: 10.13040/IJPSR.0975-8232.IJLSR.1(1).20-22.
All © 2015 are reserved by International Journal of Life Sciences and Review. This Journal licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.
Article Information
4
20-22
563
1986
English
IJLSR
B. Chaubey *, V. K. Singh, D. Singh, R. K. Varma and V. K. Yadav
Prasad Institute of Technology, Azamgarh Byepass Road Punchhatia, Jaunpur, Uttar Pradesh, India.
mailme2805@gmail.com
21 May 2014
28 July 2014
04 December 2014
http://dx.doi.org/ 10.13040/IJPSR.0975-8232.IJLSR.1(1).20-22
01 January 2015