SYNTHESIS AND CHARACTERISATION OF (MSNs) MESOPOROUS SILICA NANOPARTICLES
HTML Full TextSYNTHESIS AND CHARACTERISATION OF (MSNs) MESOPOROUS SILICA NANOPARTICLES
Surabhi Rashi * 1, Devendra Singh 1, Shashi Alok 1, Upendra Sharma 1, Dilip Kumar Chanchal 1 and Akhilesh Mishra 2
Institute of Pharmacy 1, Bundelkhand University, Jhansi - 284128, Uttar Pradesh, India.
Smt. Vidyawati College of Pharmacy 2, Jhansi - 284128, Uttar Pradesh, India.
ABSTRACT: Mesoporous silica nanoparticles are one of the widely used as carriers for drugs classified for different categories like NSAIDs, cancer, etc. They are synthesized by using four major materials, which include a surfactant, solvent, a silica source, and acidic or basic catalyst. By step by step, the addition of all MSNs is formed. The main part in synthesis is played by room temperature and particle size with its stability in solid form. They are characterized by using different instruments, which include ultraviolet (UV), FT-IR (Fourier transform- Infrared), SEM (Scanning Electron Microscopy).
Keywords: MSNs, Ibuprofen, Synthesis, Characterization
INTRODUCTION: Silicon is the second abundant element in the earth’s crust after oxygen. Silicon occurs as oxygen-containing compound termed Silicon dioxide (SiO2), which is known as silica. The first mesoporous silica nanoparticles (MSN) were developed by Mobil Oil Corporation (Mobil) scientists in 1992. This was named M41S series (the Mobil 41Series), their pore diameters ranged from 15Å to 100Å 1, 2. Mesoporous silica nanoparticles (MSNs) have been intensively explored in materials research due to their unique properties, such as high surface areas, large pore volumes, tunable pore sizes with a narrow distribution and tunable particle diameters 3, 4.
MSNs with controlled properties can be extended to a wide range of applications, such as drug delivery, catalysts supports, adsorption and separation of proteins, cell imaging, cell labeling, enzyme adsorption and immobilization 5. More specifically, the potential utilization of MSN materials in medical and pharmaceutical drug delivery systems is well documented 6-14.
The suitability of MSN for important biotechnological and biomedical applications is also due to their small size, which allows facile endocytosis by living animal and plant cells without any significant cytotoxicity. Mesoporous silica materials with different morphologies, such as sphere 15, fibers 16, film 17, and vesicles 18 have been developed.
However, one of the main and specific of drug delivery system using Mesoporous materials is the pore sizes which could not encounter all types of desired drugs which consist of bulky and different features.
Mesoporous silica structures have been known to materials scientists for over 40 years when the term was first coined to describe zeolite-silica gel mixtures with a well-defined and uniform porosity 19. It was not until 1992, following the near-simultaneous discovery of organic-templated mesoporous silicas by two groups of scientists that researchers outside the field of materials engineering and petrochemicals began to take notice of these unique materials 20, 1. The idea that mesoporous silica nanoparticles (MSNs) could be used as drug delivery devices would have to wait another 6 years until 199 when a patent was filed by Muller, Reck, and Rose stating that mesoporous silicates might contain pharmacologically active substances 21 and again speculated upon by Schuth and colleagues 22. The first account of MSNs with the ability to release a drug molecule was published in 2001 by Balkus and colleagues using the material known as Dallas Amorphous Material-1 (DAM-1) 23.
MSN of various types and exhibiting different properties have previously been synthesized via different methods, such as plasma synthesis chemical vapor deposition, microemulsion processing, combustion synthesis, sol‐gel, hydrothermal techniques, etc. Recent efforts for the preparation of nanoparticles are focused on controlling size, morphology and surface reactivity of nanoparticles 24- 34.
The Sol-gel method has been widely used and a method of choice for the preparation of nanoparticles, as it has several advantages such as synthesis may be carried out at low temperature, desired pH to yield high purity and also, the reaction kinetics of the process may be controlled by varying the composition of the reaction mixture. Mainly four ingredients are necessary for the formation of mesoporous silica materials: a surfactant, silica source, an acid or base catalyst and a solvent like ethanol or water.
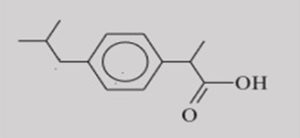
Ibuprofen (IBU) [(±)-2-(4-isobutylphenyl)] propionic acid (I) is an orally administered drug belongs to a class of non-steroidal anti-inflammatory drugs (NSAIDs), which are used to reduce the pain and swelling. It is considered to be the prototype for the family of synthetic 2 -aryl propionic acids, profens, a sub-class of the non-steroidal antiinflammatory drugs (NSAIDS). In recent, they are used to treat arthritis, muscular strain, cephalalgia, and others. Some of the available profen drugs are ibuprofen, naproxen, ketoprofen, and flurbiprofen 35. Ibuprofen is distributed over the counter and belongs to the top-ten of drugs marketed worldwide in 1989. IBU is a poorly water-soluble drug 36.
FIG. 1: CHEMICAL STRUCTURE OF IBUPROFEN
It is used to relieve the symptoms of a wide range of illnesses such as headaches, backache, period pain, dental pain, neuralgia, rheumatic pain, muscular pain, migraine, cold and flu symptoms and arthritis 37. Ibuprofen has only 2 hr as biological half-life; this shorter biological half-life makes it a suitable drug for sustained or controlled drug delivery system development. Therefore, ibuprofen has been commonly employed as a model drug in the development of sustained/controlled releases further to this its structural feature makes it easy to accommodate into the MSNs 38.
In the present work, we studied the preparation of MSNs by using Tween 20 and 80 as surfactants and ibuprofen as a drug to be carried.
Experimental:
Materials: A surfactant such as Tween 20 (Polyoxyethylene (20) sorbitan monolaurate) was obtained from HiMedia laboratories Ltd, Mumbai, Tween 80 (Polyoxyethylene (20) sorbitan monooleate was obtained from s.d. fine- chem. Ltd., Mumbai. Silica source which is Tetraethylorthosilicate (TEOS) was obtained from Yarrowchem products, Mumbai. Sodium hydroxide (NaOH) was obtained from Hi-Tech Laboratories, Delhi. Deionized water was obtained from HiTech Laboratories, Delhi and distill water was obtained from the Innovation center, Bundelkhand University, Jhansi.
Synthesis of MSNs: General sol-gel method was employed for the synthesis of Mesoporous silica nanoparticles. MSNs were prepared by dissolving Tween 20 and Tween 80 (11g) in deionized water (490 ml), then 2M NaOH (13.5g) added at 80 °C with stirring. TEOS (15 g) was added dropwise in vigorous stirring solution at 80°C for 4 h.
FIG. 2: VOLUMETRIC FLASK CONTAINED MSNs IN SOLUTION FORM
Cooled the solution formed and filtered, then washed with ethanol (10 ml) many times.
FIG. 3: FILTERED AND WASHED
Then it was dried in a vacuum oven (63°C). For surfactant removal, calcination process at 540°C was done.
Loading of the drug in MSNs: Ibuprofen (150 mg) was dissolved in ethanol (10 ml), and to that solution, dried Mesoporous silica sample (150 mg) was added. Ibuprofen loaded MSNs were recovered by filtration followed by washing with ethanol and dried for 24 hours at 40 °C.
FIG. 4: DRIED MSNs WITH DRUG
Characterization: Here we used Ultra-Violet spectroscopy (UV) Varioskan Flash 4.00.53 (Innovation center, BU, Jhansi) to determine the λmax of samples and obtain the calibration curve of IBU. Solartron Analytical FT-IR spectrometer with model number 1255B (IIT, Kanpur) for recording infrared (FT-IR) spectra of the samples. Scanning electron microscopy (SEM) Carl Zeiss EV018 (IIT, Kanpur) used to study the morphology of the MSNs synthesized for which sample preparation was done by dispersing a little amount of sample in water. A thin film of the homogenous sample solution was deposited and air dried on the reflective surfaces of silicon wafer for SEM analysis.
RESULT AND DISCUSSION:
Microscopy Observation: SEM image discloses the particle diameter of MSNs. Particle diameter was 200 μm.
FIG. 5: SEM IMAGE SHOWING PARTICLE DIAMETER
λmax of the pure drug as well as ibu loaded nanoparticle were taken by UV spectroscopy. The drug-loaded silica shows similar absorbance spectrum with ibuprofen in liquid media at the peak around 264 nm.
FIG. 6: UV SPECTRA OF PURE IBU WITH MSN LOADED IBU
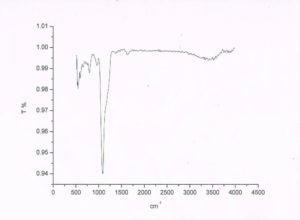
FT-IR spectra represent the MSNs loaded drug. The ibu loaded silica in this figure the bands at around 1720 cm-1 is proved the presence of carboxyl group vibration, and the vibration bands from 2950–2850 cm-1 is proved the presence of C–H stretching vibrations of Ibu. These bands provide the information of ibuprofen loading into the silica. Remaining bands from 795 and 465 cm-1 were attributed to Si–O–Si, and the band at 964 cm-1 is for Si–OH.
FIG. 7: IR SPECTRA
CONCLUSION: In our present study, we synthesized Mesoporous silica nanoparticles with tween 20 and 80 as surfactant and ibuprofen as a drug for loading. The nanoparticles obtained were in Mesoporous silica nanoparticles range, i.e., 50 – 500 nm and the wavelength at which peaks were obtained of pure drug and MSN loaded with the drug was 264 nm. So, we can say that the MSNs formed by using Tween 20 and 80 with Ibuprofen as a drug to be loaded was successful.
ACKNOWLEDGEMENT: We are grateful to Guide Mr. Devender Singh, Assistant Professor, Institute of Pharmacy, Bundelkhand University, for his assistance and encouragement. We extend our sincere thanks to Mr. Akhilesh Mishra; Dr. Upendra Sharma, Assistant professor, Institute of Pharmacy, Bundelkhand University; Mr. Chetan Prakash, Librarian, Institute of Pharmacy, Bundelkhand University; Mr. Dilip Kumar Chanchal, for critically reading the manuscript and providing the valuable suggestions.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Beck JS, Vartuli JC, Roth WJ, Leonowicz ME, Kresge CT and Schmitt KD: A new family of Mesoporous molecular sieves prepared with liquid crystal templates. J Am Chem Soc 1992; 114(27): 10834-43.
- Kresge CT, Leonowicz ME, Roth WJ, Vartuli JC and Beck JS; Ordered Mesoporous molecular sieves synthesized by a liquid- crystal template mechanism. Nature 1992; 359: 710-12.
- Bottini M, Annibale FD, Magrini A, Cerignoli F, Arimura Y, Dawson MI, Bergamaschi E, Rosato N, Bergamaschi A and Mustelin T: Quantum dot-doped silica nanoparticles as probes for targeting of T-lymphocytes. Int J Nanomed 2007; 2: 227-33.
- Gerion D, Herberg J, Bok R, Gjersing E, Ramon E, Maxwell R, Kurhanewicz J, Budinger TF, Gray JW, Shuman MA and Chen FF: Paramagnetic silica-coated nanocrystals as an advanced MRI contrast agent. J Phy Chem 2007; C111: 12542-51.
- Cauda V, Muhlstein L, Onida B and Bein T: Tuning drug uptake and release rates through different morphologies and pore diameters of confined Mesoporous silica. Micropor Mesopor Mat 2009; 118: 435-42.
- Courtney RT, Daniel PF, Jae-Hyun L, Eunjoo C, MiHyeon C, Eun SK, Stoddart JF, Jeon-Soo S, JinWoo C and Jeffery IZ: Nonvasive remote-controlled release of drug molecules in vitro using Magnetic actuation of mechanized nanoparticles. J Am Chem Soc 2010; 132: 10623-25.
- Speybroeck MV, Mellaerts R, Mois R, Thi TD, Martens JA, Humbeeck JV, Annaert P, Mooter GV and Augustijns P: Enchanced absorption of the poorly soluble drug fenofibrate by tunning its release rate from ordered Mesoporous silica; Eur J Pharm Sci 2010; 41: 623-30.
- Vallet-Regi M: Nanostructured Mesoporous silica matrices in Nanomedicines. J Int Med 2009; 267: 22-43.
- Popovici RF, Seftel EM, Mihai GD, Popvici E, Voicu VA: Controlled drug delivery system based on ordered Mesoporous inhibitor drug. J Pharm Sci 2010; 100(2): 704-14.
- Leirose GDS and Cardoso MB: Silica- maltose composites: Obtaining drug carrier systems through tailored ultrastructural nanoparticles. J Pharm Sci 2011; 100(7): 2826-34.
- Limnel T, Santos HA, Makila E, Heikkila T, Salonen J, Murzin DY, Kumar N, Laaksonen T, Peltonen L and Hirvonen J: Drug delivery formulation of ordered Mesoporous silica: Comparison of three drug loading methods. J Pharm Sci. DOI.10.1002/jps. 22577, 2011.
- Zhu CL, Lu CH, Song XY, Yang HH and Wang XR: Bioresponsive controlled release using Mesoporous silica nanoparticles capped with Aptamer-based molecular gate. J Am Che Soc 2011; 133: 1278-81.
- Shen SC, Ng WK, Chia L, Hu J and Tan RBH: Physical state and dissolution of ibuprofen formulated by co-spray drying with Mesoporous silica: Effect of pore and particle size. Int J Pharm 2011; 410: 188-95.
- Vivero-Escoto JL, Slowing II, Trewyn BG and Lin VSY: Mesoporous silica nanoparticles for Intracellular controlled drug delivery. Small 2010; 6(18): 1952-67.
- Qi LM, Ma JM, Cheng HM and Zhao ZG: Micrometer sized Mesoporous silica spheres grown under static condition. Chem Mater 1998; 10, 1623-26.
- Huo QS, Zhao DY, Feng JL, Weston K, Buratto SK., Stuck GD, Schacht F and Schuth F: Room temperature growth of Mesoporous silica fibers: a new high- surface area optical waveguide. Adv Mater 1997; 9: 974-78.
- Zhao DY, Yang PD, Melosh N, Feng JL, Chmelka BF and Stucky GD: Continuous Mesoporous silica films with highly ordered large pore structures. Adv Mater 1998; 10, 1380-85.
- Li YS, Shi JL, Hua ZL, Cheng HR, Ruan ML and Yan DS: Hollow spheres of Mesoporous aluminosilicate with a three- dimensional pore network and extraordinarily high hydrothermal stability. Nano. Lett 2003; 3: 609-12.
- Mieville RL: Measurement of microporosity in the presence of mesopores. J Colloid Interface Sci 1972; 41(2): 371-73.
- Inagaski S, Fukushima Y, Okada A, Kato C and Kuroda K: Toyota Central Research and Development Laboratories, Inc., Japan. Assign. Manufacture of layer-form silica-metal oxide porous intercalation compounds useful as adsorbents and catalysts patent JPO4238810A. 1992.
- Mulle U, Reck B and Roser J: Basf Aktiengesellschaft, Germany. Assignee. Mesoporous silica and its preparation for use as catalyst or supports for cataltst, drugs, enzymes or pigments patent EP831059A1, 1998.
- Schuth F, Ciosla U, Schacht S, Thieme M, Huo Q and Stucky G: Ordered mesporous silicas and zirconias: control on length scales between nanometer and micrometer. Mat Res Bull 1999; 34(3): 483-94.
- Balkus KJ, Coutinho D, Lucas J and Washmon-Kriel L: Synthesis and characterization of DAM-1 type materials. Mat Res Soc Sympo Proc 2001; 628. (Organic/Inorganic Hybrid Materials): CC107.1-CC.7.6.
- Igo IS, Juan LVE, Chia-Wen W and Victor SYL: Mesoporous silica nanoparticles as controlled release drug delivery and gene transfection carriers. Adv Drug Deli Rev 2008; 60: 1278-88.
- Gu J, Fan W, Shimojima A and Okubo T: Organic-inorganic Mesoporous nanocarriers integrated with biogenic ligands. Small 2007; 3: 1740-44.
- Slowing II, Trewyn BG and Lin VSY: Mesoporous silica nanoparticles for intracellular delivery of membrane-impermeable proteins. J Am Chem Soc 2007; 129: 8845-49.
- Naik SP, Elangovan SP, Okubo T and Sokolov I: Morphology control of Mesoporous silica particles. J Phys Chem 2007; C111: 11168-73.
- Yoo WC and Stein A: Solvent effects of morphologies of Mesoporous silica spheres prepared by pseudomorphic transformations. Chem Mater 2011; 23: 1761-67.
- Niu D, Ma Z, Li Y and Shi J: Synthesis of core-shell structured dual-mesoporous silica spheres with tunable pore size and controllable shell thickness. J Am Chem Soc 2010; 132: 15144-147.
- Borisova D, Mohwald H and Shchukin DG: Mesoporous silica nanoparticles for active corrosion protection. ACS NANO 2010; 5(3): 1939-46.
- Suteewong T, Sai H, Cohen R, Wang S, Bradbury M, Baird B, Gruner SM and Wiesner U: Highly animated Mesoporous silica nanoparticles with cubic pore structure. J Am Chem Soc 2011; 133: 172-75.
- Yokoi T, Sakamoto Y, Terasaki O, Kubota Y, Okubo T and Tatsumi T: Periodic arrangement of silica nanospheres assisted by amino acids. J Am Chem Soc 2006; 128: 13664-65.
- Mbaraka IK, McGuire KJ and Shanks BH: Acidic Mesoporous silica for the catalytic conversion of fatty acids in beef tallow. Ind Eng Che Res 2006; 45: 3022-28.
- Graf C, Dembski S, Hofman A and Ruehl E; A general method for the controlled embedding of nanoparticles in silica colloids. Langmuir 2006; 22: 5604-10.
- Lee X: Appendix attempts towards the synthesis of ibuprofen 2012.
- Janjikhel RK and Adeyeye CM: Dissolution of ibuprofen enantiomers from coprecipitates and suspensions containing chiral excipients. Pharm Dev Technol 1999; 4(1): 9-17.
- Ahmadi A Danialia M, Kazemia S, Azamia S and Alizadea N: Synthesis of ibuprofen with modified and economical process as an NSAID drug. J App Chem Res 2014; 8(3): 91-95.
- Ganesh M and Lee SG: Synthesis, characterization and drug release capability of new cost-effective Mesoporous silica nanoparticles for Ibuprofen drug delivery; International Journal of Control and Automation 2013; 6(5): 207-16.
How to cite this article:
Rashi S, Singh D, Alok S, Sharma U, Chanchal DK and Mishra A,: Synthesis and characterisation of (MSNs) Mesoporous Silica Nanoparticles. Int J Life Sci & Rev 2016; 2(8): 133-37. doi: 10.13040/IJPSR.0975-8232.IJLSR.2(8).133-37.
All © 2015 are reserved by International Journal of Life Sciences and Review. This Journal licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.
Article Information
2
133-137
659
1627
English
IJLSR
S. Rashi *, D. Singh, S. Alok, U. Sharma, D. K. Chanchal and A. Mishra
Department of Pharmaceutics, Institute of Pharmacy, Bundelkhand University, Jhansi, Uttar Pradesh India.
surabhir2014@gmail.com
11 April 2016
16 May 2016
28 June 2016
10.13040/IJPSR.0975-8232.IJLSR.2(8).133-37
31 August 2016